Many people eventually begin to ask a difficult question: why do doctors treat symptoms instead of causes?
I have heard this frustration countless times from individuals struggling with fatigue, weight gain, unstable glucose, or persistent metabolic issues. Their lab values may still fall within conventional ranges, yet something in their physiology clearly feels wrong.
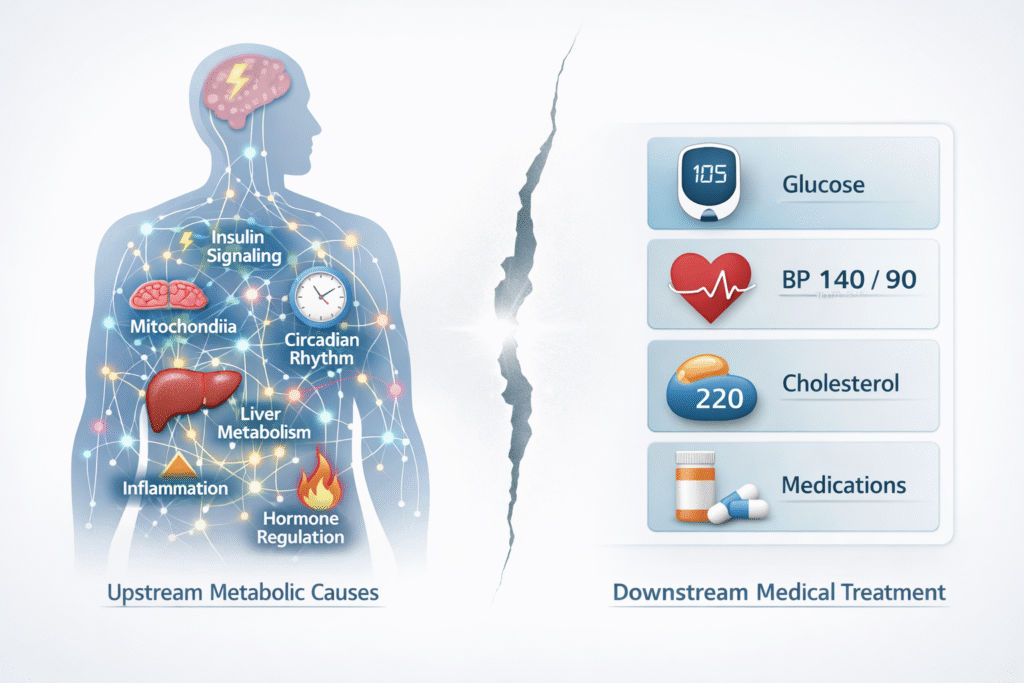
The reality is rarely that physicians are careless or uninterested. The deeper issue is that modern healthcare systems are often designed to react to downstream signals rather than investigate the upstream biological processes that created them. Understanding this distinction is essential if we want to truly understand metabolic health.
There is a pattern I have seen again and again in people struggling with metabolic dysfunction.
They feel tired, inflamed, hungry too often, foggy, heavier than they should be, less resilient than they used to be, and somehow not fully themselves. They know something is off. Yet they go from appointment to appointment and are told that everything looks fine, or at least fine enough. A standard lab panel may not trigger alarm. A drug may be offered for one isolated marker. A brief recommendation to eat more vegetables, move more, and worry less may follow. But the person still feels unwell.
I do not believe this happens primarily because individual doctors do not care. In many cases, they care deeply. The deeper problem is that modern medicine is often trained, organized, and reimbursed to react to downstream changes rather than investigate upstream biology. It is excellent at crisis care, emergency intervention, and disease management. It is far less effective when the problem is slow, layered, adaptive, and metabolic.
This is why so many patients feel unseen. And it is why a more thoughtful model of metabolic health is needed.
Clinical Perspective: What I See in Practice
In over a decade of clinical practice, the pattern that arrives most consistently is not dramatic. It is ordinary — and that is precisely what makes it so consequential. A patient comes in carrying a standard panel that has been reviewed, signed off, and filed. Fasting glucose: borderline but acceptable. Total cholesterol: flagged, statin discussed. Everything else: within normal range. The appointment lasted seven minutes. The patient was told to watch their diet.
