How Melatonin and Metabolism Are Connected Through Insulin Timing
Most people know melatonin as a sleep aid. Few know it as a metabolic hormone. That gap is clinically significant — because melatonin is not simply a signal to feel drowsy. It is the body’s primary marker of biological night, and it governs the transition from the active metabolic state of the day to the fasting and repair state of the night.
When melatonin timing is disrupted — delayed, suppressed, or fragmented — that transition fails to happen cleanly. The body stays in a mixed metabolic state at night: neither fully in repair mode nor equipped to handle fuel. Insulin signaling, glucose control, and overnight recovery all suffer as a result.
Understanding how melatonin and metabolism interact is essential for any patient whose metabolic markers are not responding to dietary effort alone — particularly when the clinical picture involves late-night energy, poor mornings, and glucose control that deteriorates through the day.
What you will learn:
The physiological role of melatonin beyond sleep | How melatonin and insulin operate as biological opposites | Why melatonin suppression impairs glucose control | The clinical profile of the melatonin-disrupted metabolic patient | What correcting melatonin timing does to metabolic markers | The clinical position on melatonin supplementation in insulin-resistant patients
What Melatonin Actually Does in the Body
Melatonin is produced by the pineal gland in response to darkness. As light fades in the evening, the suprachiasmatic nucleus releases the inhibition it holds over the pineal gland throughout the day, and melatonin secretion begins. It rises through the evening, peaks in the middle of the night, and falls in the early morning hours as light returns.
This rise and fall is not incidental. It is the central timing signal that coordinates biological night across every organ in the body.
The moment melatonin begins to rise, it communicates one message systemically: the active, feeding phase is ending. The fasting and repair phase is beginning. In response to this signal, the liver reduces glucose output, growth hormone secretion begins, mitochondrial repair processes activate, and the immune system shifts toward anti-inflammatory housekeeping. These are not separate events that happen to coincide with melatonin. They are governed by it.
Melatonin receptors — primarily MT1 and MT2 — are expressed in the brain, retina, cardiovascular tissue, immune cells, the gastrointestinal tract, and critically for metabolic function, in the pancreatic beta cells and liver. The presence of melatonin receptors in these metabolic organs is not incidental. It reflects melatonin’s role as a direct regulator of insulin secretion timing and hepatic glucose metabolism.
Melatonin and Insulin: Biological Opposites by Design
The relationship between melatonin and insulin is one of biological opposition — and that opposition is intentional.
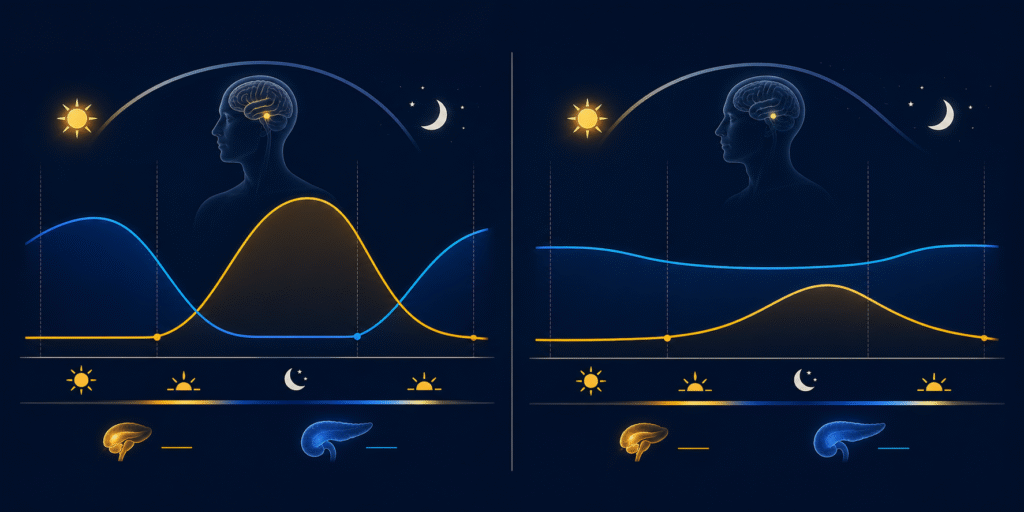
During the day, insulin is the dominant metabolic hormone. It rises in response to feeding, facilitates glucose uptake into cells, and drives nutrient storage and anabolism. Melatonin is suppressed. The body is in active metabolic mode.
