Why Chronic Hyperinsulinemia — Not Protein — Disrupts Human Physiology
Introduction: The problem is not mTOR — the problem is dysregulation
In recent years, the mTOR pathway has been increasingly portrayed as a biological villain. Protein intake is often blamed for “overstimulating mTOR,” accelerating aging, and increasing disease risk. This narrative has gained traction in longevity circles and social media, but it reflects a fundamental misunderstanding of human physiology.
mTOR and AMPK are not opposing enemies in a battle for longevity. They are complementary survival systems that evolved to help humans adapt to fluctuating environments. The real question is not whether mTOR is “good” or “bad,” but whether the natural rhythmic cycling between growth and repair has been disrupted.
Modern lifestyles — characterized by constant food availability, frequent eating, hyperinsulinemia, and physical inactivity — distort this biological rhythm. The result is not “too much protein signaling,” but a loss of metabolic flexibility.
What mTOR actually is and what it does
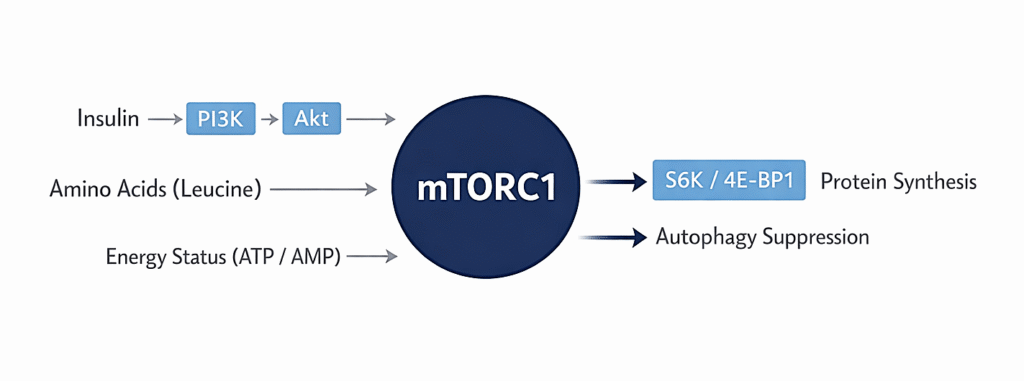
The mammalian target of rapamycin (mTOR) is a highly conserved serine/threonine kinase that functions as a central integrator of environmental signals. It exists in two complexes:
- mTORC1 (mTOR Complex 1): primarily responsible for nutrient sensing, protein synthesis, cell growth, and suppression of autophagy
- mTORC2 (mTOR Complex 2): involved in cytoskeletal organization, insulin signaling modulation, and cellular survival
Most of the controversy around nutrition relates to mTORC1.
Physiologically, mTORC1 activation is essential for:
- Muscle protein synthesis
- Tissue repair
- Immune cell proliferation
- Bone remodeling
- Wound healing
- Reproductive function
- Normal growth and development
Without mTOR signaling, complex multicellular life would not function. The issue is therefore not the existence of mTOR activation, but its chronic, unopposed activation in inappropriate contexts.
What actually activates mTOR in humans
mTORC1 does not respond to a single input. It integrates several signals simultaneously:
- Amino acids (especially leucine)
These act as a permissive signal indicating the presence of building blocks. Leucine activates mTORC1 through Rag GTPases and lysosomal sensing mechanisms. - Insulin and IGF-1
These act as strong upstream activators via the PI3K → Akt → mTOR pathway. Insulin signaling is one of the most potent and sustained drivers of mTORC1 activity. - Cellular energy status
High ATP availability favors mTOR activation. Energy scarcity suppresses it via AMPK. - Mechanical load (exercise)
Resistance training robustly activates mTORC1 in muscle tissue, supporting adaptation and strength. - Inflammatory signaling
Chronic inflammation can dysregulate mTOR activity through cytokine signaling.
A crucial nuance often missed in public discourse:
Protein provides a signal of substrate availability, but insulin determines the duration and systemic persistence of mTOR activation.
A protein-rich meal in an insulin-sensitive person produces a short-lived, physiologically appropriate mTOR pulse. Chronic hyperinsulinemia, however, can keep mTOR signaling elevated regardless of protein intake.
Insulin as a chronic driver of mTOR in modern lifestyles
From a signaling perspective, insulin activates mTORC1 through the classical pathway:
Insulin → Insulin receptor → PI3K → Akt → inhibition of TSC1/2 → activation of mTORC1
This is not controversial biology. The implication, however, is often underappreciated.
In metabolically healthy individuals, insulin rises transiently after meals and returns to baseline between eating periods. mTOR activity follows a similar pulsatile pattern. This rhythmicity is physiologically normal.
In contrast, modern dietary patterns often involve:
- Frequent eating (3 meals plus snacks)
- Refined carbohydrate intake
- Ultra-processed foods
- Liquid calories
- Chronically elevated insulin levels
- Insulin resistance with compensatory hyperinsulinemia
Under these conditions, mTOR signaling can remain persistently elevated, not because of protein intake, but because insulin signaling never truly returns to baseline.
From a systems perspective, the pathological variable is signal duration, not simply signal presence.
See also: Insulin Resistance: The Central Mechanism Behind Modern Chronic Disease
AMPK: the counter-regulatory repair system
AMP-activated protein kinase (AMPK) functions as the cellular energy sensor. It becomes activated when the ratio of AMP to ATP rises, signaling low energy availability.