What that panel also contains — sitting unremarked in the same results — is a GGT above the functional concern threshold of 20–30 U/L, an ALT trending toward 35 U/L, a TG/HDL ratio above 2.0 that has never been calculated because no one divided those two numbers, and a fasting glucose above the functional optimal of 85 mg/dL that is being monitored rather than investigated. Fasting insulin is not on the panel at all. It was never ordered. In Germany, in the majority of GP consultations I am aware of through my patients’ histories, it is almost never ordered — and when it does appear, it receives no clinical attention.
This is not a panel with nothing to say. This is a panel that is communicating early metabolic dysfunction across four separate markers simultaneously — and the communication is being missed at every level. Not because the GP does not care. But because the training, the time, and the reimbursement structure of a standard consultation do not create the conditions for this kind of cross-marker pattern recognition to occur.
What I explain to these patients — often for the first time in their clinical history — is that fasting insulin is the single most important metabolic marker that standard medicine almost never measures. It rises years, sometimes a decade, before blood sugar becomes abnormal. By the time HbA1c crosses the conventional diagnostic threshold for prediabetes, the pancreas has already been compensating with excess insulin secretion for years. GGT has already been trending upward. The TG/HDL ratio has already been above 2.0 for panel after panel. The liver has already been accumulating fat. The entire upstream trajectory has been visible in the numbers. No one connected them.
I then explain what each marker is actually measuring — not as a lecture, but in direct answer to why they feel the way they feel. Why the afternoon fatigue is real. Why the brain fog is metabolic, not psychological. Why the abdominal weight that will not shift is not a willpower problem but a hormonal one driven by chronically elevated insulin. And I explain what the intervention needs to address: not blood sugar, which is the downstream consequence, but the insulin load, the dietary quality, and the gut-liver axis disruption that is producing the entire picture.
That conversation — connecting the markers to the mechanism to the symptoms to the solution — is frequently the first time these patients have understood what is happening in their own body. And that understanding is what makes genuine engagement with the protocol possible.
The most important clinical distinction I draw is this: the diagnostic system is not failing these patients through misdiagnosis. It is failing them through non-diagnosis. The signals are in the panel. They are just not being read. And by the time they finally are — when HbA1c has crossed the threshold and a T2D diagnosis is made — the window for the most complete and durable metabolic recovery has already narrowed considerably. That is the cost of treating downstream markers while the upstream biology goes unread.
The Frustrating Experience Many Patients Share
Many patients arrive with a very similar story. They say, “I know my body. Something has changed.” They may have worsening energy, central fat gain, unstable hunger, poor sleep, brain fog, elevated triglycerides, higher fasting glucose than before, rising liver enzymes, or a sense that recovery is no longer normal. Yet because they do not yet meet a formal disease threshold, they are often told to wait, watch, or simply live a little healthier.
I understand why this happens. Conventional medicine is built around diagnosis, and diagnosis usually requires thresholds. But biology begins to shift long before a diagnosis appears. Insulin resistance does not suddenly start the day someone crosses into diabetes. Fatty liver does not begin the moment imaging finally detects it. Mitochondrial strain, circadian disruption, inflammatory signaling, and altered substrate handling can be present for years before the system formally names the problem.
This creates a painful gap between lived experience and medical recognition. The patient feels the biology before the chart reflects it.
How Modern Medicine Became a Downstream Model
Modern medicine did not become this way by accident. It evolved under enormous pressure to solve urgent, visible, life-threatening problems. Acute infections, trauma, organ failure, stroke, heart attack, severe autoimmune flares, and advanced disease demand rapid, decisive action. In that setting, downstream markers and emergency interventions save lives.
That model is powerful. But it does not translate perfectly to chronic metabolic disease.
Metabolic dysfunction is usually not explosive. It is gradual. It is often nonlinear. It affects multiple systems at once: glucose handling, liver function, hunger signaling, sleep, inflammation, mitochondrial output, body composition, reproductive hormones, vascular tone, and cognitive performance. By the time a clearly defined downstream abnormality appears, the upstream process may have been active for years.
I think this is one of the great mismatches in modern healthcare: the model is optimized for dramatic pathology, while much of chronic disease develops quietly in the background.
So what gets measured most often? The later signals. The obvious endpoints. The formal diagnoses. The biomarker that has crossed the line. The prescription target.
