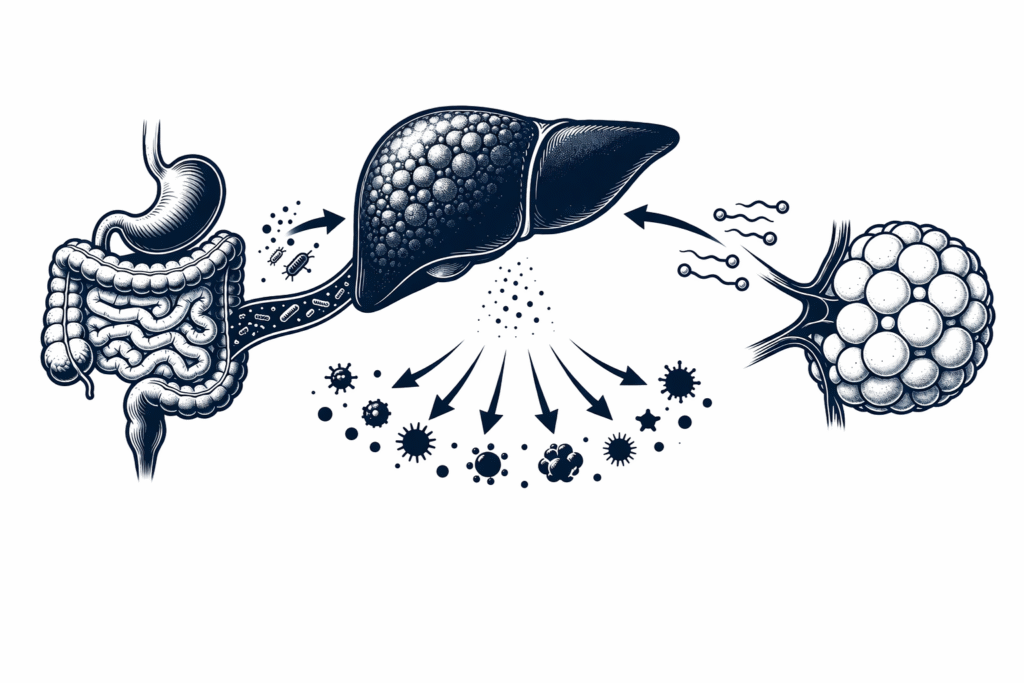
Fatty liver and metabolic dysfunction share the same upstream driver — and it is not the liver.
Fatty liver is named after one organ. That name is the problem.
It implies a local condition — a liver that has accumulated too much fat, for reasons that begin and end inside the liver itself. Eat less fat, lose some weight, and the liver normalizes. This is how most patients understand it when they are told about it at all. And it is precisely why so many people with fatty liver, elevated liver enzymes, and early metabolic dysfunction never get better: the intervention targets the organ, not the network that is driving it.
Fatty liver is not a liver disease. It is a metabolic disease that expresses itself most visibly — and most measurably — in the liver. The liver is the readout. The drivers are elsewhere: in adipose tissue releasing unregulated free fatty acids into portal circulation, in a disrupted gut microbiome flooding the liver with inflammatory signals, in chronically elevated insulin stimulating fat production inside hepatocytes, and in the downstream inflammatory cascade that impairs insulin signaling across every organ simultaneously.
Understanding fatty liver as an organ network problem changes everything. It changes which markers you measure, which organs you address, and why interventions that target only diet or only the liver consistently produce incomplete results.
What you will learn: Why the liver is the readout of a multi-organ metabolic dysfunction, not its cause | The four organ systems driving fatty liver and how they interact | Why standard liver enzyme testing misses this process until it is advanced | Which biomarkers actually reflect the network — not just the organ | Why this is the most important and most neglected pattern in standard metabolic screening
Clinical Perspective of Fatty Liver and Metabolic Dysfunction: What I See in Practice
The most striking clinical reality about fatty liver is not how serious it is. It is how invisible it remains in standard care. In over a decade of practice, I have not encountered a single patient who arrived having been told by their GP that their elevated ALT was a metabolic warning signal.
Not one who had been told that their liver enzymes were reflecting early fat accumulation driven by insulin resistance. The conversation, when it happens at all, is brief and conclusory — your liver markers are slightly elevated, we will monitor them. What is driving the elevation, what organs are involved, and what to do about it: none of that is communicated. Patients leave not knowing they have a problem.
