I see this every week: people eating less, moving more, tracking everything — and their metabolism still won’t cooperate.
The problem isn’t calories.
It’s insulin signaling.
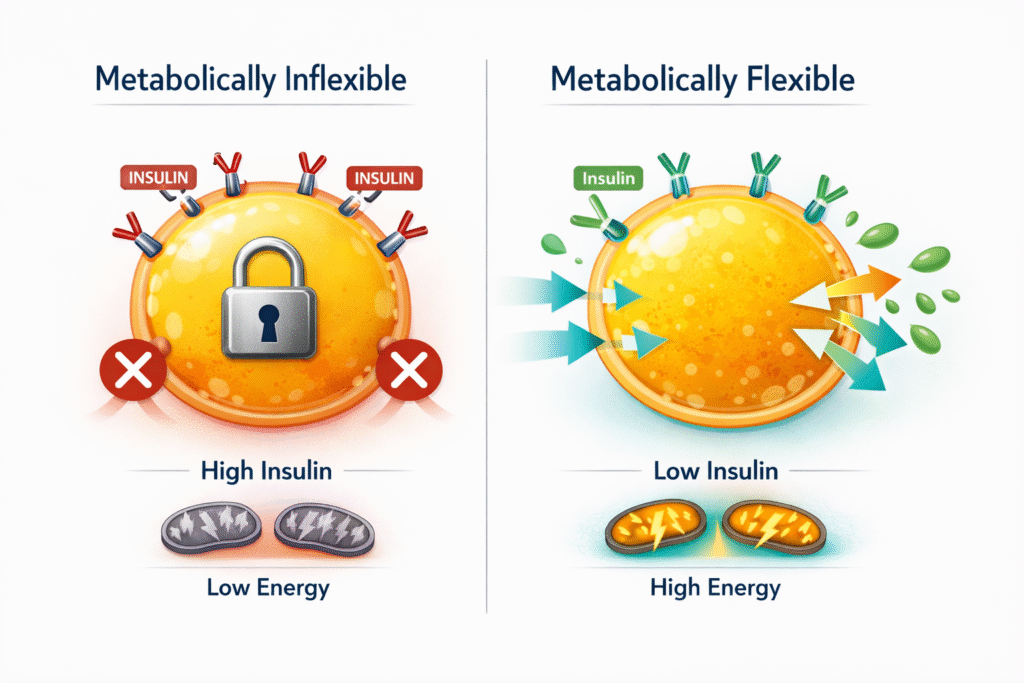
If your body cannot switch between burning glucose and burning fat, you are metabolically inflexible — and no amount of discipline fixes that long term.
If insulin stays elevated, fat cells stay closed.
The Real Problem Isn’t Weight — It’s Fuel Switching
Metabolic flexibility is the ability to switch between fuels depending on availability.
Eat carbohydrates → burn glucose.
Don’t eat for several hours → burn stored fat.
That’s how a healthy system works.
But when insulin rarely falls — because of frequent eating, refined foods, liquid calories, stress, low muscle mass — the body loses access to stored energy.
You are forced to run on sugar, even when fat is available.
The Pattern I Keep Observing
In clinic, I don’t see lazy patients.
I see exhausted ones.
They wake up hungry. Crash mid-morning. Crave carbs in the afternoon. Feel cold between meals. Gain fat centrally while arms and legs stay relatively lean.
Their glucose may still be “normal.”
Their HbA1c may still be under 6.0.
But fasting insulin is elevated. Triglycerides creep up. HDL drifts down. Waist circumference expands before the scale changes.
The metabolic shift begins years before diabetes is diagnosed.
A Real Case: 38, Cook, Germany
Seven months ago, a 38-year-old female cook in Germany came to me frustrated.
She worked long kitchen shifts surrounded by food. She ate frequently. Convenience snacks between tasks. No resistance training, but she walked all day.
Labs before:
- Fasting glucose: 107 mg/dL
- HbA1c: 5.9% (40.4 mmol/mol)
- Triglycerides: 96.9 mg/dL
- HDL: 45.2 mg/dL
- LDL: 151 mg/dL
- Total cholesterol: 206 mg/dL
She had central fat gain and described feeling “puffy,” hungry, and mentally foggy.
This was early metabolic dysfunction — not a motivation failure.
What We Changed
We removed junk food.
Stopped grazing between meals.
Prioritized protein at each meal.
Replaced seed oils with butter, ghee, and coconut oil.
Maintained her walking volume.
No calorie tracking.
No extreme dieting.
No new gym program.
We lowered chronic insulin exposure.
Seven Months Later
Labs after:
- Fasting glucose: 81 mg/dL
- HbA1c: 29.9 mmol/mol (~4.9%)
- Triglycerides: 68.7 mg/dL
- HDL: 56.4 mg/dL
- LDL: 164 mg/dL
- Total cholesterol: 228 mg/dL
Her waist changed before her weight did. The pear-shaped distribution disappeared. Hunger normalized. Energy stabilized.
Insulin signaling improved and lipid transport adapted.
The LDL Panic Moment
Halfway through this transformation she looked at her lipid panel and said:
“Why is my cholesterol higher when I feel better?”
That is a clinical moment you don’t see in textbooks.
Triglycerides dropped. HDL rose. Glucose normalized. But LDL increased modestly.
If you only track LDL in isolation, you misinterpret the physiology.
Context determines meaning.
What Actually Happens During Metabolic Repair
When insulin falls, triglycerides usually drop first.
HDL rises next.
Waist circumference improves before scale weight does.
During increased fat mobilization, LDL can rise because lipid trafficking increases.
This is not pathology by default. It is transport.
