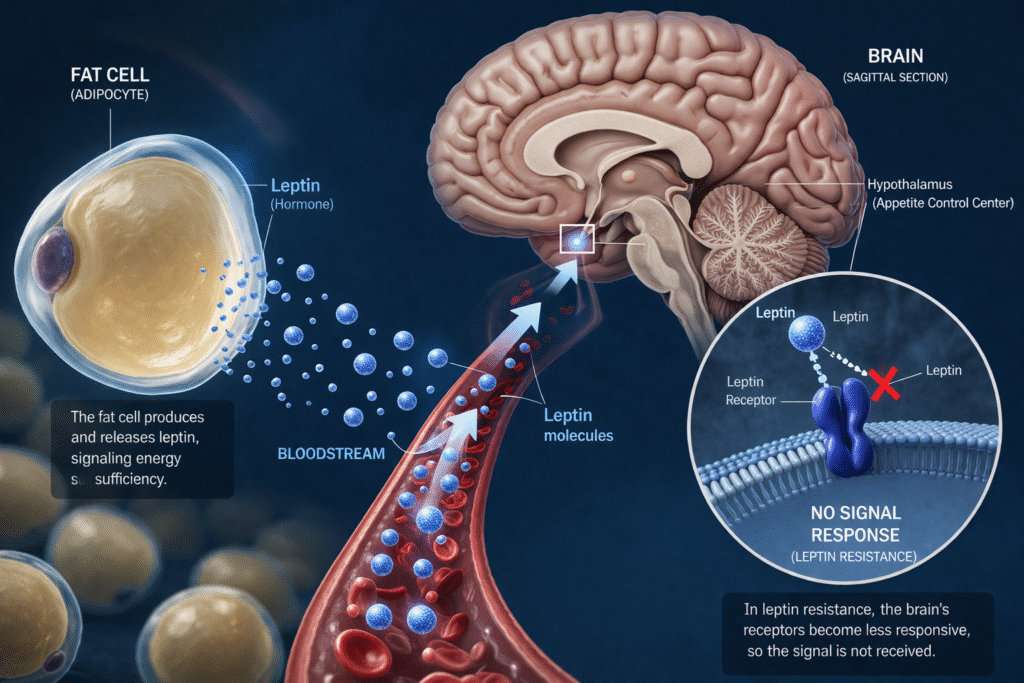
There is a hormonal signal that is supposed to tell your brain when you have stored enough fat. When fat mass rises above a threshold, fat cells release leptin into the bloodstream. Leptin travels to the hypothalamus, binds to its receptor, and sends a clear message: energy stores are sufficient, appetite should decrease, and metabolic rate should increase. In a metabolically healthy individual, this feedback loop regulates body weight with remarkable precision over months and years.
In leptin resistance, this signal is sent but never received. The fat cells are doing their job. The leptin is being produced — often at high levels, sometimes at very high levels. But the brain has stopped listening. The hypothalamus no longer responds to the signal. The feedback loop is broken. And without that signal arriving at its destination, the brain continues to behave as if the body is in a state of energy deficit — driving hunger, reducing energy expenditure, promoting fat storage, and defending against weight loss with every physiological mechanism at its disposal.
This is not a willpower problem. It is a signaling failure. And understanding the distinction between the two changes everything about how leptin resistance is approached, interpreted, and addressed.
What you will learn: What leptin is and what it is supposed to do in a functioning metabolic system | The cellular mechanisms through which leptin resistance develops | Why leptin resistance and insulin resistance almost always arrive together — and which one drives which | Why this mechanism is entirely absent from standard clinical practice and what markers reflect it indirectly | What the intervention looks like and how long it takes to restore signaling
Clinical Perspective: What I See in Practice
Leptin resistance does not exist in the conversation between a patient and their GP. It is even more absent from standard care than fasting insulin — and fasting insulin is hardly ever ordered in clinical practice. When GPs do not have time to interpret a TG/HDL ratio that is already in the panel in front of them, when ALT and GGT sitting above functional thresholds receive no clinical attention, the concept of central leptin resistance is genuinely not part of the clinical landscape. It is not a gap in knowledge. It is a gap in the entire framework of what standard medicine considers measurable and addressable.
