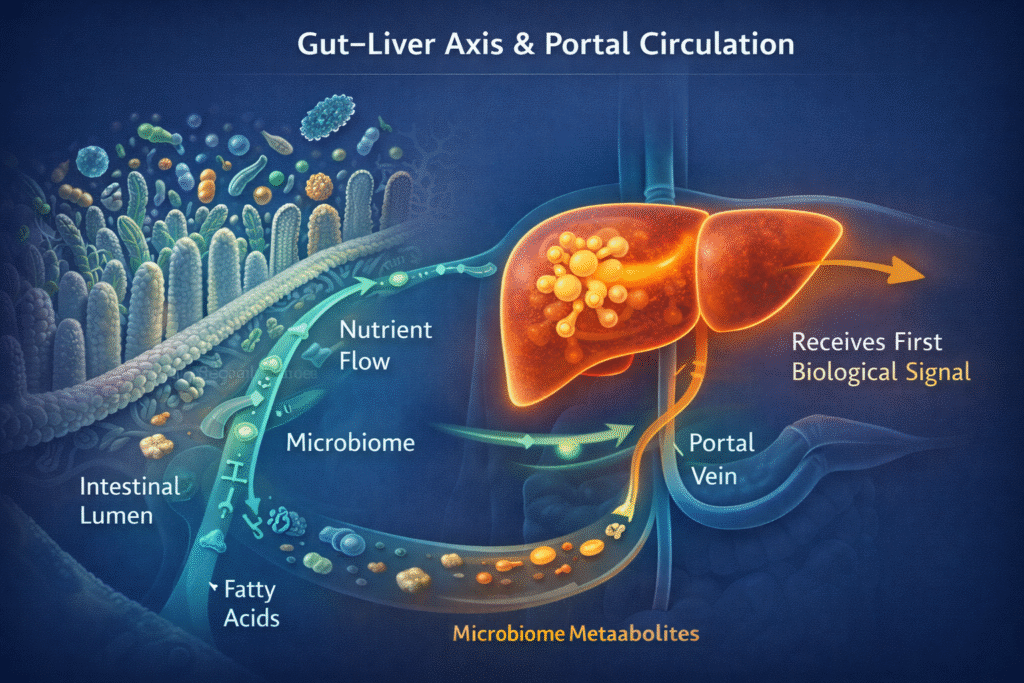
The gut health and liver connection is one of the most underappreciated relationships in metabolic medicine — and one of the most clinically relevant for anyone dealing with fatty liver, elevated liver enzymes, insulin resistance, or persistent digestive symptoms. Your liver does not exist in isolation. Through the portal vein, it receives every signal your gut produces — nutrients, bacterial metabolites, fatty acids, and inflammatory compounds — before that information reaches anywhere else in your body. What happens in your gut does not stay in your gut.
In this article we’ll break down the gut-liver axis and insulin resistance—and why the liver ‘feels’ the gut first.
Many people work hard on their metabolic health. They train. They prioritize protein. They reduce ultra-processed foods. They even lower carbohydrates.
And yet, some things refuse to normalize:
- fasting glucose stays elevated
- morning blood sugar spikes persist
- fatty liver shows up on imaging
- insulin resistance lingers despite “doing everything right”
At this point, the problem is often not muscle anymore.
To understand what’s going on, we need to move upstream—from muscle to the liver, and from the liver to the gut.
This is where the gut–liver axis becomes essential.
The liver’s unique role in metabolic control
The liver is not just another metabolic organ.
It occupies a strategic anatomical and physiological position.
Everything absorbed from the gut—nutrients, metabolites, bile acids, microbial products—travels first through the portal vein directly to the liver before entering the systemic circulation.
This means the liver is:
- the first metabolic processor of what you eat
- the first immune sensor of gut-derived signals
- a central regulator of glucose, fat, and inflammation
No other organ sits at this crossroads.
So when metabolic dysfunction persists, it often shows up in the liver first, even before muscle or fat tissue fully break down.
Hepatic insulin resistance is different from muscle insulin resistance
A crucial distinction is often missed:
Insulin resistance is not one uniform condition.
Muscle insulin resistance
- Impairs glucose uptake after meals
- Leads to higher post-meal blood sugar
- Improves strongly with resistance training and activity
Hepatic (liver) insulin resistance
- Impairs insulin’s ability to suppress glucose production
- Leads to elevated fasting glucose, especially overnight and in the early morning
- Drives the “dawn phenomenon”
- Often persists even when diet and exercise look “correct”
This explains a common clinical paradox:
Someone can eat low-carb, train regularly, and still wake up with high fasting glucose.
That is not a willpower issue.
It is a liver signaling issue.
What the gut–liver axis actually means (no buzzwords)
The gut–liver axis is not a single pathway and not a diagnosis.
It describes bidirectional communication between the gut and the liver involving:
- intestinal barrier integrity
- gut-derived metabolites and microbial products
- bile acid signaling
- immune and inflammatory pathways
- hepatic metabolic processing
Importantly, this is not binary.
The gut is not simply “leaky” or “not leaky.”
