Insulin resistance and chronic disease share a connection that conventional medicine rarely explains clearly — yet understanding it may be the single most important shift in how you think about your long-term health. Insulin resistance is not just a diabetes problem. It is the central upstream mechanism behind fatty liver, cardiovascular disease, cognitive decline, chronic inflammation, hormonal disruption, and metabolic syndrome — often developing silently for a decade or more before any single diagnosis is made.
If there’s one metabolic glitch that quietly reshapes the whole body, it’s insulin resistance.
Not because insulin is “bad.” Insulin is a survival hormone: it helps you store fuel, build tissue, and keep blood sugar in a safe range. The problem starts when the body is forced into chronic fuel surplus and chronic insulin signaling, and tissues begin to “push back.”
That pushback is insulin resistance — and it’s the common thread connecting what look like separate diagnoses:
- type 2 diabetes
- fatty liver
- PCOS
- hypertension
- dyslipidemia patterns
- gout
- sleep apnea
- neurodegeneration risk
- and a large slice of cardiovascular disease
This is why you’ll see it show up again and again across this entire platform.
Chronic insulin signaling disrupts metabolic flexibility
The most important nuance
Insulin resistance is often an adaptive response, not a character flaw and not a simple “sugar disease.”
When cells are already overloaded with energy (especially in the wrong places), becoming less responsive to insulin is one way the body tries to limit further fuel storage. The tragedy is that the compensation for insulin resistance is usually more insulin (hyperinsulinemia) — and that drives many downstream problems for years before glucose ever becomes “abnormal.” PubMed+2PubMed+2
The “sequence” that matters: insulin rises first
A classic pattern:
- Tissues become less insulin sensitive (especially liver and muscle)
- The pancreas compensates by secreting more insulin
- Blood sugar can stay “normal” for a long time
- Over years, the system breaks: beta-cells can’t keep up → glucose rises → diagnosis
This is not a controversial idea; it’s core physiology and a major reason why relying only on fasting glucose misses early disease. PubMed+1
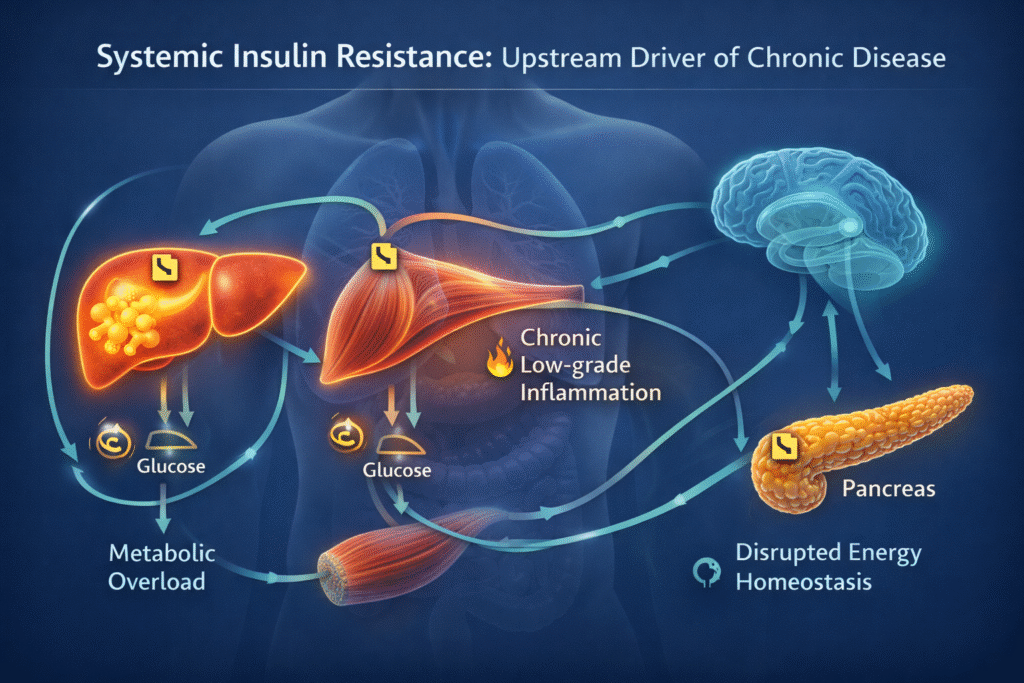
The two main battlegrounds: liver and muscle
Insulin resistance is not uniform. It’s tissue-specific.
1) Liver insulin resistance
Normally, insulin tells the liver: “stop making glucose.”
With liver insulin resistance, the liver keeps releasing glucose — especially overnight — contributing to high fasting glucose and elevated A1c.
See also The Gut-Liver Axis: Why the Liver “Feels” the Gut First
Also: the liver becomes a factory for VLDL export when it’s stuffed with fat → a common route to high triglycerides and fatty liver.
2) Muscle insulin resistance
Muscle is the main “sink” for glucose after meals. When muscle is insulin resistant, post-meal glucose runs higher, and insulin rises to compensate.
DeFronzo popularized how multiple organs get involved (the “ominous octet”), reminding us that “type 2 diabetes” is really multi-organ dysfunction, not just high sugar. PMC+1
See also Protein, Muscle, and Insulin – Why Most People Get This Wrong
What actually causes insulin resistance?
There are many contributors, but the best mechanistic work converges on a few repeatable “nodes.”
Node 1: Ectopic fat and toxic lipid signaling (the “wrong fat in the wrong place” problem)
One of the strongest mechanistic models is that insulin resistance is driven by ectopic lipid accumulation (fat inside liver and muscle cells), which creates lipid intermediates (like diacylglycerols) that interfere with insulin signaling pathways.