What gets missed? The terrain that produced it.
Why Early Metabolic Signals Are Often Ignored
There are several reasons early signals get overlooked, and most of them are structural rather than personal.
The first is time. In many healthcare systems, including Germany, physicians often work under extraordinary patient volume. When appointments are short, the clinical encounter naturally shifts toward what can be addressed quickly: the main complaint, the obvious abnormality, the billable action, the immediate next step. But metabolic dysfunction rarely reveals its full story in a few rushed minutes. It requires pattern recognition across food quality, meal timing, sleep, circadian rhythm, stress load, body composition, training, recovery, family history, and biomarker trends over time.
The second is incentive design. Healthcare systems tend to reward procedures, prescriptions, and standardized interventions more clearly than long conversations about diet, sleep, stress physiology, and behavior change. That does not mean physicians are malicious. It means systems pull attention toward what they measure and reimburse.
The third is culture. The dominant strategy for chronic disease has become increasingly medication-centered. Drugs can absolutely play an important role. But when the entire model becomes medicamentös, the patient may never receive a serious physiological explanation of why the problem emerged in the first place.
And finally, nutritional guidance itself is often oversimplified. Generic advice may sound harmless, but vague slogans are not enough for a person with significant insulin resistance, fatty liver risk, appetite dysregulation, or chronic metabolic inflexibility. These patients need physiology, not slogans.
The Difference Between Biomarkers and Biology
This distinction matters enormously.
A biomarker is not the biology itself. It is a signal. A clue. A snapshot. Sometimes a late snapshot.
One of the most common errors in medicine is to confuse numerical control with physiological resolution. A lab value may improve while the upstream process remains disturbed. A symptom may be suppressed while the metabolic stressor continues. A diagnosis may be delayed because the marker has not yet crossed the threshold, even though the biology is already deteriorating.
I often think of biomarkers as dashboard lights. They are useful, sometimes essential, but they are not the engine. If we only react once the warning light becomes bright enough, we are already late.
This is why I place so much importance on context and trend interpretation. Fasting insulin, triglyceride-to-HDL relationship, postprandial tolerance, GGT, ALT, waist change, energy variability, sleep quality, and hunger dynamics can tell us far more when viewed together than when interpreted as isolated fragments. Biology is relational. Most conventional medicine is still too compartmentalized.
Science supports this systems view strongly. Metabolic dysfunction emerges through interacting networks: insulin signaling, ectopic fat accumulation, hepatic stress, inflammatory tone, mitochondrial inefficiency, autonomic imbalance, and circadian disruption. No single biomarker can carry that whole story alone.
Metabolic Disease Is a Systems Problem
This is the center of the whole article.
Metabolic disease is not just a glucose problem. It is not just a weight problem. It is not just a willpower problem. And it is certainly not just a prescription problem.
It is a systems problem.
Insulin resistance alters fuel partitioning. Sleep loss worsens glucose control and appetite regulation. Circadian disruption affects cortisol rhythm, mitochondrial timing, and insulin sensitivity. Liver stress changes glucose output and lipid trafficking. Sedentary behavior reduces glucose disposal. Chronic overexposure to hyperpalatable processed food distorts satiety, energy intake, and inflammatory tone. Loss of muscle mass reduces metabolic buffering capacity. Stress biology changes behavior, recovery, and substrate use.
This is why reductionism fails here. You cannot fully understand a systems disease by treating each downstream fragment as if it were independent.
I have seen this repeatedly: one clinician looks at glucose, another at lipids, another at blood pressure, another at fatigue, another at body weight, and the patient is left carrying a dozen disconnected labels. But the body is not running twelve separate diseases. Often it is running one deteriorating metabolic pattern expressing itself through multiple channels.
That changes everything. It changes how we listen. It changes what we measure. It changes what we prioritize. And it changes how early we intervene.
Why This Gap Makes Patients Feel Gaslit
Most patients do not use the word gaslit lightly. They use it when their lived reality is repeatedly minimized.
They are tired, but told their labs are normal.
They are gaining central fat, but told aging is the issue.
They are constantly hungry, but told simply to eat less.
They are metabolically deteriorating, but told to come back when the problem becomes clearer.
This creates an emotional injury on top of the biological one. The patient begins to distrust not only the system, but sometimes their own perception.