The patients who arrive in this practice with leptin resistance as a significant driver look, in their symptom profile, like metabolic syndrome moving in the direction of obesity. Persistent hunger that does not resolve with what a reasonable meal should satisfy. Fatigue that is disproportionate to sleep quality. A sugar roller coaster — energy that rises briefly after eating and crashes within an hour or two, driving the next craving before the previous meal has been metabolized.
Weight that has been resistant to every conventional intervention — not because the patient lacks motivation, but because their hypothalamus is operating under a hormonal instruction set that makes fat mobilization physiologically difficult regardless of what they eat or how little of it.
What strikes me consistently in these consultations is the moment of recognition when I explain that their hunger is not a character trait. The brain genuinely believes there is insufficient energy stored. The leptin signal that should be correcting that belief is being generated by the fat cells — sometimes at very high circulating levels — but is not arriving at its destination in the hypothalamus. When I explain this directly — that the problem is not how much they eat, but that the signal telling the brain to stop wanting to eat is not getting through — the response is almost always the same. They ask why their GP has never explained this to them. And then they ask, practically and directly, what they need to do about it.
These are patients who are, to a significant degree, fed up with suppressing symptoms without seeing meaningful change. Many of them have watched their metabolic picture worsen on medications that were managing numbers without addressing the mechanism producing those numbers. Some of them have arrived specifically because they have heard of functional medicine and its upstream approach. They want the biology explained — not as a lecture, but as a direct answer to why their body is behaving the way it is. And the leptin resistance story, told correctly, is one of the most empowering explanations I can offer — because it reframes the entire experience from failure to physiology.
In practice, I do not confirm leptin resistance with a leptin measurement. Leptin testing is not part of standard panels and its interpretation requires context that a single value cannot provide. What tells me leptin resistance is operating is the clinical combination: the metabolic syndrome picture, the obesity or central adiposity, the out-of-range metabolic markers — elevated TG/HDL ratio, elevated fasting insulin when it has been measured, borderline or elevated ALT and GGT — and the symptomatic pattern of unresolvable hunger, fatigue, and weight resistance. Insulin resistance and leptin resistance arrive together in virtually every case I see.
I address the insulin resistance upstream, knowing that restoring insulin sensitivity in the liver and the periphery is the primary lever for restoring leptin sensitivity at the same time.
The objective signal that tells me the system is beginning to respond comes not from a single lab value or a single moment, but from the cumulative picture between weeks 8 and 12: the CGM trends stabilizing, the daily logs showing reduced hunger and improved satiety, the patient’s own report that food noise — the constant background pull toward eating — has significantly diminished. Visible physical changes in body composition follow on a similar timeline. There is no single moment. There is a direction that becomes unmistakable.
What Leptin Is and What It Is Supposed to Do
Leptin is a 16-kilodalton peptide hormone produced almost exclusively by adipocytes — white fat cells. Its production is directly proportional to fat mass: the more fat tissue a person carries, the more leptin their fat cells secrete. It is released primarily into the bloodstream during the fed state and during periods of positive energy balance, and it circulates to multiple target tissues — most importantly the hypothalamus, where it exerts its primary effects on appetite and energy expenditure.
In the hypothalamus, leptin acts on neurons in the arcuate nucleus — specifically on pro-opiomelanocortin (POMC) neurons, which it activates, and on agouti-related peptide (AgRP) neurons, which it suppresses. POMC neurons produce alpha-melanocyte-stimulating hormone (α-MSH), which binds to melanocortin 4 receptors (MC4R) in the paraventricular nucleus and sends a satiety signal — reducing appetite, increasing energy expenditure, and promoting a shift toward fat oxidation.
AgRP neurons produce the opposite signal — stimulating appetite and promoting energy conservation. Leptin activates the former and suppresses the latter, creating a coordinated central signal that communicates the body’s energy sufficiency to the brain.