I’ve watched this sequence unfold across dozens of real patients. It has a pattern. It has timing. It has context.
Lab interpretation without longitudinal observation is guesswork.
Why Calorie Culture Fails
Calorie restriction without insulin regulation often produces short-term weight loss followed by metabolic slowdown.
Chronic caloric deficit without addressing signaling leads to:
- Increased hunger
- Reduced thyroid output
- Lower resting energy expenditure
- Eventual rebound weight gain
Energy balance matters. But it is downstream of hormonal regulation.
You cannot out-calculate dysregulated insulin.
What Metabolic Flexibility Actually Means
Metabolic flexibility means:
You can go several hours without hunger.
You can burn stored fat between meals.
You do not experience energy crashes after eating.
Your triglyceride/HDL ratio reflects stable lipid handling.
It is a sign of mitochondrial efficiency and appropriate insulin signaling.
When it’s lost, the body defaults to glucose dependence.
You are metabolically stuck, not metabolically broken.
The Silent Phase Before Diabetes
One of the most dangerous myths is that normal glucose equals metabolic health.
You can have:
- Normal HbA1c
- Normal fasting glucose
- Elevated fasting insulin
- Rising triglyceride/HDL ratio
- Increasing visceral fat
This phase can last 5–10 years.
By the time glucose rises clearly, insulin resistance has already been present.
Screening glucose alone misses the problem.
The Clinical Sequence I Watch For
In early metabolic dysfunction, the pattern often unfolds like this:
- Frequent eating
- Chronic hyperinsulinemia
- Reduced metabolic flexibility
- Central fat gain
- Rising triglycerides
- Lower HDL
- Elevated fasting glucose
This is predictable.
Intervening early prevents downstream disease.
If Your Labs Show This Pattern
If your labs show:
- Fasting glucose above 95–100
- TG/HDL ratio above 2
- Central fat accumulation
- Energy instability
This is exactly what we address inside the Functional Medicine Nutrition Blueprint.
We restore insulin rhythm.
We improve lipid handling.
We rebuild metabolic flexibility.
We do not chase single numbers. We repair signaling.
Strategy beats restriction.
Why This Cannot Be Solved by “Eating Less”
Eating less while insulin remains elevated simply increases stress signaling.
You feel deprived.
You feel hungry.
You feel like you failed.
But the physiology was never corrected.
When insulin normalizes, hunger stabilizes naturally.
Fat loss becomes a consequence, not a fight.
Hormones govern appetite more than willpower does.
The Hard Truth
Modern environments are designed to keep insulin elevated:
Constant food access.
Liquid carbohydrates.
Refined industrial oils.
Low muscle engagement.
Disrupted circadian rhythm.
This is not a moral issue. It is a biological mismatch.
Your metabolism adapted to the environment you placed it in.
The Real Question
When I look at labs, I don’t ask:
“How do we lower weight?”
I ask:
“Can this body switch fuels?”
If the answer is no, we correct signaling first.
Because fat burning is not a trick.
It is a metabolic state.
So here is the question that matters:
Is your metabolism flexible — or are you trapped in sugar-burning mode?
And if one lab marker moves in the “wrong” direction while the entire system improves — are you looking at disease… or adaptation?
People Also Ask
What is metabolic flexibility?
Metabolic flexibility is the ability of the body to switch between burning glucose and burning fat depending on energy availability. In a healthy metabolic state, the body uses carbohydrates after meals and shifts toward fat oxidation between meals or during fasting. Loss of this flexibility is commonly seen in insulin resistance, obesity, and type 2 diabetes.
What causes metabolic inflexibility?
Metabolic inflexibility is usually driven by chronically elevated insulin levels. Frequent eating, refined carbohydrates, sedentary lifestyle, sleep disruption, and excess visceral fat can keep insulin elevated throughout the day. When insulin remains high, fat cells cannot release stored energy, and the body becomes dependent on glucose as its primary fuel.
How do you know if you are metabolically inflexible?
Common indicators include persistent hunger, energy crashes between meals, difficulty losing fat, and central fat accumulation. Laboratory markers may include elevated fasting glucose, elevated fasting insulin, high triglycerides, low HDL cholesterol, and a triglyceride-to-HDL ratio above 2. These markers often appear years before diabetes is diagnosed.
Can metabolic flexibility be restored?
Yes. Metabolic flexibility can often be restored by improving insulin signaling and mitochondrial function. Strategies include prioritizing protein intake, reducing ultra-processed foods, spacing meals to allow insulin to fall, building muscle mass through resistance training, and maintaining regular physical activity. Over time, these changes allow the body to access stored fat again.
Why can LDL cholesterol increase when metabolic health improves?
During metabolic improvement, triglycerides often decrease and HDL increases as insulin levels fall. In some individuals, LDL cholesterol may temporarily rise because increased fat mobilization leads to greater lipid transport in the bloodstream. Interpreting LDL in isolation can therefore be misleading; it should always be evaluated within the broader metabolic context.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Scientific References
Galgani et al., 2008 — Metabolic flexibility and insulin resistance
Metabolic Inflexibility in Obesity and Type 2 Diabetes, Goodpaster & Sparks, 2017 — Cell Metabolism
Plasma triglyceride/HDL-cholesterol ratio, insulin resistance, and cardiometabolic risk in young adults, Murguía-Romero et al., 2013 — Journal of Lipid Research
Metabolic Flexibility and Its Impact on Health Outcomes, Palmer et al., 2022 — Mayo Clinic Proceedings
Mitochondrial Function and Insulin Resistance, Sergi et al., 2019 — Frontiers in Physiology
