The most persistent misconception in the entire conversation about fatty liver and metabolic dysfunction is also the most damaging one: that the fat accumulating inside the liver arrived there from the fat you ate. Remove dietary fat, eat less butter, switch to oat milk, avoid eggs and meat — and the liver will clear. This is what millions of patients are told. It is what most dietary guidelines still imply. And it is, in the context of metabolic fatty liver, largely wrong.
The fat that accumulates in a metabolically dysfunctional liver is not primarily dietary in origin. It is manufactured there. The liver itself is producing fat — from carbohydrate substrate, driven by chronically elevated insulin, through a biochemical process called de novo lipogenesis. The word means, precisely, new fat synthesis. The liver is not storing what you ate. It is building fat from what your insulin levels are signaling it to build, using the carbohydrate and fructose substrate that your diet is continuously supplying.
Understanding de novo lipogenesis — what triggers it, how it proceeds at the molecular level, why it accelerates in the context of insulin resistance, and why fructose is a disproportionately potent substrate — changes the entire clinical conversation about fatty liver. It explains why reducing dietary fat without reducing carbohydrate load and insulin produces minimal hepatic improvement. It explains why fructose in particular is metabolically destructive in a way that most nutritional messaging has never communicated. And it explains why the standard dietary advice given to patients with fatty liver — avoid animal fat, use plant-based alternatives, reduce saturated fat — is not only unhelpful but actively misdirects the patient away from the intervention that would actually work.
What you will learn
What de novo lipogenesis is and why it exists as a physiological process | The molecular machinery through which insulin activates hepatic fat synthesis | Why fructose is a uniquely potent DNL substrate and how it bypasses regulatory control | How the insulin-resistant liver creates a paradox of selective sensitivity that drives simultaneous glucose dysregulation and fat overproduction | What the lab markers look like when DNL is the dominant mechanism | Why standard dietary advice gets this entirely backwards — and what the correct intervention actually addresses
Clinical Perspective: What I See in Practice
The patient who arrives with de novo lipogenesis as the dominant driver of their fatty liver has a recognizable clinical picture. Liver enzymes above the functional concern threshold — ALT above 35 U/L, GGT above 20–30 U/L, sometimes significantly higher. An hsCRP that reflects low-grade systemic inflammation. A TG/HDL ratio above 2.0, frequently well above 2.0 — often in the range of 3.0 to 4.0 — reflecting the VLDL overproduction that results directly from excess hepatic triglyceride synthesis.
And behind all of it, a dietary pattern built around high carbohydrate intake, refined foods, and HFCS-containing products from the ultra-processed food environment that has become the default nutritional landscape in both Europe and the United States.
What is consistent across these patients is not that they have been eating large amounts of animal fat. What is consistent is that animal fat has been blamed for their condition — and they believe it. When I ask what their previous practitioner told them about their elevated liver enzymes, the answer is almost always the same: avoid butter, avoid dairy, avoid meat, avoid eggs. Drink oat milk. Use industrial seed oils. The reasoning offered is the one that has been repeated so many times it has become unquestioned: fat makes fat. The liver is full of fat because you ate too much fat.
I shake my head when I hear this, because the underlying mechanism is so precisely the opposite. The very simple fact that fructose is almost entirely metabolized by the liver — and that when hepatic glycogen is already full from continuous carbohydrate loading, the liver has no choice but to convert that incoming fructose to fat faster than it can export it — is not part of the conversation these patients have ever had with their GPs.
The insulin-resistant liver, which cannot suppress its own glucose production in response to insulin’s signal, continues manufacturing fat from carbohydrate substrate regardless of dietary fat intake. This is the mechanism. And it is the mechanism that the recommendation to switch from butter to oat milk addresses not at all.
When I explain this — in plain language, without jargon, connecting the mechanism directly to their specific lab values — the response is consistently one of stunned recognition. They have never heard that fructose goes almost exclusively to the liver. They have never heard that when glycogen storage is full, the liver turns carbohydrate overflow into fat. They have never heard that insulin — not dietary fat — is the signal driving that fat production. The reframe is genuine and significant.