The physiological consequence of this system operating correctly is elegant: as fat mass rises, leptin rises, POMC neurons are activated, AgRP neurons are suppressed, appetite falls, metabolic rate is maintained or increased, and fat mass returns toward its set point. As fat mass falls — during caloric restriction or fat loss — leptin falls, POMC neurons are suppressed, AgRP neurons are activated, appetite increases, metabolic rate is reduced, and the body resists further fat loss. Leptin is the central feedback signal in a long-loop energy homeostasis system that maintains body composition with remarkable stability over time in a healthy metabolic environment.
The downstream signaling pathway through which leptin acts in the hypothalamus involves the JAK2–STAT3 pathway. Leptin binds to its receptor (LepRb), activates Janus kinase 2 (JAK2), which phosphorylates STAT3, which translocates to the nucleus and drives transcription of the genes that produce the satiety response. This is the molecular chain that leptin resistance disrupts.
How Leptin Resistance Develops
In obesity and metabolic syndrome, circulating leptin levels are elevated — often substantially. This is the expected consequence of increased fat mass: more fat cells producing more leptin. But the downstream signaling response to that leptin is impaired. The hypothalamus no longer responds proportionally to the elevated leptin signal. The JAK2–STAT3 pathway is blunted. The satiety signal does not arrive with the intensity it should, despite the fact that leptin itself is present in abundance.
This is leptin resistance: high leptin, low leptin sensitivity. The signal is being sent. The receiver is not responding.
Several mechanisms contribute to this resistance, and they are not entirely independent of one another.
SOCS3 upregulation. One of the genes activated by the JAK2–STAT3 pathway is the suppressor of cytokine signaling 3 (SOCS3). SOCS3 is a negative feedback regulator — it inhibits JAK2 and reduces the sensitivity of the leptin receptor to subsequent signaling. In a normal metabolic context, this feedback is modest and self-limiting. In the context of chronically elevated leptin — as occurs in obesity — SOCS3 is constitutively upregulated, creating persistent suppression of leptin receptor signaling. The more leptin is secreted, the more the receptor is desensitized to it. This is the core molecular mechanism of central leptin resistance.
PTP1B activation. Protein tyrosine phosphatase 1B (PTP1B) is a phosphatase that dephosphorylates JAK2, terminating its activity and reducing downstream signaling. PTP1B is upregulated by chronic inflammation — specifically by TNF-alpha and other pro-inflammatory cytokines produced by metabolically active visceral adipose tissue. As visceral fat expands and its inflammatory output increases, PTP1B activity rises, JAK2 is more aggressively inactivated, and leptin signaling is further blunted.
Hypothalamic inflammation. Chronic exposure to saturated fatty acids and gut-derived lipopolysaccharide — both of which enter the central nervous system — activates TLR4-mediated NF-κB signaling in hypothalamic neurons. This produces local neuroinflammation that directly impairs leptin receptor function in the arcuate nucleus. Hypothalamic inflammation is now recognized as a central feature of diet-induced leptin resistance, and it develops early — before obesity is fully established — in response to the same dietary patterns that drive insulin resistance and gut-liver axis dysfunction.
Impaired leptin transport across the blood-brain barrier. Leptin must cross the blood-brain barrier to reach its hypothalamic targets, and it does so via a saturable receptor-mediated transport system. In the context of chronically elevated circulating leptin, this transport system becomes saturated and its efficiency declines. The result is that even high circulating leptin levels fail to produce proportionally high hypothalamic leptin concentrations — a peripheral-to-central delivery failure that compounds the intracellular signaling impairment described above.
These mechanisms interact and amplify each other. Visceral adiposity drives inflammatory cytokine production, which activates PTP1B and promotes hypothalamic inflammation, which impairs leptin receptor signaling, which prevents the satiety signal from arriving, which fails to suppress appetite and energy conservation, which maintains the positive energy balance that expands visceral fat further. The loop is self-reinforcing, and it operates independently of whether the person is consciously overeating or not.
