Chronic low-grade inflammation is one of the most misunderstood findings in routine blood work. If your CRP came back elevated and your doctor shrugged, said “watch it,” or blamed it on stress, you were not given the full picture. Chronic low-grade inflammation is not random immune activation — it is a measurable signal that your metabolism is under sustained overload, and it connects directly to insulin resistance, visceral fat, liver stress, and circadian disruption.
Most people hear the word inflammation and think of pain, swelling, or an infection that needs to be suppressed.
In the conventional medical model, chronic inflammation is often treated as:
- a risk factor
- a lab value
- or a “silent danger” that must be lowered
Supplements companies tell people they need antioxidants, detox powders, and exotic anti-inflammatory blends.
But when you zoom out from symptom-based thinking and look at the body through a systems-biology lens, something becomes very clear:
Chronic low-grade inflammation is not random immune activation.
It is a metabolic adaptation to stress, nutrient overload, circadian disruption, and impaired energy handling.
In other words — the body is not malfunctioning.
It is responding to an environment it can no longer regulate efficiently.
And the longer this state persists, the more metabolism shifts away from health-promoting, flexible energy use — and toward protective, conservative survival physiology.
Let’s break down what that actually means.
Acute inflammation vs chronic low-grade inflammation — two completely different states
Inflammation is not “good” or “bad”.
It is contextual biology.
Acute inflammation
The kind you learn in textbooks:
- short-term
- targeted
- triggered by injury, infection, or tissue damage
- intense — but temporary
It is how the body protects and repairs.
And when the threat is resolved, the immune response shuts down and tissues return to baseline.
That is healthy physiology.
Chronic low-grade inflammation is different
This is not injury, infection, or tissue damage.
It is subtle.
It is persistent.
It does not cause dramatic symptoms — it reshapes how the body:
- allocates energy
- handles nutrients
- manages stress
- regulates metabolism
The immune system is not attacking anything.
It is responding to chronic metabolic and environmental load.
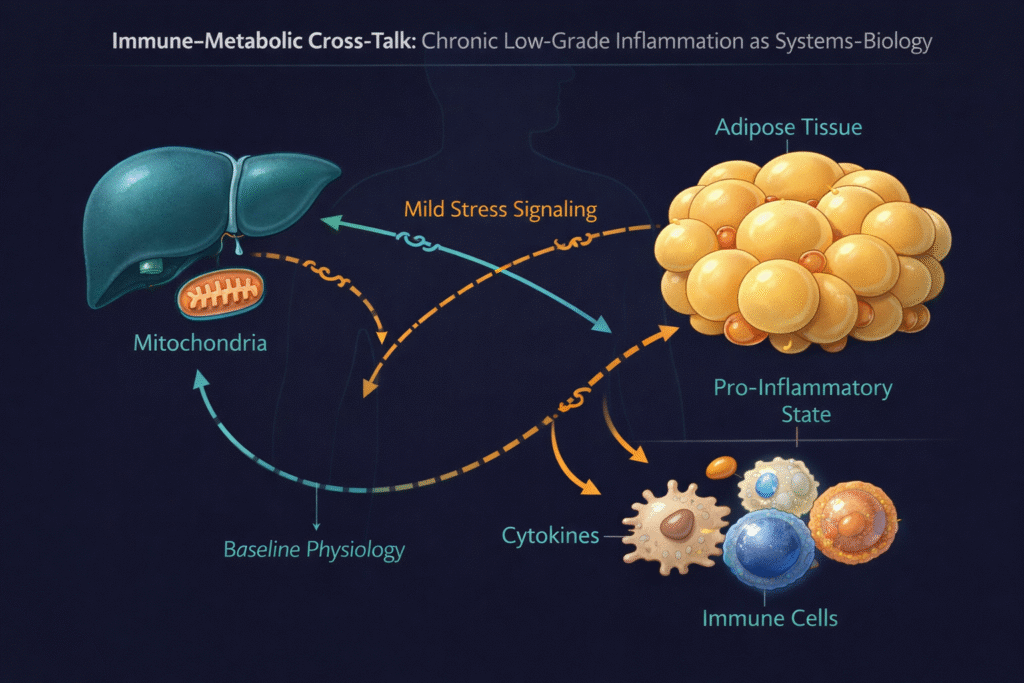
And that response changes how:
- the liver processes fuel
- the muscle utilizes glucose
- adipose tissue stores and releases energy
- mitochondria regulate oxidation
- the brain interprets fatigue and motivation
Chronic inflammation isn’t a “disease”.
It is a signal.
Where chronic inflammation really comes from (mechanistic view)
When we step out of symptom-based thinking and map physiology upstream, chronic low-grade inflammation simply reflects the cost of long-term metabolic tension.
The contributors are rarely isolated.
They stack.