Intestinal permeability exists on a spectrum, influenced by:
- metabolic stress
- inflammation
- bile flow
- circadian disruption
- nutritional context
When that balance shifts, the liver is the first organ that has to deal with the consequences.
LPS, inflammation, and why nuance matters
One gut-derived signal that receives a lot of attention is lipopolysaccharide (LPS)—a component of the outer membrane of certain bacteria.
Here is the careful, adult framing:
- LPS can enter portal circulation in small amounts
- The liver is designed to neutralize it
- Under conditions of metabolic stress, higher exposure can activate hepatic immune pathways
- This activation can interfere with insulin signaling in the liver
However—and this matters greatly:
- LPS is not the only driver
- Measuring it in humans is technically difficult
- Its effects depend on context: diet, bile flow, liver fat, mitochondrial capacity
So the correct statement is not:
“LPS causes insulin resistance.”
The correct statement is:
“Gut-derived inflammatory signals can contribute to hepatic insulin resistance when metabolic buffering capacity is impaired.” That distinction separates physiology from internet mythology.
For US professionals with fatty liver, triglyceride elevation, or insulin resistance — I address these gut–liver mechanisms inside the Metabolic Reset Blueprint using a structured, lab-anchored approach.
Fatty liver (MASLD) as a metabolic amplifier
The modern term MASLD (metabolic dysfunction–associated steatotic liver disease) reflects an important shift in thinking.
Fatty liver is not just fat storage.
It reflects:
- impaired lipid handling
- mitochondrial overload
- oxidative stress
- inflammatory signaling
When fat accumulates in the liver:
- insulin signaling becomes less effective
- hepatic glucose output becomes harder to suppress
- fasting glucose rises
- the entire metabolic system becomes more brittle
This is why fatty liver often sits at the center of metabolic syndrome, even in people who do not appear severely overweight.
See also Metabolic Flexibility
Why “fixing the gut” alone often fails
This is where your work clearly separates from mainstream functional medicine clichés.
Many people try to “heal the gut” while ignoring:
- muscle mass and function
- hepatic energy handling
- circadian disruption
- overall metabolic load
In that context:
- probiotics disappoint
- elimination diets expand endlessly
- symptoms fluctuate but glucose control does not stabilize
Because the gut–liver axis does not operate in isolation.
The liver cannot process signals properly if:
- mitochondrial capacity is low
- glucose overflow remains high
- circadian cues are misaligned
You don’t fix a systems problem by pulling one lever repeatedly.
Practical implications (without turning this into a protocol)
At a conceptual level, improving the gut–liver axis means:
- reducing chronic inflammatory load reaching the liver
- improving hepatic mitochondrial function
- supporting bile flow and nutrient signaling
- lowering unnecessary glucose production by the liver
- offloading glucose disposal to skeletal muscle
- aligning food timing and light exposure
Notice what’s missing:
- no magic supplements
- no extreme restriction
- no single villain
This is systems biology, not symptom chasing.
Final thought
If muscle is the primary glucose sink,
and mitochondria are the energy engine,
then the liver is the metabolic traffic controller—
and the gut is one of its loudest input signals.
When the liver is overwhelmed, insulin resistance becomes a rational response, not a moral failing.
Understanding this changes how we intervene—and why superficial fixes so often disappoint.
If you want a structured approach that addresses muscle, liver, gut, and circadian biology together, start with the Metabolic Road Map.
It’s designed to rebuild metabolic function at the systems level—not chase isolated markers.
Author bio
Morteza Ariana is a Functional Nutrition Practitioner specializing in insulin resistance, type 2 diabetes, and systems-based metabolic restoration. His work focuses on identifying upstream drivers of metabolic dysfunction — including insulin load, liver–gut axis disruption, circadian misalignment, and micronutrient gaps — rather than masking symptoms.
He works with high-performing professionals through a structured 12-week Metabolic Restoration Blueprint designed to restore metabolic flexibility and long-term resilience.
If this resonates, the next step is clarity.
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Scientific References
Nature npj Metabolic Health & Disease (2022) — solid review on host–microbiome interactions and the gut–liver axis; covers dysbiosis, permeability, portal delivery, hepatic immunity/inflammation. Nature
NIH PubMed Central review (2025) — “Unveiling the gut-liver axis”; good overview of portal venous link, bile acids, immune cross-talk. PMC
American Journal of Physiology GI & Liver Physiology (2010) — “Kupffer cell activation is a causal factor for hepatic insulin resistance”; directly supports the “hepatic immune activation → insulin resistance” framing. Physiology Journals
TLR4 and insulin resistance review (2010, PMC) — classic mechanistic overview linking TLR4 activation to inflammation/insulin resistance pathways. PMC
Multinational liver societies / AASLD announcement (June 24, 2023) — describes the change to SLD/MASLD terminology. aasld.org
EASL–EASD–EASO Clinical Practice Guidelines (Journal of Hepatology, 2024) — top-tier guideline update on definitions, screening, diagnosis, and management for MASLD (excellent “authority” citation). journal-of-hepatology.eu+1