At night, melatonin rises and insulin falls. The body enters fasting mode. Glucose is not arriving from food, so the system shifts toward mobilizing stored fuel and running repair processes that require minimal insulin signaling. This anti-phase relationship — melatonin up, insulin down — is how the body cleanly separates the metabolic day from the metabolic night.
Melatonin achieves part of this separation through a direct effect on pancreatic beta cells. When melatonin binds to MT1 and MT2 receptors on beta cells, it reduces cAMP and cGMP signaling — two pathways that drive insulin secretion. In practical terms: rising melatonin suppresses insulin secretion. This is normal, appropriate, and precisely timed to coincide with the period when food intake should have stopped.
This is why late eating in the presence of rising melatonin creates a specific metabolic conflict. The pancreas receives two opposing signals simultaneously: glucose from a late meal demanding insulin secretion, and melatonin signaling the beta cells to reduce output. Insulin secretion is compromised exactly when it is needed. Glucose clearance slows. The postprandial glucose response extends into the night. And the melatonin signal — already suppressed by the light exposure that typically accompanies late meals — loses its clean amplitude.
The result is a body that cannot make a clean metabolic transition from day to night. It stays in a confused intermediate state — not properly in feeding mode, not properly in repair mode — and insulin resistance accumulates in that confusion.
The timing conflict described here is one dimension of a larger circadian desynchronization. For the full picture of how peripheral clock misalignment drives metabolic dysfunction, see Circadian Rhythm Disruption and Metabolism.
The MTNR1B Gene and Individual Metabolic Vulnerability
Not all patients are equally vulnerable to melatonin-insulin disruption. A specific genetic variant — the MTNR1B risk allele — significantly amplifies the metabolic consequences of disrupted melatonin timing.
MTNR1B encodes the MT2 melatonin receptor expressed on pancreatic beta cells. Carriers of the risk variant show higher baseline fasting glucose, reduced first-phase insulin secretion, and a greater deterioration in glucose handling under conditions of circadian disruption or late eating. Studies in large population cohorts have consistently associated MTNR1B risk allele carriage with elevated type 2 diabetes risk — and the mechanism runs directly through the melatonin-insulin interaction described above.
The clinical implication is important: in patients who carry this variant, the standard advice to “have a light snack before bed” or to eat later in the evening is not merely suboptimal. It is mechanistically harmful. Their beta cell response to melatonin suppression is more pronounced, their insulin secretory capacity under late-eating conditions is more compromised, and their overnight glucose regulation is more sensitive to melatonin timing than the average patient.
Even without genetic testing, the patient who shows disproportionate fasting glucose elevation and poor postprandial control in the context of late eating and evening light exposure should be considered a likely candidate for heightened melatonin-metabolic sensitivity. The intervention — earlier meals, evening darkness, melatonin timing correction — is the same regardless of genotype. The urgency is higher.
Clinical Perspective: What I See in Practice
Before a patient mentions their sleep habits or light exposure, the clinical picture often already points to melatonin disruption.
The presenting pattern is consistent. They describe a second wind in the evening — energy and mental clarity that arrive around 21:00 to 23:00, at precisely the time melatonin should be rising and the brain should be transitioning toward sleep pressure. They are not sleepy at night even when physically tired. They describe shallow, non-restorative sleep despite adequate hours — the kind of sleep that leaves them foggy in the morning, slow to start, and feeling like the day only properly begins at 10am or later. The melatonin offset is delayed into the morning, and what should be the sharpest period of metabolic activation — the first two hours after waking — is blunted.
The metabolic hints are equally consistent. Glucose control deteriorates through the day, with the worst readings in the evening. Late-night hunger and cravings appear not from caloric deficit but from the melatonin-insulin mismatch — the body is signaling night while glucose handling is still misaligned. And the characteristic plateau: doing everything correctly nutritionally, energy inconsistent, markers not moving.
The pattern underneath all of it is the same: sleepiness, metabolism, and cognition are all phase-shifted later than biology intends. The melatonin signal is either delayed, suppressed, or of insufficient amplitude to drive a clean metabolic transition into the night phase.
The cases that demonstrate this most clearly are those where diet is already structured and the remaining variable is timing. Fasting glucose sitting between 100 and 110 mg/dL, HbA1c borderline, energy flat in the mornings. The intervention involves no dietary change — only signal correction: morning light within 30 minutes of waking, dim lights after 20:30, screens minimized in the final hour before sleep, a consistent wake time, and the last meal moved earlier.