And then something interesting happens: even after they understand it intellectually, even after they trust me and change their eating habits and begin eating animal fat and protein without fear — a dietician appears on Instagram saying avoid eggs, and the fear reasserts itself. The years of conditioning do not dissolve in one consultation. What changes the behavior durably is not the explanation alone — it is seeing the lab markers move. When their triglycerides fall from 280 to 110, when their ALT drops from 78 to 32, and they understand why those numbers moved and which intervention caused it, the intellectual understanding becomes embodied conviction.
The fructose picture in Europe deserves specific attention. In the United States, HFCS consumption has been significantly higher for longer, and the metabolic consequences are correspondingly more severe and more established. In Germany and Western Europe, HFCS consumption has historically been lower — but it is rising.
It is rising not because patients are choosing it deliberately, but because it is present in almost every ultra-processed product on supermarket shelves, and because the nutritional messaging that dominates mainstream German media is simultaneously blaming animal products for metabolic disease and creating the cultural and moral pressure to replace them with exactly the high-carbohydrate, HFCS-laden alternatives that drive de novo lipogenesis.
The cultural situation in Germany around meat is extraordinary and deserves to be named directly. On mainstream television, in cookbooks, in the German language textbooks used to teach immigrants, meat is being reframed as a historical habit of poverty and a current threat to the climate — without a single scientific reference. When I was studying type 2 diabetes at a German university, the textbooks presented dietary recommendations without citations.
In this environment, patients arrive having internalized a dietary framework that directs them away from the foods that would protect their liver and toward the foods that are silently driving the de novo lipogenesis producing the fatty liver they came in to address. The diagnostic and dietary system is not just failing to fix the problem. It is, in specific and traceable ways, creating the conditions for the problem to worsen.
Once the protocol is in place — ultra-processed food eliminated, refined carbohydrates significantly reduced, HFCS-containing products removed, animal-based protein and fat prioritized — the liver markers begin to move. For patients who apply the protocol with genuine consistency, meaningful improvement in triglycerides and ALT is visible within 8 weeks. For others — particularly those who have been sitting with this pattern for years, or whose compliance is partial — it can take up to 6 months. The timeline is not primarily about the biology. The biology responds predictably and consistently. The timeline is about the patient’s ability and willingness to sustain the behavioral change in a food environment that is actively working against it.
What De Novo Lipogenesis Is and Why It Exists
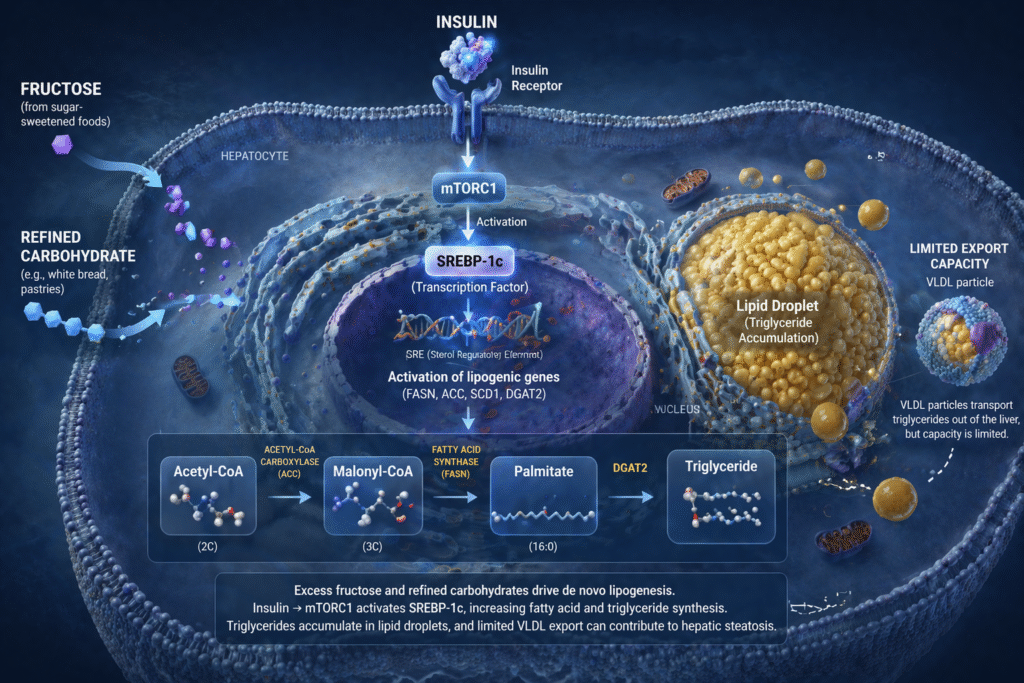
De novo lipogenesis is not a pathological process. It is a normal and essential feature of hepatic metabolism — the biochemical mechanism through which the liver converts excess carbohydrate substrate into fatty acids when carbohydrate intake exceeds the combined capacity of glycogen storage and immediate energy needs. In evolutionary terms, it provided a mechanism for storing seasonal carbohydrate abundance as fat during periods when carbohydrate was intermittent and fat was the primary fuel between feeding episodes.
