
Why treating symptoms while ignoring insulin is failing millions of women — and what the evidence actually says.
Over the course of my work with women struggling with PCOS, I have noticed a consistent pattern: by the time they reach me, most have been told the same things. Take the pill to regulate your cycle. Manage your weight. Come back if you want to get pregnant. What they were rarely told — and what changed everything for them — was this: the root driver of their condition was not their ovaries, not their hormones in isolation, and certainly not a lack of willpower. It was insulin.
Polycystic Ovary Syndrome affects an estimated 8 to 13 percent of women of reproductive age worldwide. It is the most common endocrine disorder in women. And yet, in standard clinical practice, it is still predominantly managed as a hormonal or gynaecological condition — when the evidence has been telling us for decades that metabolic dysfunction, specifically hyperinsulinemia and insulin resistance, is the dominant driver.
This post is for two audiences: clinicians who want a sharper evidence-based framework, and women with PCOS who are tired of being managed rather than genuinely treated.
The Conventional Story — and Where It Falls Short
The standard diagnostic criteria for PCOS — the Rotterdam criteria — require two of three features: irregular ovulation, clinical or biochemical signs of hyperandrogenism, and polycystic ovarian morphology on ultrasound. Insulin resistance does not appear in the diagnostic criteria at all. This is a significant clinical blind spot.
The result is that women are diagnosed based on downstream symptoms — the irregular cycles, the elevated androgens, the cysts — while the upstream metabolic driver continues unchecked. Treatments are then aimed at those symptoms: oral contraceptives to regulate the cycle, spironolactone for androgen excess, metformin in some cases. These approaches can be useful, but they do not address the underlying dysfunction. When the medication stops, the condition returns.
Conclusion: Treating PCOS without addressing insulin resistance is treating the alarm without investigating the fire. Symptom control is not the same as metabolic recovery.
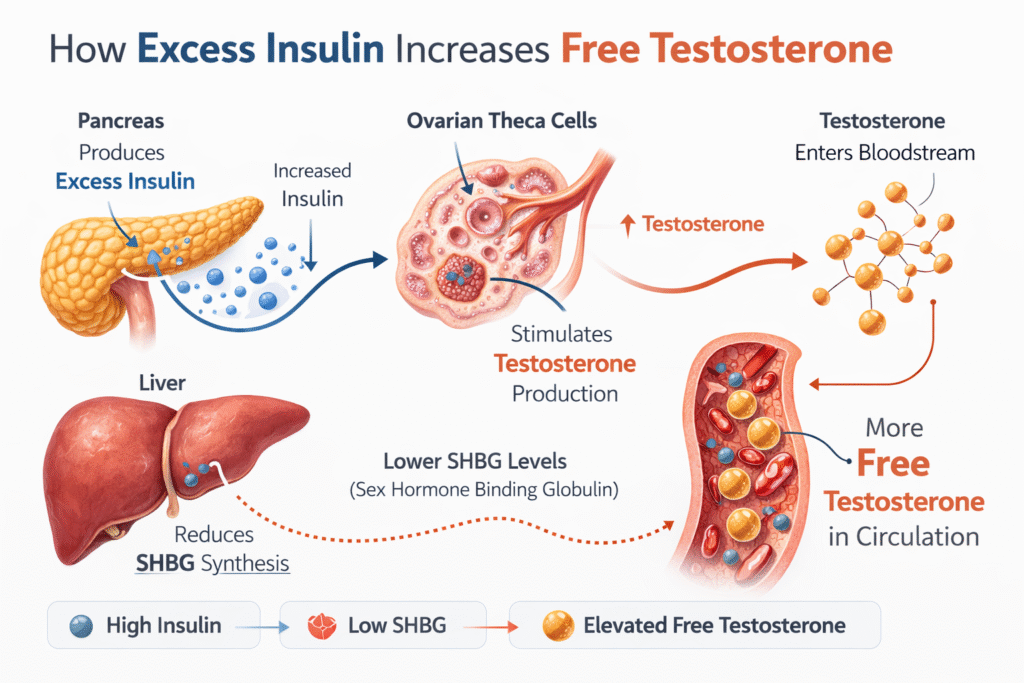
The Insulin-Androgen Loop The core hyperinsulinemia → androgen excess mechanism
How Insulin Drives the Condition — The Mechanism
The physiological chain linking hyperinsulinemia to PCOS is well-established. Insulin acts on the ovarian theca cells to stimulate androgen production — particularly testosterone. Simultaneously, elevated insulin suppresses hepatic synthesis of sex hormone-binding globulin (SHBG), the protein that binds and neutralises circulating androgens. The result is a double mechanism: more androgens being produced, and fewer being bound and inactivated. Bioavailable testosterone rises, and the clinical signs of hyperandrogenism follow.