Why Leptin Resistance and Insulin Resistance Arrive Together
In clinical practice, leptin resistance and insulin resistance almost never present independently. They arrive as a pair — and understanding the mechanistic reason for this co-occurrence clarifies why addressing one without the other produces incomplete results.
The connection between the two operates in both directions.
Insulin directly modulates leptin sensitivity in the hypothalamus. Insulin receptors are expressed on hypothalamic POMC neurons, and insulin signaling in the hypothalamus normally potentiates the leptin response — the two signals work synergistically to produce central satiety. When hypothalamic insulin resistance develops — driven by the same inflammatory mechanisms and lipid intermediates that impair peripheral insulin signaling — this potentiation is lost. The hypothalamus becomes simultaneously resistant to both insulin and leptin, and the central satiety system loses two of its primary inputs at once.
At the same time, hyperinsulinemia drives visceral fat expansion through the mechanisms described in the post on adipose tissue insulin resistance and FFA spillover. Expanding visceral fat secretes more leptin and more pro-inflammatory cytokines. More leptin drives SOCS3 upregulation. More cytokines drive PTP1B activation and hypothalamic inflammation. Leptin resistance deepens.
The upstream cause, in the clinical picture I see consistently, is insulin resistance — specifically in the liver. Hepatic insulin resistance drives compensatory hyperinsulinemia. Hyperinsulinemia drives visceral fat deposition. Visceral fat drives leptin hypersecretion and the inflammatory environment that impairs leptin receptor signaling. Leptin resistance at the hypothalamic level then removes the central brake on appetite and energy conservation, which maintains the positive energy balance that sustains all the upstream mechanisms.
This is why the intervention that addresses insulin resistance first — reducing the insulin load through dietary quality, carbohydrate reduction, elimination of ultra-processed foods and HFCS, and structured fasting — is also the intervention that restores leptin sensitivity. It does not target leptin directly. It removes the hyperinsulinemia and the visceral inflammatory environment that are preventing leptin from being heard.
What Leptin Resistance Does to the Body
The clinical consequences of leptin resistance extend significantly beyond persistent hunger and weight gain. Because leptin is a pleiotropic hormone — acting on multiple tissues beyond the hypothalamus — its resistance produces a wide-ranging metabolic and physiological disruption that standard medicine does not frame as a unified syndrome.
Appetite and food intake dysregulation. The most immediately visible consequence is the failure of satiety regulation. Without effective leptin signaling, AgRP neurons are tonically active, driving persistent appetite that is not proportional to energy stores. Patients experience hunger that does not resolve normally after meals, cravings that are driven by neurochemistry rather than genuine energy need, and a subjective experience of never quite feeling satisfied — a pattern that has nothing to do with portion size or dietary willpower and everything to do with the hormonal environment in which eating is occurring.
Reduced energy expenditure. The hypothalamus, in the absence of adequate leptin signaling, reduces energy expenditure through multiple mechanisms — decreasing thermogenesis, reducing spontaneous physical activity, and lowering basal metabolic rate. This metabolic adaptation is the biological basis of the weight loss plateau that most patients on caloric restriction eventually encounter. The body is not failing to respond to the intervention. It is responding exactly as it is designed to respond when the brain believes it is in a state of energy scarcity — by reducing energy output to match the reduced input.
Impaired fatty acid oxidation. Leptin normally promotes fat oxidation in peripheral tissues — particularly in skeletal muscle — by activating AMPK and suppressing malonyl-CoA, which would otherwise inhibit CPT-1 and block mitochondrial fatty acid entry. In leptin resistance, this pro-oxidative signal is absent. Fatty acid oxidation in muscle is reduced, intramyocellular lipid accumulates, and insulin resistance in skeletal muscle is compounded. This creates a direct metabolic link between leptin resistance and the impaired muscle glucose disposal that characterizes peripheral insulin resistance.