The process begins with the conversion of excess acetyl-CoA — derived from carbohydrate and other substrates — into malonyl-CoA by acetyl-CoA carboxylase (ACC). Malonyl-CoA is then elongated by fatty acid synthase (FAS) in a series of condensation, reduction, and dehydration reactions that add two-carbon units with each cycle, building palmitate (a 16-carbon saturated fatty acid) as the primary product. Palmitate is then modified — elongated, desaturated, or directly incorporated into triglycerides — for storage inside hepatocytes or packaging into VLDL particles for export into the circulation.
In a metabolically healthy individual with normal insulin sensitivity, De novo lipogenesis is a minor contributor to hepatic fat — accounting for less than 5% of hepatic triglyceride in the fasting state. It activates transiently in the postprandial period when carbohydrate intake genuinely exceeds storage and oxidation capacity, and it is suppressed during fasting and exercise when fat oxidation is the dominant hepatic fuel pathway. The regulatory system is designed to keep DNL tightly controlled.
What disrupts this tight control — and converts De novo lipogenesis from a minor physiological process into a major pathological driver — is chronic hyperinsulinemia in the context of sustained carbohydrate loading. This is the metabolic context that most people in Western food environments now inhabit continuously.
How Insulin Activates De Novo Lipogenesis
Insulin is the primary hormonal activator of hepatic de novo lipogenesis. Its downstream effects on hepatic fat synthesis are mediated through two transcription factors that work in concert: SREBP-1c and ChREBP.
SREBP-1c (Sterol Regulatory Element-Binding Protein 1c) is the master transcriptional regulator of lipogenic gene expression. Insulin activates SREBP-1c through the PI3K–Akt–mTORC1 signaling pathway — the same downstream signaling cascade that mediates insulin’s effects on glucose metabolism. For a comprehensive understanding of how insulin resistance drives fatty liver, refer to our detailed guide.
SREBP-1c, once activated, translocates to the nucleus and drives transcription of the full lipogenic enzyme program: fatty acid synthase (FAS), acetyl-CoA carboxylase (ACC), stearoyl-CoA desaturase (SCD-1), and ATP-citrate lyase (ACLY). The entire enzymatic machinery of fat synthesis is upregulated simultaneously by a single insulin-driven transcriptional event.
ChREBP (Carbohydrate Response Element-Binding Protein) is activated by carbohydrate metabolites directly — specifically by xylulose-5-phosphate, a pentose phosphate pathway intermediate that rises when glucose flux through the liver is high. ChREBP induces many of the same lipogenic genes as SREBP-1c and additionally activates pyruvate kinase, which accelerates the conversion of carbohydrate to acetyl-CoA and thus increases the substrate supply for De novo lipogenesis. ChREBP-mediated De novo lipogenesis activation is, in this sense, carbohydrate-autonomous — it operates even in the absence of insulin signaling, driven purely by the flux of carbohydrate substrate through the hepatocyte.
The combination of SREBP-1c and ChREBP activation in the insulin-resistant, carbohydrate-loaded liver creates a state of constitutive, self-reinforcing DNL. Insulin drives the transcription machinery. Carbohydrate flux drives the substrate supply and additional enzymatic activation. Both signals are chronic and uninterrupted in the metabolic environment produced by continuous eating of refined carbohydrates and ultra-processed foods.