Insulin also disrupts the delicate hormonal signalling required for normal follicular development. In a healthy cycle, follicles mature sequentially, with a dominant follicle responding to luteinising hormone (LH) at around 9.5mm and proceeding to ovulation. In the presence of chronic insulin excess, follicles respond to LH prematurely — at just 4mm — and development becomes arrested. The ovary fills with small, immature follicles. This is not the cause of PCOS. It is the consequence of a metabolic environment that insulin has fundamentally altered.
The adrenal gland is also implicated. Insulin stimulates adrenal androgen production, adding a second source of androgen excess beyond the ovary. Meanwhile, the hypothalamic-pituitary axis is disrupted, with elevated LH pulsatility further compounding the ovulatory dysfunction. This is not a single-organ problem. Hyperinsulinemia creates a systemic hormonal environment that is hostile to normal reproductive function.
Conclusion: Hyperinsulinemia does not merely correlate with PCOS — it mechanistically drives androgen excess, suppresses SHBG, disrupts follicular development, and dysregulates the HPG axis. The evidence for this pathway is robust.
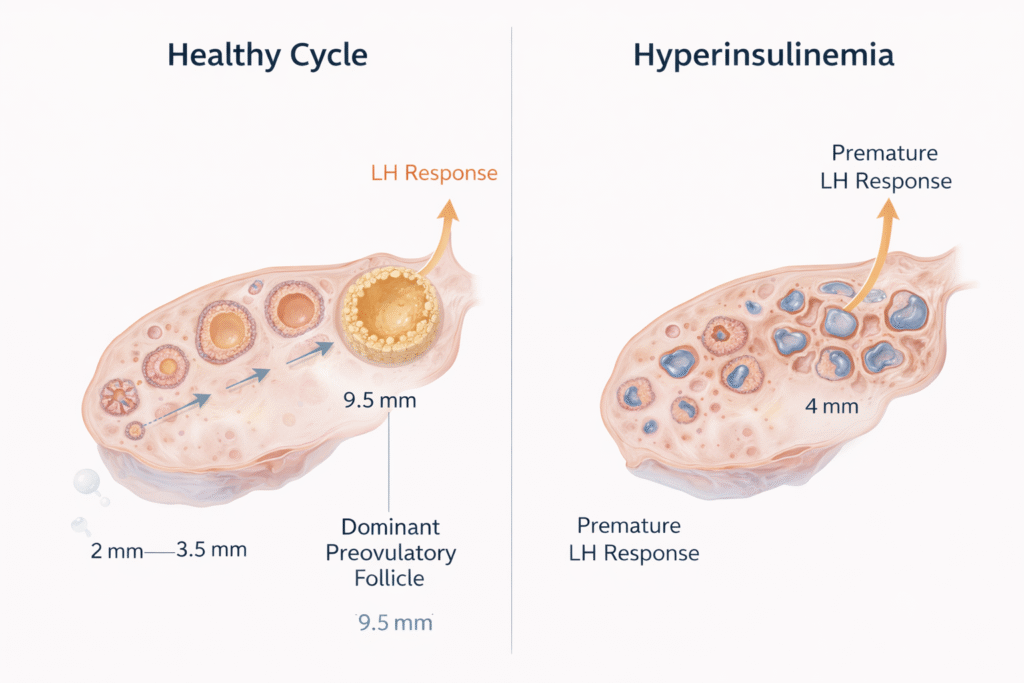
Follicle Development Arrested Normal vs PCOS follicular development comparison
PCOS Is Not Just an Obese Woman’s Condition
One of the most persistent and damaging misconceptions about PCOS is that it is primarily a condition of obesity. This framing causes two problems: it leads clinicians to underdiagnose PCOS in lean women, and it causes women at normal weight to be dismissed entirely when they present with classic symptoms.
The data tells a different story. While PCOS prevalence does increase with BMI — rising sharply from overweight to moderately and extremely obese categories — it is present across all weight classes, including underweight and normal-weight women. What matters is not body weight per se, but metabolic phenotype. A lean woman with high visceral adiposity, poor insulin sensitivity, and a diet driving chronic hyperinsulinemia can have as severe a PCOS presentation as a woman with a BMI of 35.