What I find consistently in these patients is that the liver is never the only thing telling the story. By the time ALT is trending above the functional concern threshold of 35 U/L, there are almost always other signals present that standard screening either ignores or interprets in isolation. Triglycerides creeping toward or above 100 mg/dL. A TG/HDL ratio above 2.0. Fasting glucose above the functional optimal of 85 mg/dL. These markers are present in the same panel. They are not connected. The GP sees cholesterol and blood sugar. The organ network — adipose tissue, gut, liver, inflammatory signaling — remains invisible.
The two mechanisms I find most underappreciated in this picture are the gut-liver axis and adipose tissue insulin resistance, in that order. Microbiome dysbiosis, intestinal barrier disruption, LPS translocation into portal circulation, TLR4 activation on hepatic Kupffer cells — this inflammatory cascade arriving directly at the liver from the gut is the mechanism I encounter most consistently in patients whose fatty liver picture does not fit the classic overweight, high-carbohydrate profile.
The inflammatory pathway that drives hepatic insulin resistance is downstream of both the gut-liver axis disruption and the adipose tissue FFA spillover. It is not a separate driver. It is what both of those mechanisms produce when they are left unaddressed.
What happens when I explain this to a patient is one of the most consistent experiences in clinical practice. They listen carefully. They are surprised — not by the complexity, but by the fact that no one has explained any of it before. For the first time, the pieces connect. The fatigue, the belly fat, the elevated liver enzymes, the slightly high triglycerides — they are not separate problems requiring separate management. They are one problem expressed across multiple organs. That shift in understanding is often the moment the patient becomes genuinely motivated to act, because for the first time the intervention makes biological sense to them.
The case that captures this organ network story most precisely is one I return to often. A patient arrived with what a standard panel would describe as broadly unremarkable — ALT at 41.6 U/L, fasting glucose at 107 mg/dL, triglycerides at 96.9 mg/dL, HDL at 45.2 mg/dL, HbA1c at 5.9%. Nothing flagged except the glucose and total cholesterol. No mention of liver function as a metabolic concern. Shared with patient permission, all identifying information removed.
After structured metabolic intervention addressing dietary quality, gut-liver axis disruption, and insulin load, the follow-up panel told a different story: ALT dropped from 41.6 to 20.9 — from above the functional concern threshold into the optimal range. Fasting glucose normalized from 107 to 81 mg/dL. Triglycerides fell from 96.9 to 68.7. HDL rose from 45.2 to 56.4. The liver did not improve in isolation. Everything improved together — because the intervention addressed the organ network, not the individual markers. That is the story fatty liver tells when you read it correctly.
Why the Liver Is the Wrong Starting Point
The liver sits at an anatomical crossroads that no other organ occupies. Everything absorbed from the gastrointestinal tract — every fatty acid, every glucose molecule, every bacterial product, every dietary toxin — travels through the portal vein to the liver before it reaches the systemic circulation. The liver processes it all first.
This position makes the liver the most sensitive metabolic sensor in the body. It is also what makes it the most vulnerable. When the systems that feed into it are dysregulated — when adipose tissue is releasing excess free fatty acids, when the gut microbiome is generating inflammatory signals, when insulin is chronically elevated — the liver absorbs the consequences before any other organ does.
This is why fatty liver appears early in the trajectory of insulin resistance — not late. It is not a complication of advanced metabolic disease. It is one of the earliest detectable signs that the upstream organ network is under strain. By the time a standard ultrasound confirms hepatic steatosis, the metabolic dysfunction driving it has typically been operating for years.
The clinical implication is precise: to understand and address fatty liver, you must understand the organs that are driving it. The liver is where the problem becomes visible. The problem itself is elsewhere.
The Four Organ Systems Driving Fatty Liver
Adipose Tissue: The Unregulated Supply Line
In a metabolically healthy individual, insulin suppresses lipolysis in adipose tissue after meals — keeping free fatty acid release regulated and time-gated. In insulin-resistant adipose tissue, this suppression fails. Hormone-sensitive lipase remains active regardless of insulin levels, releasing a continuous, unregulated stream of free fatty acids into portal and systemic circulation around the clock.
The liver is the first organ downstream of this spillover. It receives a fat load that far exceeds its oxidative capacity. What it cannot burn for energy it re-esterifies into triglycerides — storing them inside hepatocytes or packaging them into VLDL for export. When the rate of incoming free fatty acids chronically exceeds both capacity limits, fat accumulates inside the liver.
This is the mechanism through which fatty liver develops in lean individuals — people with a normal BMI and a moderate diet — who have insulin-resistant adipose tissue. The liver is not receiving too much dietary fat. It is receiving too much adipose-derived fat, driven by a signaling failure in the fat cells themselves. The full mechanistic account of this pathway is covered in the post on how insulin resistance drives fatty liver.