The further consequence of active De novo lipogenesis — specifically the accumulation of malonyl-CoA as an intermediate — is that it inhibits CPT-1 (carnitine palmitoyltransferase 1), the enzyme that transports fatty acids into mitochondria for beta-oxidation. When De novo lipogenesis is running at high rates, malonyl-CoA simultaneously blocks the only exit route for fatty acid clearance. The liver is manufacturing fat at an elevated rate and clearing it at a reduced rate simultaneously. Hepatic triglyceride accumulation under these conditions is not a matter of probability. It is a biochemical inevitability.
The Selective Insulin Resistance Paradox
One of the most clinically important and least discussed features of hepatic insulin resistance is that it is selective — the liver does not become uniformly resistant to all of insulin’s actions simultaneously. It becomes resistant to insulin’s glucose-suppressing effects while remaining fully sensitive to insulin’s fat-producing effects through SREBP-1c activation.
This selective pattern has a specific molecular basis. The insulin signaling branch that suppresses hepatic glucose production (gluconeogenesis and glycogenolysis) operates through IRS-1/IRS-2 → PI3K → Akt → FoxO1 phosphorylation. This branch is impaired early in hepatic insulin resistance, primarily through the DAG–PKCε mechanism that diacylglycerol accumulation activates. IRS-1 serine phosphorylation reduces downstream Akt activation, and FoxO1 is no longer adequately suppressed — allowing it to remain transcriptionally active and drive continued gluconeogenesis regardless of insulin levels.
The SREBP-1c branch, by contrast, receives its insulin signal through a different downstream pathway — specifically through mTORC1 activation — that is preserved significantly longer in the progression of hepatic insulin resistance. The result is that chronically elevated insulin, secreted by the pancreas in compensatory response to the worsening insulin resistance, continues to drive SREBP-1c-mediated lipogenesis through the intact mTORC1 pathway even as the glucose-suppressing branch becomes increasingly unresponsive.
In clinical terms this manifests as the patient whose fasting glucose is rising and whose triglycerides are simultaneously elevated — two findings that a standard panel reports as separate issues but that are both outputs of the same selective insulin resistance pattern. The liver cannot stop producing glucose. And it is simultaneously manufacturing fat at an elevated rate. Both consequences are driven by the same hyperinsulinemia that the worsening insulin resistance is generating. Managing fasting glucose with medication while leaving the insulin load unaddressed does not touch the SREBP-1c activation driving the triglyceride elevation. Both markers will continue on their trajectories until the upstream insulin signal is corrected.
Why Fructose Is a Disproportionately Potent DNL Substrate
Of all the dietary inputs that drive hepatic de novo lipogenesis, fructose occupies a specific and disproportionate position — not because it is categorically toxic in all contexts, but because of how it is metabolized and what that metabolism does in the context of an insulin-resistant, glycogen-replete liver.
Fructose is absorbed from the intestine via GLUT5 transporters and enters the hepatocyte without requiring insulin. Inside the hepatocyte, it is phosphorylated by fructokinase (KHK) to fructose-1-phosphate — a reaction that has no feedback inhibition. Unlike glucose phosphorylation by glucokinase, which is regulated by glucose-6-phosphate accumulation and insulin status, fructokinase-mediated fructose phosphorylation proceeds at a rate determined entirely by substrate availability. As long as fructose is arriving, the reaction runs.
Fructose-1-phosphate is then cleaved by aldolase B into dihydroxyacetone phosphate (DHAP) and glyceraldehyde, both of which enter the lower portion of glycolysis and provide acetyl-CoA for lipogenic pathways. Because this substrate delivery bypasses phosphofructokinase — the primary regulatory checkpoint of glycolysis — fructose effectively circumvents the metabolic brake that limits glucose-driven De novo lipogenesis under normal circumstances. The result is unrestricted substrate delivery to the lipogenic machinery regardless of the cell’s energy status.