Within two to four weeks the changes are consistent. Sleep onset accelerates. Awakenings reduce. The night-time “wired” feeling disappears — often within days, which is the first signal that the melatonin phase is beginning to shift. Fasting glucose drops 8 to 12 mg/dL. Evening glucose elevation reduces. Fasting insulin readings become more stable. Morning clarity returns.
Nothing was added nutritionally. The metabolic night signal was restored. The body could finally make the transition it was designed to make — and glucose control followed.
The reframe I use in consultation that consistently lands is this: melatonin is not a sleep hormone. It is the body’s signal that the fasting and repair phase has begun. When that signal is delayed or suppressed, the body stays in a metabolically confused state — still partly in day mode, unable to fully enter night mode. The analogy that makes it concrete: melatonin is the switch that flips the body from “eat and store” to “fast and repair.” If the switch never fully flips, or flips two hours late every night, the metabolic consequences accumulate — regardless of how clean the diet is.
Melatonin disruption rarely exists in isolation. In most patients it is accompanied by sleep deprivation, which triggers its own distinct hormonal cascade. That mechanism is covered in detail in How Poor Sleep Causes Insulin Resistance.
On Melatonin Supplementation in Metabolic Patients
The question of exogenous melatonin supplementation comes up consistently in this patient group, and it requires a more nuanced answer than either “take it” or “avoid it.”
The MTNR1B data has made some practitioners cautious about melatonin supplementation in insulin-resistant patients — and that caution is warranted in the right context. Melatonin acutely reduces insulin secretion through its action on beta cell receptors. In a patient who is still eating late, taking melatonin near food intake can worsen glucose clearance by suppressing the insulin response at a moment when food is still arriving. The problem in that scenario is not melatonin. It is the combination of melatonin with mistimed eating — a conflict that replicates exactly what the circadian mismatch is already producing.
Used correctly, melatonin supplementation is not a metabolic risk. It is a temporary timing tool.
The first intervention is always behavioral — light correction, earlier meals, fixed wake time, evening darkness. That combination addresses 80 to 90 percent of melatonin timing issues without supplementation. When the rhythm is clearly delayed or unstable and behavioral correction is producing slow progress, low-dose melatonin used as a phase-shifting tool can accelerate realignment.
The dose matters significantly. The standard commercial doses of 3 to 10 mg are pharmacological — they produce supraphysiological melatonin levels that can overshoot the timing correction and produce morning grogginess. The physiologically appropriate dose for phase shifting is 0.3 to 1 mg, taken 60 to 90 minutes before the desired sleep time. At this dose, the effect is primarily chronobiotic rather than sedating — it shifts the clock rather than forcing sleep.
Duration should be limited to two to four weeks, used as a bridge while behavioral corrections are taking effect, then tapered. Melatonin supplementation without the underlying behavioral correction is a band-aid. The clock will drift back to its disrupted phase as soon as supplementation stops.
The framing for patients: melatonin is not a sleeping pill. It is a signal. If it is used, the goal is to teach the body when night begins — not to override wakefulness. And the key practical warning: if meal timing has not been corrected, taking melatonin close to food intake can worsen glucose control rather than improve it.
Melatonin, Aging, and the Perimenopausal Patient
Melatonin production declines with age — a well-documented process that begins in the mid-thirties and accelerates through the fifties and beyond. This age-related decline in melatonin amplitude has direct metabolic consequences, because the strength of the biological night signal weakens progressively over decades.
In older adults, the melatonin peak is lower, its duration is shorter, and the transition between day and night phases becomes less defined. This blunting of the melatonin signal partially explains why insulin resistance tends to worsen with age even in the absence of major dietary or lifestyle changes — the circadian regulation of glucose metabolism becomes less precise as the night signal loses its amplitude.
In perimenopausal and menopausal women, this process is compounded by hormonal shifts that independently affect circadian signaling and sleep architecture. Night awakenings from hot flashes fragment the sleep during which melatonin should be peaking. Estrogen decline reduces the sensitivity of central clock mechanisms. The combination of reduced melatonin amplitude, fragmented sleep, and altered hormonal context produces a circadian and metabolic vulnerability that is distinct from, and often more complex than, the melatonin disruption seen in younger patients.