What is critical to understand at the organ network level is that adipose tissue insulin resistance and hepatic fat accumulation form a self-reinforcing loop. Portal FFA spillover drives hepatic fat accumulation. Hepatic fat accumulation drives hepatic insulin resistance. Hepatic insulin resistance drives compensatory hyperinsulinemia. Hyperinsulinemia drives further visceral fat deposition. More visceral fat means more FFA spillover. The cycle tightens with each turn.
The Gut-Liver Axis: The Inflammatory Supply Line
The portal vein does not only deliver nutrients and fatty acids to the liver. It delivers everything the gut produces — including the products of a disrupted microbiome.
When the intestinal barrier is compromised — by ultra-processed food components, industrial seed oils, emulsifiers, HFCS, chronic stress, or antibiotic exposure — lipopolysaccharide from gram-negative gut bacteria translocates across the gut wall and enters portal circulation. The liver receives this LPS load first. Kupffer cells, the liver’s resident macrophages, express TLR4 receptors that bind LPS and activate the NF-κB inflammatory cascade, producing hepatic TNF-alpha and IL-6. These inflammatory signals directly impair insulin receptor signaling in hepatocytes through IRS-1 serine phosphorylation — producing hepatic insulin resistance through an inflammatory mechanism that is entirely independent of fat accumulation.
This is the pathway that explains why patients who eat a relatively controlled diet and maintain a reasonable body weight can still develop fatty liver and hepatic insulin resistance. If gut barrier integrity is compromised, the liver is receiving a chronic inflammatory stimulus regardless of macronutrient composition. The gut-liver axis is not a secondary consideration in the fatty liver story. For a significant proportion of patients, it is the primary driver.
Microbiome dysbiosis compounds this through a second mechanism: altered bile acid metabolism. The gut microbiome plays a central role in bile acid transformation, and dysbiosis disrupts the bile acid signaling that regulates hepatic lipid metabolism through FXR and TGR5 receptors. Impaired bile acid signaling reduces the liver’s capacity to manage its own fat load — adding a regulatory failure on top of the inflammatory burden already arriving through the portal vein.
Hyperinsulinemia: The Internal Fat Production Signal
While adipose tissue and the gut represent external fat delivery systems to the liver, the liver also has an internal fat production mechanism that is driven directly by elevated insulin — and this mechanism operates independently of dietary fat intake.
De novo lipogenesis — the conversion of carbohydrate substrate into fat inside the liver — is stimulated by insulin through the transcription factor SREBP-1c. In a metabolically healthy individual with normal fasting insulin, this process is modest and tightly regulated. In a state of chronic hyperinsulinemia — where the pancreas is compensating for peripheral insulin resistance by secreting excess insulin — hepatic SREBP-1c activation is sustained, and de novo lipogenesis runs at an elevated rate continuously.
The metabolic paradox at the center of this mechanism is that the liver develops selective insulin resistance: it becomes resistant to insulin’s glucose-suppressing effects while remaining fully sensitive to insulin’s fat-producing effects. The same elevated insulin signal that fails to suppress hepatic glucose output continues to drive fat production inside the liver. This is why fasting glucose rises and triglycerides accumulate simultaneously in the same patient — not as separate problems but as two outputs of the same selective resistance pattern.
The practical implication is that addressing dietary fat alone does not resolve this pathway. The driver is insulin. Reducing the insulin load — through dietary quality, carbohydrate reduction, intermittent fasting, and restoration of insulin sensitivity — is what switches off the hepatic fat production signal at its source.
Chronic Inflammation: The Signal That Locks the Network In Place
The inflammatory cascade generated by both the gut-liver axis disruption and the adipose tissue FFA spillover does not remain local. It becomes systemic — and once it does, it impairs insulin signaling across all insulin-sensitive tissues simultaneously.
Pro-inflammatory cytokines including TNF-alpha and IL-6, produced by both hepatic Kupffer cells responding to LPS and by visceral adipose tissue macrophages responding to lipid overload, activate JNK and NF-κB signaling in liver, muscle, and adipose tissue. JNK phosphorylates IRS-1 at serine 307, converting it from a signal activator to a signal inhibitor. Every organ that depends on intact insulin signaling is affected.
This is what locks the organ network into a self-perpetuating dysfunction. Adipose tissue insulin resistance worsens because inflammation impairs its insulin signaling. Hepatic insulin resistance deepens for the same reason. Compensatory hyperinsulinemia increases in response, driving more visceral fat deposition, more FFA spillover, and more hepatic fat production. The inflammatory signal does not resolve spontaneously because the organ conditions generating it — adipose tissue dysfunction and gut barrier disruption — remain in place.