In a liver where glycogen storage is already full — as it routinely is in someone eating a high-carbohydrate pattern throughout the day — there is nowhere to direct incoming fructose except toward lipogenesis. The glycogen stores are saturated. Energy expenditure cannot absorb the flux. The only remaining route is fat synthesis. Fructose drives triglyceride accumulation faster than the liver’s VLDL export machinery can clear it, producing a net positive fat balance inside hepatocytes that accumulates with each successive exposure.
The clinical relevance of this is direct and practical. High-fructose corn syrup is present in virtually every category of ultra-processed food and soft drink in the modern food supply — in the United States at historically high levels, and in Europe at levels that are rising rapidly as ultra-processed foods displace traditional dietary patterns.
The patient who is drinking sweetened beverages, eating commercially processed bread, packaged snacks, and convenience foods — and who has simultaneously been told by their GP to avoid animal fat — is receiving a dietary pattern that maximally activates hepatic fructose metabolism, continuously fills glycogen stores, maintains chronic hyperinsulinemia, and drives de novo lipogenesis throughout the day, while simultaneously depriving the liver of the animal-based nutritional inputs that support its metabolic machinery.
This is not a complicated story. It is, however, one that requires explaining — because the mainstream nutritional framework that most patients inhabit does not contain it.
What the Lab Panel Looks Like
De novo lipogenesis leaves a specific and consistent fingerprint in standard lab panels. The challenge is not that the information is unavailable — it is that the combination is almost never interpreted in terms of the hepatic fat manufacturing process producing it.
Elevated triglycerides are the most direct consequence. When the liver synthesizes triglycerides through DNL faster than it can package and export them as VLDL, circulating triglycerides rise. A fasting triglyceride above 150 mg/dL is above the conventional threshold; above 100 mg/dL is above the functional medicine optimal. In patients where DNL is the dominant mechanism, triglycerides are often significantly elevated — in the range of 200 to 300 mg/dL or higher — and respond more rapidly to carbohydrate reduction than to any other single intervention.
Suppressed HDL accompanies the triglyceride elevation through the cholesteryl ester transfer protein mechanism. Elevated VLDL triglycerides facilitate lipid exchange with HDL particles, depleting HDL cholesterol. The TG/HDL ratio above 2.0 in mg/dL units — and especially above 3.0 — is the most accessible proxy for the hepatic insulin resistance and hyperinsulinemia driving DNL. It is present in every standard lipid panel. It is almost never interpreted in this context.
Elevated ALT and GGT reflect the hepatocyte stress and oxidative burden that accompany intrahepatic fat accumulation. ALT above the functional concern threshold of 35 U/L indicates hepatic metabolic stress. GGT above 20–30 U/L reflects the oxidative load and adaptive response of the glutathione system to the increased lipid flux through the hepatocyte. The gap between functional and conventional thresholds — ALT functional concern at 35 U/L versus conventional upper limit at 56 U/L, GGT functional concern at 20–30 U/L versus conventional upper limit above 55 U/L — is the diagnostic window where DNL is operating and the standard panel is reporting normal.
Elevated hsCRP reflects the systemic low-grade inflammation driven by the visceral adiposity and gut-liver axis disruption that co-occur with chronic DNL activation. An hsCRP above 1.0 mg/L in the context of the lipid pattern above confirms that the inflammatory environment is compounding the hepatic metabolic burden.
Fasting insulin above the functional optimal of 5 µIU/mL — and especially above 10 µIU/mL — is the direct measure of the compensatory hyperinsulinemia that is driving SREBP-1c activation and maintaining the DNL program. This is the upstream signal. Everything else in the panel is downstream of it.
When these markers appear together — elevated TG/HDL ratio, elevated ALT and GGT, elevated hsCRP, and elevated fasting insulin in a patient eating a high-carbohydrate ultra-processed diet — the picture is coherent and specific. The liver is manufacturing fat. The mechanism is operating. And the intervention that addresses it is not dietary fat restriction.