In this population, the clinical picture typically includes sleep fragmentation, inconsistent energy, worsening fasting glucose despite unchanged diet, and an increasing sensitivity to late eating and evening light. The behavioral interventions remain the foundation — morning light, evening darkness, meal timing, wake consistency. The threshold for considering low-dose melatonin as a short-term tool may be lower in this group given the physiological reduction in endogenous production.
What Melatonin Disruption Looks Like in the Lab
Standard metabolic panels do not measure melatonin. There is no routine marker that directly flags melatonin timing disruption in clinical practice. The diagnosis is made clinically — through the symptom pattern, the sleep history, and the combination of metabolic markers that suggest timing is the variable.
The lab constellation that points toward melatonin-metabolic disruption includes mildly elevated fasting glucose between 100 and 115 mg/dL despite dietary compliance, HOMA-IR showing insulin resistance disproportionate to dietary intake, triglycerides mildly elevated without clear dietary explanation, and HbA1c trending between 5.7 and 6.1% without progression or resolution.
Where formal assessment is warranted, dim-light melatonin onset (DLMO) — measured through saliva or urine samples collected in controlled dim light conditions — is the gold standard for quantifying circadian phase. In clinical practice outside of research settings, DLMO testing is not widely available, and the clinical history remains the most practical diagnostic tool. A patient who reliably experiences a second wind after 21:00, cannot fall asleep before midnight, and wakes unrefreshed is describing a delayed DLMO as clearly as any laboratory measurement.
Melatonin is one of three hormones — alongside cortisol and insulin — that form the hormonal control panel of circadian-metabolic coordination, a system explored in full in the cornerstone on how sleep and circadian rhythm govern metabolic health.
A Note on Uncertainty
The role of melatonin in sleep regulation and circadian timing is among the most robustly established areas of chronobiology. The direct relationship between melatonin receptor signaling on pancreatic beta cells and insulin secretion is well-characterized mechanistically.
What carries more uncertainty is the precise clinical magnitude of melatonin timing correction as an isolated intervention on insulin resistance outcomes in heterogeneous patient populations. Most clinical evidence comes from studies combining multiple circadian interventions simultaneously — light, sleep timing, meal timing — making it methodologically difficult to attribute outcome to melatonin correction specifically.
The MTNR1B genetic data is robust in associating the risk variant with elevated diabetes risk. The translation of that association into precise individual clinical recommendations remains an active area of research.
The clinical observations described in this post are consistent with the mechanistic literature and with what is observed repeatedly in practice. They are not a substitute for individualized assessment. Decisions regarding melatonin supplementation, particularly in patients with existing metabolic conditions or who are taking medications affecting glucose regulation, should involve the treating physician.
People Also Ask
How does melatonin affect metabolism and blood sugar?
Melatonin signals biological night to every organ in the body, including the liver and pancreas. It directly suppresses insulin secretion through melatonin receptors on beta cells — a normal and intentional mechanism that prepares the body for overnight fasting. When melatonin timing is disrupted, this suppression occurs at the wrong time or with insufficient amplitude, and the clean transition from feeding mode to fasting mode fails. Glucose clearance is impaired and overnight insulin regulation deteriorates.
Can poor melatonin timing cause insulin resistance?
Yes. Chronic melatonin suppression or delay — driven primarily by evening light exposure and late eating — sustains a metabolic state in which the body cannot cleanly enter its fasting and repair phase. The liver continues producing glucose when it should be reducing output. The pancreas receives conflicting signals between food arrival and melatonin suppression of beta cell function. Over time, this timing disruption contributes directly to insulin resistance independent of dietary composition.
Is it safe to take melatonin supplements if you have insulin resistance?
Context determines the answer. Melatonin taken at the wrong time — near food intake, particularly late in the evening when eating has not been corrected — can worsen glucose handling by suppressing insulin secretion when glucose is still arriving. Melatonin used correctly as a phase-shifting tool — low dose (0.3 to 1 mg), 60 to 90 minutes before desired sleep, away from meals, paired with light and timing corrections — is not harmful and can support circadian realignment. Behavioral correction of light and meal timing should always precede supplementation.
What is the connection between the MTNR1B gene and diabetes risk?
MTNR1B encodes the MT2 melatonin receptor on pancreatic beta cells. Carriers of the risk variant show reduced first-phase insulin secretion, higher fasting glucose, and greater metabolic vulnerability to circadian disruption and late eating. The risk allele is one of the most replicated genetic associations with type 2 diabetes, and the mechanism runs directly through melatonin’s suppressive effect on insulin secretion at night.