Immune and inflammatory dysregulation. Leptin is also a significant immune modulator — it promotes pro-inflammatory Th1 responses and suppresses anti-inflammatory regulatory T cell activity. In leptin resistance, this immune balance shifts toward a chronic pro-inflammatory state that compounds the metabolic inflammation already driven by visceral adiposity and gut-liver axis disruption. The systemic inflammation that characterizes metabolic syndrome is not only a cause of leptin resistance — it is also one of its consequences.
Reproductive and hormonal effects. The hypothalamus integrates leptin signaling into the regulation of the hypothalamic-pituitary-gonadal axis. Impaired leptin signaling contributes to the hormonal dysregulation seen in PCOS — a condition in which hyperinsulinemia, visceral adiposity, and impaired gonadotropin regulation converge — and to the testosterone suppression observed in men with significant metabolic syndrome. These are not separate hormonal problems. They are downstream consequences of the same upstream signaling failure.
Why This Is Invisible in Standard Clinical Practice
Leptin resistance has no diagnostic code in standard clinical practice. There is no routine leptin measurement on any standard metabolic panel. There is no GP consultation in which leptin resistance is named, explained, or addressed. It does not exist, clinically, in the world most patients inhabit before they arrive at a functional medicine assessment.
This invisibility is not primarily a knowledge problem. Most GPs are aware that leptin exists. The problem is structural — the standard consultation is not designed to address mechanisms that do not have a pharmaceutical treatment target and that require time, explanation, and dietary intervention to correct. When the system rewards the management of downstream numbers with prescriptions, the upstream hormonal signaling environment is not where attention is directed.
The markers that reflect leptin resistance indirectly — the metabolic syndrome cluster, the TG/HDL ratio, the elevated fasting insulin, the borderline liver enzymes, the waist circumference above functional threshold — are all already in the standard panel. They are just not being connected to the mechanism producing them. The diagnostic information is present. The interpretive framework to read it is not.
The Intervention: What Restores Leptin Sensitivity
There is no pharmacological intervention that restores leptin sensitivity in the way that the physiological interventions described below do. Exogenous leptin administration does not work in leptin-resistant individuals — the receptor is unresponsive, and injecting more of a signal that is already being ignored does not restore the receptor’s sensitivity to it. The only interventions that have demonstrated meaningful restoration of central leptin sensitivity are those that address the mechanisms driving the resistance.
Elimination of ultra-processed foods, industrial seed oils, and HFCS. These are the primary dietary drivers of the hypothalamic inflammation and gut barrier disruption that initiate and sustain leptin resistance. Their elimination reduces the LPS translocation that drives TLR4-mediated hypothalamic neuroinflammation, reduces the saturated and trans-fatty acid exposure that directly activates hypothalamic inflammatory pathways, and removes the fructose overload that drives hepatic lipogenesis and visceral fat expansion. This is the first and most consequential dietary intervention — not because of its caloric effect, but because of its direct impact on the inflammatory environment in which leptin signaling operates.
Significant carbohydrate reduction. Reducing carbohydrate intake to approximately 50 grams per day lowers the insulin signal that drives visceral fat deposition and the hyperinsulinemia that impairs hypothalamic insulin-leptin synergy. Lower insulin allows visceral fat to begin mobilizing. Shrinking visceral fat reduces the pro-inflammatory cytokine output that drives PTP1B activation and leptin receptor desensitization. The process is not immediate — visceral fat reduction takes weeks — but the direction is consistent and the lab markers that reflect it, including the TG/HDL ratio and fasting insulin, begin to move within the first 8 to 12 weeks.
Animal-based, leucine-rich protein at adequate intake. Protein at 1.6 grams per kilogram of ideal body weight, prioritizing animal-based sources rich in leucine and other branched-chain amino acids, provides the substrate for muscle protein synthesis while exerting direct effects on appetite regulation through GLP-1 and PYY secretion. Adequate muscle mass is also a direct determinant of the peripheral fatty acid oxidation that leptin resistance impairs — more muscle means greater capacity to oxidize the fat that leptin resistance is otherwise directing toward ectopic storage.