The glucose tolerance data within PCOS populations reinforces this. Even at a BMI below 25, a meaningful proportion of women with PCOS demonstrate impaired glucose tolerance or frank type 2 diabetes. As BMI increases beyond 30, the proportion with normal glucose tolerance falls below 50 percent. The metabolic burden of this condition is significant and is not adequately captured by weight alone.
Conclusion: PCOS is a metabolic condition that expresses across all body weights. Dismissing lean women, or focusing exclusively on weight loss in heavier women, misses the underlying insulin dysfunction that needs to be addressed.
What Drives the Insulin Resistance? Diet and Lifestyle.
Insulin resistance does not appear from nowhere. In the majority of women with PCOS, it is the product of a sustained dietary pattern that chronically elevates insulin — one dominated by refined carbohydrates, ultra-processed foods, and high glycaemic load — compounded by insufficient physical activity, disrupted sleep, and chronic psychological stress.
In my clinical experience, when I investigate the dietary histories of women presenting with PCOS, the pattern is remarkably consistent: years of blood glucose volatility driven by poor nutritional architecture, with the body compensating by producing ever-increasing amounts of insulin to manage the glucose load. Over time, cells become less responsive to insulin’s signal. The pancreas compensates further. Hyperinsulinemia becomes the new baseline — and the ovarian and adrenal consequences follow.
This matters because it means PCOS is, in many cases, a condition with a modifiable root cause. It is not simply a genetic fate. The genetic predisposition may exist, but the dietary and lifestyle environment determines whether and how severely it expresses.
Conclusion: For most women with PCOS, the condition is diet and lifestyle-driven at its metabolic core. This is the most empowering clinical truth in this field — because it means it is addressable.
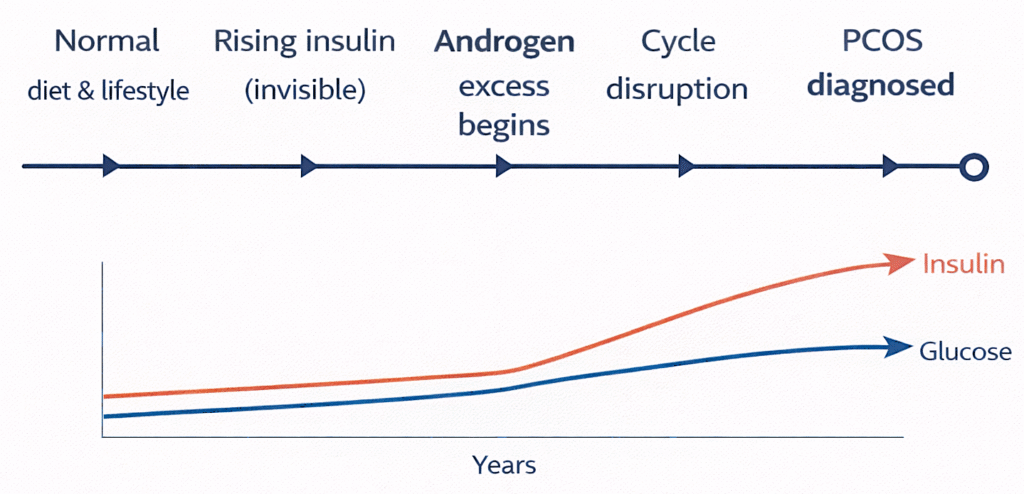
The Silent Timeline Years of compensated hyperinsulinemia before diagnosis
What I Have Observed in Practice — And What Is Possible
I have worked with women who had been told, sometimes for years, that their only path to pregnancy was IVF. Women who had been cycling through contraceptives without anyone explaining why their body was behaving the way it was. What I observed consistently was that when the metabolic root — hyperinsulinemia — was genuinely addressed through targeted nutritional intervention, sleep optimisation, and lifestyle restructuring, the hormonal picture changed. Androgens fell. Cycles regularised. And in multiple cases, women who had been told they could not conceive naturally did exactly that.
This is not anecdote divorced from science. The evidence base for low-glycaemic and low-carbohydrate dietary interventions in PCOS is growing and consistent: improvements in insulin sensitivity, reductions in free androgen index, restoration of ovulatory function. These are not marginal effects. They are the expected physiological consequence of removing the primary metabolic stressor.
Conclusion: PCOS reversal — not just symptom management — is achievable when the metabolic root is treated directly. The evidence supports it. Clinical outcomes confirm it.