What are the signs that melatonin disruption is affecting metabolism?
The key clinical pattern is phase-shifted energy and glucose regulation: a second wind in the evening when sleepiness should be rising, non-restorative sleep despite adequate hours, morning brain fog and sluggish metabolic activation, glucose control that deteriorates through the day, and metabolic markers that plateau despite dietary compliance. The underlying signal is a melatonin onset that is delayed, suppressed, or of insufficient amplitude to drive a clean metabolic transition into the night phase.
What is the most effective way to restore melatonin timing without supplements?
Morning light exposure within 30 minutes of waking is the most powerful circadian anchor — 10 to 20 minutes of direct outdoor light resets the central clock and advances melatonin onset progressively over days. Evening darkness — dim, warm lighting after 20:30 and reduced screen use in the final hour before sleep — removes the primary suppressive signal. A fixed wake time seven days per week maintains the anchor. Earlier meal timing removes the feeding-melatonin conflict. These four inputs address the majority of melatonin timing disruption without supplementation.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
- Reppert SM, Weaver DR. Coordination of circadian timing in mammals. Nature. 2002;418(6901):935–941. 🔗 https://pubmed.ncbi.nlm.nih.gov/12198538/
- Peschke E, Bähr I, Mühlbauer E. Melatonin and pancreatic islets: interrelationships between melatonin, insulin and glucagon. International Journal of Molecular Sciences. 2013;14(4):6981–7015. 🔗 https://pubmed.ncbi.nlm.nih.gov/23535335/
- Bouatia-Naji N, Bonnefond A, Cavalcanti-Proença C, et al. A variant near MTNR1B is associated with increased fasting plasma glucose levels and type 2 diabetes risk. Nature Genetics. 2009;41(1):89–94. 🔗 https://pubmed.ncbi.nlm.nih.gov/19060909/
- Lyssenko V, Nagorny CLF, Erdos MR, et al. Common variant in MTNR1B associated with increased risk of type 2 diabetes and impaired early insulin secretion. Nature Genetics. 2009;41(1):82–88. 🔗 https://pubmed.ncbi.nlm.nih.gov/19060908/
- Rubio-Sastre P, Scheer FAJL, Gómez-Abellán P, Madrid JA, Garaulet M. Acute melatonin administration in humans impairs glucose tolerance in both the morning and evening. Sleep. 2014;37(10):1715–1719. 🔗 https://pubmed.ncbi.nlm.nih.gov/25197811/
- Lewy AJ, Bauer VK, Ahmed S, et al. The human phase response curve (PRC) to melatonin is about 12 hours out of phase with the PRC to light. Chronobiology International. 1998;15(1):71–83. 🔗 https://pubmed.ncbi.nlm.nih.gov/9493716/
- Arendt J, Skene DJ. Melatonin as a chronobiotic. Sleep Medicine Reviews. 2005;9(1):25–39. 🔗 https://pubmed.ncbi.nlm.nih.gov/15649736/
- Tan DX, Manchester LC, Fuentes-Broto L, Paredes SD, Reiter RJ. Significance and application of melatonin in the regulation of brown adipose tissue metabolism. Obesity Reviews. 2011;12(3):167–188. 🔗 https://pubmed.ncbi.nlm.nih.gov/20880126/
- Garaulet M, Gomez-Abellan P, Alburquerque-Bejar JJ, et al. Timing of food intake predicts weight loss effectiveness. International Journal of Obesity. 2013;37(4):604–611. 🔗 https://pubmed.ncbi.nlm.nih.gov/23357955/
- Hardeland R. Melatonin and the pathologies of weakened or dysregulated circadian oscillators. Journal of Pineal Research. 2017;62(1). 🔗 https://pubmed.ncbi.nlm.nih.gov/27706852/
- Karasek M. Melatonin, human aging, and age-related diseases. Experimental Gerontology. 2004;39(11–12):1723–1729. 🔗 https://pubmed.ncbi.nlm.nih.gov/15582288/
- Reiter RJ, Tan DX, Korkmaz A, Ma S. Obesity and metabolic syndrome: association with chronodisruption, sleep deprivation, and melatonin suppression. Annals of Medicine. 2012;44(6):564–577. 🔗 https://pubmed.ncbi.nlm.nih.gov/21955093/