They interact.
They form a pattern.
The most common upstream drivers include:
Hepatic insulin resistance & lipid turnover
When the liver is exposed to excess substrate load — glucose, alcohol, refined carbohydrates, frequent eating windows, stress hormones — it increases:
- gluconeogenesis
- de novo lipogenesis
- VLDL export
- triglyceride turnover
This is the pattern many people see long before “disease” appears.
The TG:HDL ratio begins to shift.
Energy becomes trapped in circulation instead of being used efficiently.
Inflammatory signaling increases not because the body is broken —
but because the system is under load.
See also Beyond LDL: Why the TG:HDL ratio Exposes Hidden Insulin Resistance (Long Before “Disease” Is Diagnosed)
The gut–liver axis & immune signaling
Everything absorbed from the gut travels first to the liver.
The portal vein is not just a nutrient highway —
it is an immune-metabolic communication channel.
What reaches the liver isn’t only:
- glucose
- amino acids
- dietary fats
…but also microbial metabolites and small amounts of endotoxin (LPS) — especially when:
- circadian rhythm is disrupted
- meals are ultra-processed, late or irregular
- intestinal barrier stress increases
- the liver is already under metabolic load
The liver does not treat this as infection.
It treats it as a safety-context signal.
Kupffer cells, hepatocytes, and immune-associated cells respond by:
- increasing inflammatory tone
- modifying lipid turnover
- changing glucose export
- altering metabolic signaling to the rest of the body
This isn’t pathology.
It is the liver saying:
“Input quality and timing are uncertain —
we are operating in a stressed environment.”
Inflammation in this context is not the problem.
It is the translation layer between the gut and metabolic regulation.
Circadian misalignment & sleep fragmentation
Sleep is not recovery.
Sleep is metabolic regulation.
When circadian rhythm drifts, even slightly:
- cortisol rhythm shifts
- autonomic tone changes
- oxidative stress increases
- metabolic flexibility decreases
And the immune system reads this as:
“We are operating in a stressed environment —
conserve resources, limit sensitivity, move into protection mode.”
That mode is inflammation-associated metabolism.
Mitochondrial overload & redox stress
Mitochondria are not “powerhouses”.
They are sensors.
They constantly evaluate:
- nutrient excess
- oxygen availability
- stress load
- substrate choice
- mechanical demand
- cellular safety
When energy demand and substrate load don’t match…
…they send out danger-adaptation signals.
Those signals look like inflammation.
Not because damage is occurring —
—but because the mitochondria are saying:
“We cannot safely continue running like this.”
Inflammation is the brake pedal.
It slows the system down.
It forces a different metabolic strategy.
See also Metabolic Flexibility: The Missing Foundation of Modern Metabolic Health
See also The Gut-Liver Axis: Why the Liver “Feels” the Gut First
Why inflammation and insulin resistance travel together
These two states are rarely independent.
They co-evolve.
They reinforce one another.
Under chronic metabolic strain:
- immune cells shift fuel preference
- liver increases glucose + lipid output
- skeletal muscle becomes less insulin-sensitive
- adipose tissue releases more circulating energy
- triglycerides rise
- HDL remodeling decreases
- mitochondrial oxidation becomes less flexible
This is not failure.
It is defensive mode.
And in that mode, the body prioritizes:
- survival
- stability
- energy containment
Rather than:
- performance
- resilience
- metabolic adaptability
Inflammation is the language the body uses to orchestrate that shift.
See also Insulin Resistance: The Central Mechanism Behind Modern Chronic Disease
The symptom pattern most people feel (long before disease)
Most of the people who eventually find their way to functional medicine don’t say:
“I’m sick.”
They say things like:
- “I crash in the afternoon.”
- “I can’t lose belly fat even though I’m disciplined.”
- “I wake up at 3 a.m. several nights a week.”
- “My triglycerides are creeping up.”
- “My energy feels shallow.”
- “My brain feels dull under stress.”
Sometimes CRP is elevated.
Sometimes ferritin climbs.
Sometimes TG:HDL ratio shifts.
Sometimes everything looks “normal”.
But biologically — the pattern is already changing.
Inflammation is not the problem.
It is the translation of the problem.
Why conventional medicine struggles with this state
Not because doctors lack intelligence.
But because the framework is different.
Healthcare interprets physiology as categories:
- cardiology
- endocrinology
- gastroenterology
- psychiatry
- sleep medicine
Every organ.
Every symptom.
Owned by a different discipline.
But chronic inflammation is not an organ problem.
It is a coordination signal.
A systems-level language.
So instead of seeing:
- hepatic insulin resistance
- rising triglycerides
- circadian fragmentation
- gut–liver immune signaling
- mitochondrial redox stress
…medicine sees:
- mild dyslipidemia
- anxiety
- fatigue
- reflux
- poor sleep
And the pattern becomes invisible.