Resistance training. Progressive resistance training restores peripheral insulin sensitivity by increasing GLUT4 expression and muscle glucose disposal capacity. It also activates AMPK in skeletal muscle independently of leptin, partially compensating for the reduced AMPK activation that leptin resistance produces. Increased muscle mass improves body composition in a direction that reduces the visceral fat burden and its inflammatory output, creating a positive feedback toward leptin sensitivity restoration.
Structured and irregular fasting. Intermittent fasting — applied irregularly rather than rigidly — reduces the chronic hyperinsulinemia that drives visceral fat deposition and impairs hypothalamic leptin sensitivity. Fasting also reduces the circulating triglyceride and free fatty acid levels that contribute to hypothalamic lipotoxicity. The mechanism through which fasting lowers insulin and the downstream consequences for metabolic restoration are covered in detail in the post on how fasting lowers insulin.
A Note on Uncertainty
The mechanistic account of leptin resistance presented in this post — SOCS3-mediated receptor desensitization, PTP1B activation by inflammatory cytokines, hypothalamic neuroinflammation, impaired blood-brain barrier transport — is well-established in the research literature. What is less precisely quantified in clinical practice is the degree to which leptin resistance is the primary driver of a given patient’s metabolic picture versus a compounding factor in a predominantly insulin-resistance-driven presentation. In practice, the two are so consistently co-present that the distinction has limited clinical utility — the intervention addresses both simultaneously. What matters clinically is the direction of progress, not the precise attribution of causality between two mechanisms that are mutually reinforcing.
Practical Implications
If you have persistent hunger that does not resolve with what should be a satisfying meal, energy that crashes predictably in the afternoon, weight that resists every conventional intervention, and a metabolic panel that shows elevated triglycerides, a TG/HDL ratio above 2.0, and fasting glucose above the functional optimal — leptin resistance is almost certainly part of your picture. It will not be named by your GP. It will not appear in your diagnosis. But the markers that reflect the metabolic environment in which it develops are already in your panel, waiting to be read correctly.
The intervention does not require a leptin measurement. It requires addressing the upstream insulin load and the inflammatory environment that is preventing the leptin signal from reaching its destination. Eliminate the ultra-processed food environment. Reduce the carbohydrate load. Prioritize animal-based protein. Build muscle. Structure fasting irregularly. These are not generic lifestyle recommendations. They are specific mechanistic interventions targeting the biological pathways through which leptin resistance develops and is sustained.
The fat cells are not failing. They are signaling correctly. The problem is upstream — in the inflammatory environment and the hormonal signals that have made the brain deaf to what the fat cells are trying to say. Restore that environment, and the conversation resumes.
People Also Ask
What is leptin resistance?
Leptin resistance is a state in which the brain — specifically the hypothalamus — no longer responds normally to the leptin signal produced by fat cells. In a healthy metabolic state, rising fat mass triggers rising leptin, which signals the brain to reduce appetite and increase energy expenditure. In leptin resistance, this signal is produced — often at high levels — but the hypothalamic leptin receptor no longer responds to it adequately. The result is persistent hunger, reduced energy expenditure, and fat storage that continues despite elevated leptin levels.
What causes leptin resistance?
Leptin resistance develops through several converging mechanisms: chronic elevation of leptin itself triggers SOCS3-mediated desensitization of the leptin receptor; pro-inflammatory cytokines from visceral adipose tissue activate PTP1B, which deactivates JAK2 and blunts leptin signaling; saturated fatty acids and gut-derived LPS produce hypothalamic neuroinflammation that directly impairs leptin receptor function; and chronically elevated circulating leptin saturates the blood-brain barrier transport system, reducing central leptin delivery. These mechanisms are driven by the same upstream factors that produce insulin resistance — visceral adiposity, chronic inflammation, and gut-liver axis disruption.
What is the connection between leptin resistance and insulin resistance?
Leptin resistance and insulin resistance almost always co-present. Insulin signaling in the hypothalamus normally potentiates the leptin response — when hypothalamic insulin resistance develops, leptin sensitivity is simultaneously impaired. At the same time, hyperinsulinemia drives visceral fat expansion, which drives leptin hypersecretion and the pro-inflammatory cytokine output that desensitizes the leptin receptor. The upstream driver is insulin resistance — particularly in the liver — and the intervention that restores insulin sensitivity is the same intervention that restores leptin sensitivity.