This isn’t “fat is bad.” It’s misplaced fuel. The details here matter because it explains why people can look different on the outside and still have severe metabolic dysfunction on the inside.
If you want the best deep mechanistic reading on this: Samuel & Shulman (JCI) and Petersen & Shulman’s physiology review are top-tier. JCI+1
Node 2: Mitochondrial overload and redox stress
When energy intake chronically exceeds what you can oxidize, mitochondria are forced into a high-pressure state (high NADH/NAD⁺ ratio, higher electron pressure), which raises oxidative stress and disrupts normal metabolic flexibility. That feeds back into insulin signaling dysfunction.
(We’ll connect this directly to your mitochondria pillar content — because insulin resistance is, in many cases, a fuel-handling disorder.)
Node 3: Inflammation and cellular stress pathways (often downstream, sometimes amplifying)
Inflammatory signaling and ER stress are real players, but they often ride on top of the fuel-overflow problem — they’re not always the first domino. Samuel & Shulman’s mechanistic review is excellent on how these pathways link together. PubMed+1
For the complete systems-biology framework, see our comprehensive insulin resistance overview.
Insulin resistance is the hub that makes “separate diseases” look connected
Here’s the practical map:
- Fatty liver (NAFLD / MAFLD): tightly linked with hepatic insulin resistance and VLDL overproduction
- High triglycerides + low HDL pattern: classic insulin-resistant lipid traffic
- Hypertension: sympathetic tone, renal sodium handling, endothelial effects — frequently clustering with IR
- PCOS: hyperinsulinemia drives ovarian androgen production and worsens ovulatory dysfunction
- “Normal glucose” but progressive fatigue, belly fat, cravings, brain fog: often the compensation phase (high insulin, normal glucose)
The metabolic syndrome concept grew precisely because these patterns co-travel around insulin resistance. PubMed+2ScienceDirect+2
How reliable is this science?
Very.
Not because “guidelines say so,” but because:
- the physiology is coherent (insulin’s known actions)
- the clinical pattern reproduces across populations
- mechanistic human studies show predictable defects in insulin signaling with ectopic lipid load
- interventions that reduce the fuel overflow (especially liver fat) can produce remission in many people
This doesn’t mean insulin resistance has only one cause — it doesn’t. But the centrality of IR as a disease hub is among the most stable concepts in modern metabolism research. Physiological Journals+2JCI+2
The hopeful part: this is often reversible (especially early)
Two clean lines of evidence (different methods, same reality):
1) Intensive dietary weight-loss intervention in primary care can drive remission
The DiRECT trial showed substantial remission at 12 months in a primary-care program, with durability data at 2 years. The Lancet+2PubMed+2
2) Carbohydrate restriction / nutritional ketosis models can markedly improve glycemia and medication use (with support)
Remote-care models using carbohydrate restriction have shown meaningful improvements in glycemic markers and medication reduction in peer-reviewed publications. PubMed+2Springer+2
Different strategies, same underlying point: when you reduce the metabolic pressure (fuel overflow + chronic insulin demand), the system can recover.
The lab reality: why “normal glucose” can still be metabolic disease
A practical way to say it:
Glucose is a late marker. Insulin is an early marker.
Many people sit for years in a state of:
- normal fasting glucose
- “borderline” A1c
- but high fasting insulin, high TG/HDL ratio, fatty liver, central adiposity, or rising blood pressure
And the body is already paying the price.
There’s also prospective evidence linking higher fasting insulin to higher cardiovascular risk in some cohorts (not perfectly in all studies, but enough that it should make clinicians take insulin seriously as a signal). JAMA Network+1
People Also Ask
What is insulin resistance?
Insulin resistance is a metabolic state in which cells respond less effectively to insulin, requiring higher insulin levels to manage blood glucose and fuel storage. It often develops years before blood sugar becomes abnormal.
Is insulin resistance the same as type 2 diabetes?
No. Insulin resistance usually precedes type 2 diabetes by many years. During this time, insulin levels rise to compensate while blood glucose can remain within the normal range.
What causes insulin resistance?
Insulin resistance is driven by chronic metabolic overload, ectopic fat accumulation in organs such as the liver and muscle, mitochondrial stress, and impaired fuel handling—not simply sugar intake or body weight.
Can insulin resistance be reversed?
In many cases, yes—especially in earlier stages. Reducing metabolic pressure through appropriate nutrition, movement, sleep, and circadian alignment can significantly improve insulin sensitivity.
Why can insulin resistance exist with normal blood sugar?
Because the pancreas compensates by producing more insulin. Blood glucose is a late marker of metabolic dysfunction, while insulin resistance and hyperinsulinemia develop much earlier.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Scientific References
- Reaven GM. Role of insulin resistance in human disease (Banting Lecture, 1988). PubMed
- Reaven GM. Role of insulin resistance in human disease (Syndrome X) (1993). PubMed
- DeFronzo RA. From the triumvirate to the ominous octet (Diabetes, 2009). PMC+1
- Samuel VT, Shulman GI. Mechanisms for insulin resistance: common threads and missing links (2012). PubMed
- Samuel VT, Shulman GI. The pathogenesis of insulin resistance (JCI, 2016). JCI
- Petersen MC, Shulman GI. Mechanisms of Insulin Action and Insulin Resistance (Physiol Rev, 2018). Physiological Journals
- Lean MEJ et al. DiRECT Trial, 12-month remission (Lancet, 2018). The Lancet+1
- Lean MEJ et al. DiRECT Trial, 2-year durability (2019). PubMed+2ScienceDirect+2
- Hallberg SJ et al. Continuous care / carbohydrate restriction model (2018). PubMed+1
- Lakka HM et al. Hyperinsulinemia and cardiovascular death risk (2000). JAMA Network