Addressing chronic inflammation in the fatty liver picture therefore requires addressing its sources — not suppressing its downstream markers. CRP, the accessible clinical proxy for this systemic inflammatory state, should always be interpreted in the context of the full organ network picture, not as a standalone finding.
What Standard Screening Misses and Why
Standard liver enzyme testing — ALT, AST, GGT — is designed to detect hepatocellular damage. It is not designed to detect metabolic dysfunction. These are not the same thing.
ALT begins to rise when hepatocytes are under sufficient stress to release their cytoplasmic contents. By the time ALT is elevated above the conventional upper limit of 56 U/L, meaningful hepatic fat accumulation has typically already been present for years. Studies using liver biopsy and imaging have consistently demonstrated that a significant proportion of individuals with confirmed hepatic steatosis have normal ALT values. The enzyme test is a late signal masquerading as an early one.
The functional medicine threshold for ALT concern is above 35 U/L — the level at which hepatic metabolic stress becomes clinically meaningful even if structural damage has not yet occurred. At 41.6 U/L, as in the case described above, the liver is already under strain that a conventional panel would describe as normal. This is the diagnostic gap that produces the pattern seen in practice every day: patients reassured that their liver function is fine, while the organ network driving hepatic fat accumulation continues to operate undetected.
GGT is a more sensitive early marker — reflecting oxidative stress and hepatic metabolic burden before ALT rises. The functional concern threshold for GGT is above 20 U/L, compared to a conventional upper limit that typically sits above 60. A GGT of 25 U/L in the context of a TG/HDL ratio above 2.0 and a fasting glucose above 100 mg/dL is a coherent early metabolic signal. Standard screening reads it as normal.
The marker panel that actually reflects the organ network is not liver enzymes in isolation. It is liver enzymes in context: alongside fasting insulin, HOMA-IR, TG/HDL ratio, hsCRP, waist circumference, and fasting glucose. Together, these markers provide a picture of how many organ systems are under strain and how far the dysfunction has progressed. Individually, each one can appear unremarkable while the network is actively deteriorating.
The Biomarker Map of the Organ Network
Reading the organ network requires reading the right markers in combination. Here is what each layer of the network produces in the lab:
Adipose tissue insulin resistance shows up as: elevated fasting insulin, rising triglycerides, suppressed HDL, TG/HDL ratio above 2.0 in mg/dL units, increasing waist circumference. These are the lipid fingerprint of unregulated FFA spillover and compensatory hyperinsulinemia operating together.
Hepatic fat accumulation and hepatic insulin resistance shows up as: ALT trending above 35 U/L, GGT above 20 U/L, rising fasting glucose despite normal or borderline HbA1c, elevated triglycerides from excess VLDL export, and HOMA-IR above 2.0 when fasting insulin is available.
Gut-liver axis disruption does not have a single direct biomarker in standard panels. It shows up indirectly as: elevated hsCRP without an obvious acute cause, elevated GGT reflecting oxidative stress from gut-derived toxin exposure, and the overall clinical pattern of metabolic dysfunction in a patient whose diet appears reasonable by conventional standards.
Systemic chronic inflammation shows up as: hsCRP above 1.0 mg/L, elevated ferritin in the context of elevated CRP (reflecting acute phase response rather than true iron excess), and the combination of suppressed HDL with normal or borderline triglycerides that suggests inflammation-driven rather than insulin-driven dyslipidemia.
No single marker captures the network. The picture emerges from reading all of them together — which is precisely what standard screening is not designed to do.
A Note on Uncertainty
The organ network model described in this post reflects the current mechanistic understanding of NAFLD pathogenesis. The individual pathways — adipose tissue FFA spillover, de novo lipogenesis, gut-derived LPS translocation, inflammatory IRS-1 impairment — are each well-characterized in the research literature. What is less precisely quantified is the relative contribution of each pathway in any given individual. In some patients, gut-liver axis disruption dominates. In others, adipose tissue insulin resistance is the primary driver. In most patients with established fatty liver, multiple pathways are operating simultaneously and reinforcing each other.
Clinical assessment — integrating the full biomarker panel with dietary history, body composition, gut health history, and inflammatory markers — is required to identify which drivers are most active and to prioritize intervention accordingly. The organ network model is a framework for asking the right questions, not a formula that applies identically to every patient.