How do I know if I have leptin resistance?
Leptin resistance is almost never directly tested in standard clinical practice. The clinical picture that suggests it is operating includes: persistent hunger disproportionate to food intake, energy crashes after meals, weight resistance despite dietary effort, and the metabolic syndrome marker cluster — elevated TG/HDL ratio, elevated fasting insulin when measured, borderline liver enzymes, and central adiposity. No single marker confirms it, but the combination of symptoms and markers read together in the context of the full metabolic picture provides a reliable clinical inference.
Can leptin resistance be reversed?
Yes — through interventions that address the mechanisms driving it rather than the leptin level itself. Exogenous leptin administration does not restore leptin sensitivity because the receptor is unresponsive. What restores sensitivity is reducing the upstream hyperinsulinemia and visceral inflammatory environment that are driving receptor desensitization: elimination of ultra-processed foods, industrial seed oils, and HFCS; significant carbohydrate reduction; adequate animal-based protein; progressive resistance training; and structured fasting. The timeline for meaningful improvement in satiety signaling, energy stability, and body composition is typically 8 to 12 weeks of consistent implementation.
Why does my hunger not go away even when I eat enough?
In leptin resistance, the hypothalamus does not receive an adequate signal that sufficient energy is stored, regardless of what or how much is eaten. The leptin produced by fat cells is not reaching its target receptors in the brain at sufficient intensity to suppress AgRP neuron activity — the neurons that drive appetite — or to activate POMC neurons that produce the satiety signal. The hunger is neurobiological, not caloric. It is driven by a signaling failure, not by an actual energy deficit.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Myers MG Jr, Leibel RL, Seeley RJ, Schwartz MW. Obesity and leptin resistance: distinguishing cause from effect. Trends in Endocrinology and Metabolism. 2010;21(11):643–651. 🔗 https://pubmed.ncbi.nlm.nih.gov/20846876/
2. Münzberg H, Myers MG Jr. Molecular and anatomical determinants of central leptin resistance. Nature Neuroscience. 2005;8(5):566–570. 🔗 https://pubmed.ncbi.nlm.nih.gov/15856064/
3. Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006;444(7121):860–867. 🔗 https://pubmed.ncbi.nlm.nih.gov/17167474/
4. Thaler JP, Yi CX, Schur EA, et al. Obesity is associated with hypothalamic injury in rodents and humans. Journal of Clinical Investigation. 2012;122(1):153–162. 🔗 https://pubmed.ncbi.nlm.nih.gov/22201683/
5. Frühbeck G, Catalán V, Rodríguez A, et al. Adiponectin-leptin ratio is a functional biomarker of adipose tissue inflammation. Nutrients. 2019;11(2):454. 🔗 https://pubmed.ncbi.nlm.nih.gov/30791665/
6. Schwartz MW, Woods SC, Porte D Jr, Seeley RJ, Baskin DG. Central nervous system control of food intake. Nature. 2000;404(6778):661–671. 🔗 https://pubmed.ncbi.nlm.nih.gov/10766253/
7. Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiological Reviews. 2018;98(4):2133–2223. 🔗 https://pubmed.ncbi.nlm.nih.gov/30067154/
8. Enriori PJ, Evans AE, Sinnayah P, Cowley MA. Leptin resistance and obesity. Obesity. 2006;14(S5):254S–258S. 🔗 https://pubmed.ncbi.nlm.nih.gov/17021368/
9. Cani PD, et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56(7):1761–1772. 🔗 https://pubmed.ncbi.nlm.nih.gov/17456850/
10. Farooqi IS, O’Rahilly S. Leptin: a pivotal regulator of human energy homeostasis. American Journal of Clinical Nutrition. 2009;89(3):980S–984S. 🔗 https://pubmed.ncbi.nlm.nih.gov/19176740/