I want to be careful here. The answer is not physician-bashing. Many clinicians are trying to do good work inside a model that leaves little room for nuance. But from the patient’s side, the experience can still feel invalidating. The system often waits too long, sees too little, explains too little, and fragments what should have been understood as one pattern.
When a person senses that something is wrong and repeatedly hears that nothing meaningful is wrong, trust erodes. And once trust erodes, care becomes harder for everyone.
A Better Way to Think About Metabolic Health
A better model begins with humility. It recognizes that the body gives signals long before disease labels appear. It respects symptoms, trends, context, and physiology. It does not wait for collapse before becoming interested.
This does not mean rejecting all of modern medicine. That would be foolish. Modern medicine can be extraordinary. It means using a broader lens. We need a model that asks upstream questions earlier: How is the person sleeping? What is their meal pattern? How metabolically flexible are they? What do their hunger rhythms tell us? What is happening with liver stress, visceral fat, muscle mass, circadian timing, glucose excursions, and recovery capacity?
It also means restoring diet and lifestyle to the center of metabolic medicine rather than treating them as decorative advice added after the prescription. In many cases, food quality, meal timing, sleep restoration, resistance training, walking, stress reduction, and body composition improvement are not secondary. They are foundational biology.
And perhaps most importantly, we need to speak to patients in a way that restores trust. Thoughtful medicine does not dismiss. It interprets. It investigates. It explains. It connects dots.
That is the real purpose of this final pillar article. To close the loop. To show that many people were never imagining their dysfunction. They were feeling upstream biology in a downstream system.
People Also Ask
Why do doctors often treat symptoms instead of underlying causes?
Modern healthcare systems are largely designed around diagnosing and managing measurable disease states. This often leads clinicians to address downstream markers such as glucose, cholesterol, or blood pressure rather than investigating the upstream metabolic processes that produced them.
Can metabolic disease develop before blood tests become abnormal?
Yes. Metabolic dysfunction can develop years before conventional diagnostic thresholds are crossed. Early changes such as hyperinsulinemia, liver stress, or reduced metabolic flexibility may exist long before conditions like type 2 diabetes are formally diagnosed.
What does “downstream noise” mean in medicine?
Downstream noise refers to late-stage signals of disease such as abnormal biomarkers or symptoms. These signals often appear after deeper biological disturbances—such as insulin resistance or mitochondrial stress—have been developing for years.
Why is metabolic disease considered a systems problem?
Metabolic disease affects multiple interconnected systems including insulin signaling, liver metabolism, inflammation, sleep regulation, and energy balance. Because these systems interact, focusing on a single biomarker rarely captures the full biological picture.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Vaidya RA, Desai S, Moitra P, et al. Hyperinsulinemia: an early biomarker of metabolic dysfunction. Frontiers in Clinical Diabetes and Healthcare. 2023;4:1159664. 🔗 https://pmc.ncbi.nlm.nih.gov/articles/PMC10186728/
2. Masenga SK, Kabwe LS, Chakulya M, Kirabo A. Metabolic dysfunction biomarkers as predictors of early diabetes. Biomolecules. 2021;11(11):1589. 🔗 https://www.mdpi.com/2218-273X/11/11/1589
3. Ferrer-García JC, Romero-Roca P, Abellán-Huerta J, et al. Early identification of metabolic syndrome risk: a review of reviews and proposal for defining pre-metabolic syndrome status. Nutrition, Metabolism and Cardiovascular Diseases. 2021;31(8):2237–2246. 🔗 https://www.sciencedirect.com/science/article/pii/S0939475321002453
4. Zhao W, Zhang X, Liang Y, et al. Trends in insulin resistance: insights into mechanisms and therapeutic strategy. Signal Transduction and Targeted Therapy. 2022;7(1):216. 🔗 https://www.nature.com/articles/s41392-022-01073-0
5. Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiological Reviews. 2018;98(4):2133–2223. 🔗 https://pubmed.ncbi.nlm.nih.gov/30067154/
6. Liang H, Ward WF. A conceptual review on systems biology in health and diseases: from biological networks to modern therapeutics. PMC. 2014. 🔗 https://pmc.ncbi.nlm.nih.gov/articles/PMC3933630/