What gets treated are downstream effects.
Not the system that created them.
What actually calms chronic inflammation (there is no shortcut)
Most people search for:
- anti-inflammatory supplements
- detox powders
- miracle antioxidants
- magic diets
But if chronic inflammation is an adaptive response…
…forcing it down is like muting a fire alarm
while the building is still overheating.
Inflammation quiets when the environment becomes safer.
That usually means:
- reducing hepatic substrate overload
- stabilizing sleep and circadian signals
- improving fat oxidation capacity
- restoring genuine metabolic flexibility
- eating in a rhythm that matches physiology
- expanding stress capacity instead of tolerating more stress
This is not about perfection.
Or discipline.
Or willpower.
It’s about bringing metabolism back into a state where the body feels safe enough to relax.
When energy handling improves…
…the immune system stops holding the brake.
Inflammation doesn’t have to be suppressed —
—it simply becomes unnecessary.
The real takeaway
Chronic low-grade inflammation is not a mistake.
It is not noise.
It is not a random lab abnormality.
It is:
a biological strategy the body uses
to stabilize itself under energy and stress overload.
And if you understand why the body chose that strategy…
…you can help it choose a better one.
Not by fighting the signal —
—but by changing the conditions that created it.
Is chronic inflammation the same as acute inflammation?
No. Acute inflammation is a short-term immune response to injury or infection. Chronic low-grade inflammation is a persistent immune-metabolic signal reflecting ongoing metabolic load, stress, and energy imbalance rather than tissue damage or infection.
How does the gut–liver axis contribute to metabolic inflammation?
Signals from the gut — including microbial metabolites and small amounts of endotoxin such as lipopolysaccharide (LPS) — travel through the portal vein directly to the liver. This immune-metabolic cross-talk influences hepatic lipid handling and inflammatory signaling, contributing to chronic low-grade immune activation. Frontiers
Why is the TG:HDL ratio relevant to inflammation and metabolism?
The TG:HDL ratio reflects lipid export dynamics and insulin resistance. Studies support its use as a surrogate biomarker for metabolic dysfunction and its association with cardiometabolic risk independent of triglyceride or HDL levels alone. PMC+1
Can inflammation and insulin resistance occur together?
Yes — chronic low-grade inflammation and insulin resistance often co-exist and potentiate each other. Markers like hsCRP correlate with insulin resistance indices, and both independently contribute to cardiometabolic risk. PMC
What lifestyle patterns contribute to chronic metabolic inflammation?
Circadian misalignment, frequent eating windows, high substrate load (e.g., refined carbohydrates and excess energy), poor sleep, and stress hormone patterns can all contribute to systemic immune-metabolic signaling that manifests as chronic low-grade inflammation.
Author bio
Morteza Ariana is a Functional Nutrition Practitioner specializing in insulin resistance, type 2 diabetes, and systems-based metabolic restoration. His work focuses on identifying upstream drivers of metabolic dysfunction — including insulin load, liver–gut axis disruption, circadian misalignment, and micronutrient gaps — rather than masking symptoms.
He works with high-performing professionals through a structured 12-week Metabolic Restoration Blueprint designed to restore metabolic flexibility and long-term resilience.
If this resonates, the next step is clarity.
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Scientific References
Inflammation as a metabolic adaptation
- Chronic low-grade inflammation is a hallmark of metabolic disorders such as obesity, type 2 diabetes, and NAFLD, linking metabolism and immune signaling early in disease progression. Thieme Connect
- Obesity-associated systemic inflammation contributes to insulin resistance, β-cell dysfunction, and multiple metabolic comorbidities, including NAFLD and cardiovascular disease. ScienceDirect
Gut–liver axis, microbiota, and LPS/endotoxin signaling
- The gut–liver axis plays a central role in metabolic regulation; immune-metabolic cross-talk via microbial metabolites — including LPS — is implicated in chronic low-grade inflammation, obesity-related metabolic disorders, and insulin resistance. Frontiers+1
- Intestinal microbial products can modulate host metabolism and inflammatory signaling, contributing to chronic metabolic inflammation in obesity and insulin resistance. PMC
TG:HDL ratio as an insulin resistance biomarker
- The TG/HDL ratio is supported by systematic analyses as a simple surrogate biomarker for insulin resistance across diverse populations. PMC
- Elevated TG/HDL ratio is linked to metabolic syndrome components and cardiometabolic risk indicators independent of individual triglyceride or HDL levels. nature.com
Inflammation + insulin resistance interactions
- High-sensitivity C-reactive protein (hsCRP) and insulin resistance metrics show overlapping effects on cardiometabolic risk, indicating a physiological interaction between chronic inflammation and metabolic dysfunction. PMC