Physiological activators of AMPK include:
- Fasting
- Caloric restriction
- Exercise
- Glycogen depletion
- Mitochondrial stress
Once activated, AMPK promotes:
- Autophagy (cellular cleanup and recycling)
- Mitochondrial biogenesis (via PGC-1α)
- Increased fatty acid oxidation
- Improved insulin sensitivity
- Inhibition of mTORC1
- Enhanced cellular stress resistance
This is not a longevity “hack.” It is a fundamental survival program that allows organisms to maintain integrity during periods of scarcity.
The problem in modern lifestyles is that AMPK is rarely meaningfully activated because energy availability is almost constant.
See also: Metabolic Flexibility: The Missing Foundation of Modern Metabolic Health
Health depends on rhythmic cycling, not suppression
Biological systems are governed by oscillation:
- Circadian rhythms
- Hormonal pulsatility
- Sleep–wake cycles
- Feast–fast cycles
- Sympathetic–parasympathetic balance
mTOR and AMPK follow this same logic.
Healthy physiology looks like:
- Feeding → transient mTOR activation
- Exercise → localized mTOR activation in muscle
- Fasting → AMPK activation
- Movement → AMPK activation
- Rest → cellular repair
Pathological physiology looks like:
- Constant eating → persistent insulin
- Persistent insulin → persistent mTOR
- Persistent mTOR → suppressed autophagy
- Suppressed autophagy → impaired cellular renewal
- Impaired renewal → metabolic dysfunction
The issue is not that mTOR exists.
The issue is that the biological rhythm between growth and repair has collapsed.
The evolutionary mismatch
Humans evolved in environments characterized by:
- Food unpredictability
- Periods of scarcity
- High physical activity
- Natural fasting intervals
Our signaling systems are adapted to cycles of abundance and scarcity.
The modern environment introduces:
- Continuous food availability
- Artificially hyperpalatable foods
- Constant stimulation of insulin
- Sedentary lifestyles
- Disrupted sleep cycles
This mismatch leads to chronic activation of growth pathways without the necessary counterbalancing repair phases.
From an evolutionary physiology perspective, this is not a protein problem. It is a context problem.
Clinical implications
When viewed through this lens, several clinical patterns become more coherent:
- Insulin resistance and type 2 diabetes
Chronic insulin elevation distorts both metabolic and growth signaling. - NAFLD
Hyperinsulinemia drives hepatic lipogenesis and suppresses fat oxidation. - Sarcopenia in aging
Fear of protein and excessive caloric restriction may suppress necessary mTOR pulses required for muscle maintenance. - Metabolic inflexibility
The inability to switch between fed-state growth and fasted-state repair reflects impaired signaling dynamics. - Aging biology
Not “mTOR activity” per se, but loss of oscillatory balance appears central to many age-related dysfunctions.
This suggests that therapeutic strategies should prioritize:
- Restoring insulin sensitivity
- Creating fasting windows
- Encouraging physical activity
- Supporting adequate protein intake
- Rebuilding natural signaling rhythms
Not indiscriminately suppressing mTOR.
Why the “protein is the problem” narrative persists
The persistence of this narrative reflects broader issues in modern health discourse:
- Reductionism (single pathway explanations for complex systems)
- Misinterpretation of animal models
- Overextension of caloric restriction research
- Fear-based communication
- Social media amplification of simplified ideas
mTOR is easier to demonize than hyperinsulinemia, lifestyle patterns, and metabolic dysfunction — but biology does not conform to ideological simplicity.
See also: Protein, Muscle, and Insulin – Why Most People Get This Wrong
Conclusion: Restore physiology, don’t fear biology
mTOR is not a pathological pathway. It is a fundamental growth regulator essential to human function. Protein is not inherently dangerous. It is biologically indispensable.
The real problem in modern societies is the loss of physiological rhythm:
- Chronic hyperinsulinemia
- Constant feeding
- Lack of fasting periods
- Lack of physical stress signals
- Suppression of AMPK-mediated repair
Health is not achieved by suppressing growth pathways.
It is achieved by restoring the dynamic balance between growth and repair that human biology evolved to depend on.
Author bio
Morteza Ariana is a Functional Nutrition Practitioner specializing in insulin resistance, type 2 diabetes, and systems-based metabolic restoration. His work focuses on identifying upstream drivers of metabolic dysfunction — including insulin load, liver–gut axis disruption, circadian misalignment, and micronutrient gaps — rather than masking symptoms.
He works with high-performing professionals through a structured 12-week Metabolic Restoration Blueprint designed to restore metabolic flexibility and long-term resilience.
If this resonates, the next step is clarity.
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Scientific References
- Saxton RA, Sabatini DM. mTOR Signaling in Growth, Metabolism, and Disease. Cell. 2017.
- Laplante M, Sabatini DM. mTOR signaling in growth control and disease. Cell. 2012.
- Hardie DG. AMP-activated protein kinase: maintaining energy homeostasis. Nat Rev Mol Cell Biol. 2014.
- Kahn BB, Alquier T, Carling D, Hardie DG. AMP-activated protein kinase: ancient energy gauge provides clues to modern understanding of metabolism. Cell Metabolism. 2005.
- Johnson SC, Rabinovitch PS, Kaeberlein M. mTOR is a key modulator of ageing and age-related disease. Nature. 2013.
- Kraft TE, et al. Leucine, not insulin, regulates mTOR in skeletal muscle in vivo. American Journal of Physiology. 2018.
- Ludwig DS, Ebbeling CB. The Carbohydrate-Insulin Model of Obesity. JAMA Internal Medicine. 2018.
- Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiological Reviews. 2018.
- Longo VD, Mattson MP. Fasting: molecular mechanisms and clinical applications. Cell Metabolism. 2014.