The Markers That Actually Matter
If you have PCOS — or suspect you do — the standard workup is often insufficient. A normal fasting glucose tells you almost nothing about your insulin status. The tests worth requesting or ordering are:
Fasting insulin — not just fasting glucose. Hyperinsulinemia precedes glucose dysregulation by years. You can have perfectly normal blood sugar while your insulin is chronically elevated and driving androgen excess.
HOMA-IR — a calculated index of insulin resistance using fasting glucose and fasting insulin. A value above 2 warrants attention.
Fasting triglycerides and HDL — the triglyceride-to-HDL ratio is one of the most accessible proxies for insulin resistance. A ratio above 2 in European populations is a meaningful signal.
SHBG — low SHBG reflects high insulin and predicts elevated bioavailable androgen even when total testosterone appears normal.
hsCRP — chronic low-grade inflammation is both a companion and amplifier of insulin resistance in PCOS.
Conclusion: If your PCOS workup did not include fasting insulin and HOMA-IR, your metabolic picture has not been properly assessed. These are the markers that reveal the root.
Where to Go From Here
If you are a woman with PCOS who has been managed symptomatically for years without resolution, or if you are a clinician who wants a more rigorous metabolic framework for this population, the starting point is the same: investigate insulin.
The condition is not a life sentence. I have seen the evidence in the literature, and I have seen it confirmed in practice. When the metabolic environment changes, the hormonal environment follows — and with it, the possibility of restored cycles, reduced androgen burden, and yes, natural pregnancy.
If you want to understand your own metabolic picture, or if you are ready to take a genuinely root-cause approach to your PCOS, I invite you to book a consultation. We start with the markers that matter, and we build from there.
PCOS is one of the most clinically recognizable expressions of the broader principle that many hormonal problems originate in metabolic dysfunction — a framework that reframes how reproductive, thyroid, and adrenal symptoms should be approached.
People Also Ask
Is PCOS caused by insulin resistance?
In the majority of cases, yes. Insulin resistance and the resulting excess insulin in the bloodstream — known as hyperinsulinemia — is the dominant metabolic driver of PCOS. It directly stimulates the ovaries to produce too much testosterone, disrupts normal follicle development, and suppresses the proteins that keep androgens under control. Treating PCOS without addressing insulin means treating the symptoms while ignoring the cause.
Can thin or normal-weight women have PCOS?
Beyond the standard hormone panel, ask specifically for fasting insulin, HOMA-IR, SHBG, fasting triglycerides, HDL cholesterol, and hsCRP. These markers reveal the metabolic picture that standard PCOS workups routinely miss. A normal fasting glucose does not rule out hyperinsulinemia — insulin can be chronically elevated for years before blood sugar moves at all.
Can PCOS be reversed naturally without medication?
For many women, yes — when the root cause is genuinely addressed. Targeted nutritional changes that reduce insulin load, combined with improved sleep, movement, and stress management, can restore ovulatory function, reduce androgen excess, and normalise cycles. I have worked with women who achieved natural pregnancy after being told IVF was their only option. Medication can play a supporting role, but it is not the only path.
Why does my doctor focus on the pill rather than insulin?
Because standard PCOS guidelines are built around symptom management — regulating the cycle, reducing androgen-related symptoms, and managing fertility when needed. Insulin resistance does not even appear in the official diagnostic criteria. This is a genuine gap in how the condition is conventionally approached. It does not mean your doctor is wrong — it means the framework most clinicians work within has not yet caught up with the metabolic evidence.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Selected References
Azziz R, et al. Polycystic ovary syndrome. Nature Reviews Disease Primers. 2016.
Dunaif A. Insulin resistance and the polycystic ovary syndrome: mechanism and implications for pathogenesis. Endocrine Reviews. 1997;18(6):774–800.
Franks S, et al. Development of polycystic ovary syndrome: involvement of genetic and environmental factors. International Journal of Andrology. 2006.
Nestler JE, Jakubowicz DJ. Decreases in ovarian cytochrome P450c17 alpha activity and serum free testosterone after reduction of insulin secretion in polycystic ovary syndrome. New England Journal of Medicine. 1996.
Moran LJ, et al. Dietary composition in restoring reproductive and metabolic physiology in overweight women with polycystic ovary syndrome. Journal of Clinical Endocrinology & Metabolism. 2003.
Teede HJ, et al. Recommendations from the international evidence-based guideline for the assessment and management of polycystic ovary syndrome. Human Reproduction. 2018.