Next Steps
If your liver enzymes have been described as normal or borderline, if your triglycerides are rising, if your fasting glucose is above 90 mg/dL, and if no one has connected these findings into a coherent picture — the organ network described in this post is almost certainly operating in your case. The question is not whether intervention is indicated. It is which layer of the network to address first and how to measure progress objectively.
The posts in this cluster cover each component of the network in the depth it deserves: adipose tissue insulin resistance and free fatty acid spillover, visceral fat versus subcutaneous fat, de novo lipogenesis, choline deficiency and liver fat, gut-liver axis disruption, and the biomarker interpretation framework for ALT, AST, and GGT.
The liver is telling you something. Standard screening is not designed to hear it. This cluster is.
People Also Ask
What is the connection between fatty liver and metabolic disease?
Fatty liver is one of the earliest structural manifestations of insulin resistance and metabolic dysfunction. It develops when multiple organ systems — adipose tissue, the gut microbiome, the insulin signaling network, and the hepatic lipid metabolism system — are simultaneously under strain. It is not a standalone liver condition. It is a readout of systemic metabolic dysregulation that begins years before any conventional diagnostic threshold is crossed.
Can you have fatty liver with normal liver enzymes?
Yes — and this is one of the most clinically important gaps in standard screening. Multiple studies using liver biopsy and imaging have confirmed that a significant proportion of individuals with confirmed hepatic steatosis have ALT and AST values within the conventional normal range. Liver enzymes reflect hepatocellular damage, not fat accumulation. Fat can accumulate for years before enzyme elevation occurs. Functional medicine thresholds — ALT concern above 35 U/L rather than the conventional upper limit of 56 U/L — improve early detection but do not eliminate this gap entirely.
What causes fatty liver in people who are not overweight?
Lean fatty liver — hepatic steatosis in individuals with a normal BMI — is driven by the same organ network mechanisms as fatty liver in overweight individuals: adipose tissue insulin resistance producing chronic FFA spillover into portal circulation, gut-liver axis disruption generating hepatic inflammation, and hyperinsulinemia stimulating de novo lipogenesis. The difference is that in lean individuals, the visceral fat component may be relatively small while the gut-liver axis disruption and insulin signaling impairment are the dominant drivers.
What is the TG/HDL ratio and why does it matter for fatty liver?
The triglyceride-to-HDL ratio is one of the most accessible clinical proxies for insulin resistance and the dyslipidemia pattern associated with hepatic fat accumulation. A ratio above 2.0 in mg/dL units reflects the lipid fingerprint of hyperinsulinemia — elevated hepatic VLDL triglyceride synthesis and suppressed HDL. In the context of borderline liver enzymes and rising fasting glucose, a TG/HDL ratio above 2.0 is a coherent early signal that the organ network is under strain, even when no individual marker has crossed a conventional diagnostic threshold.
Why does fatty liver keep coming back after treatment?
Fatty liver recurs when intervention addresses the liver without addressing the organ network driving it. Dietary changes that reduce hepatic fat temporarily but do not restore adipose tissue insulin sensitivity, gut barrier integrity, or systemic insulin signaling will produce transient improvement followed by recurrence. Durable resolution requires identifying and correcting the upstream drivers — which vary by patient — not managing the downstream hepatic consequence in isolation.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
- Donnelly KL, et al. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. Journal of Clinical Investigation. 2005;115(5):1343–1351.
- Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiological Reviews. 2018;98(4):2133–2223.
- Cani PD, et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56(7):1761–1772.
- Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006;444(7121):860–867.
- Fabbrini E, Sullivan S, Klein S. Obesity and nonalcoholic fatty liver disease: biochemical, metabolic, and clinical implications. Hepatology. 2010;51(2):679–689.
- Targher G, Byrne CD, Tilg H. NAFLD and increased risk of cardiovascular disease: clinical associations, pathophysiological mechanisms and pharmacological implications. Gut. 2020;69(9):1691–1705.
- Musso G, Gambino R, Cassader M. Gut microbiota as a regulator of energy homeostasis and ectopic fat deposition. Current Opinion in Lipidology. 2010;21(1):76–83.
- Browning JD, Horton JD. Molecular mediators of hepatic steatosis and liver injury. Journal of Clinical Investigation. 2004;114(2):147–152.
- Kotronen A, Yki-Järvinen H. Fatty liver: a novel component of the metabolic syndrome. Arteriosclerosis, Thrombosis, and Vascular Biology. 2008;28(1):27–38.
- Stefan N, et al. Causes and metabolic consequences of fatty liver. Endocrine Reviews. 2008;29(7):939–960.