Why Standard Dietary Advice Gets This Backwards
The standard dietary advice for fatty liver — reduce saturated fat, avoid animal products, use plant-based alternatives — is built on a causal model that has the mechanism precisely inverted. It assumes that the fat in the liver arrived from dietary fat. The tracer data — most definitively the Donnelly et al. 2005 study — demonstrates that dietary fat accounts for approximately 15% of hepatic triglyceride in established NAFLD, while adipose-derived free fatty acids account for approximately 60% and de novo lipogenesis accounts for approximately 25%. The dominant driver is not what was eaten. It is what the insulin-resistant adipose tissue is releasing and what the insulin-stimulated liver is manufacturing.
Recommending oat milk over dairy removes one of the most bioavailable sources of protein, vitamin B12, and calcium from the patient’s diet while adding a carbohydrate load that — in the context of chronic hyperinsulinemia — directly activates ChREBP and SREBP-1c-mediated lipogenesis. Recommending industrial seed oils over animal fat replaces a stable, non-inflammatory fat source with a highly oxidizable omega-6-rich fat that contributes to the lipid peroxidation and oxidative stress that GGT is measuring. Recommending reduced meat intake removes the leucine-rich, bioavailable protein that supports muscle mass — the primary peripheral sink for glucose that would otherwise drive hepatic carbohydrate overload.
These recommendations do not improve the hepatic fat manufacturing mechanism. They worsen its substrate environment while simultaneously depriving the patient of the nutritional inputs that would support recovery.
The Intervention: What Actually Addresses De Novo Lipogenesis
The intervention that addresses de novo lipogenesis is mechanistically specific. It does not target fat. It targets insulin.
Elimination of ultra-processed foods, HFCS, and refined carbohydrates removes the primary substrate for DNL — the fructose that bypasses glycolytic regulation and the refined carbohydrate that saturates glycogen stores and drives ChREBP activation. This is the first and most consequential intervention. In patients who implement it genuinely, the triglyceride response is rapid and often dramatic — reflecting the direct reduction in hepatic lipogenic substrate supply.
Carbohydrate reduction to approximately 50 grams per day lowers the sustained carbohydrate flux that maintains ChREBP activity and the chronic insulin elevation that maintains SREBP-1c activation. Lower insulin reduces mTORC1-mediated SREBP-1c induction. Lower carbohydrate flux reduces ChREBP-mediated lipogenic gene expression. Both branches of the DNL activation machinery are addressed simultaneously.
Animal-based protein at 1.6 grams per kilogram of ideal body weight provides the substrate for lean mass maintenance while exerting a significantly lower insulinogenic effect than carbohydrate. Adequate muscle mass improves peripheral glucose disposal, reducing the fraction of carbohydrate that must be processed by the liver and reducing the compensatory insulin secretion that drives SREBP-1c activation. Leucine-rich animal protein also independently activates mTOR in muscle for protein synthesis purposes — directing the mTOR pathway toward anabolism in peripheral tissue rather than lipogenesis in the liver.
Structured and irregular fasting creates the conditions under which malonyl-CoA falls, CPT-1 inhibition is released, and the liver shifts from lipogenesis to beta-oxidation. During fasting, insulin falls, SREBP-1c activation is suppressed, and the liver begins clearing the intrahepatic fat it has accumulated. The metabolic rest that fasting provides is not primarily caloric. It is hormonal — the reduction in insulin is what switches the hepatic metabolic program from fat manufacturing to fat burning.
A Note on Uncertainty
The mechanistic account of de novo lipogenesis presented in this post — SREBP-1c and ChREBP activation, malonyl-CoA-mediated CPT-1 inhibition, selective insulin resistance, fructose’s bypass of phosphofructokinase — is among the most extensively characterized in metabolic biochemistry. The tracer data quantifying the relative contributions of DNL, adipose FFA spillover, and dietary fat to hepatic triglyceride is from carefully controlled human studies.
What varies between individuals is the relative contribution of DNL versus adipose FFA spillover as the dominant mechanism in any given patient — and therefore the relative emphasis the intervention should place on carbohydrate reduction versus insulin sensitivity restoration in adipose tissue. This variability is particularly evident in conditions such as lean fatty liver disease, where the presentation differs from typical NAFLD.In practice, the protocol addresses both simultaneously, and both respond to the same upstream intervention.
Practical Implications
If your triglycerides are elevated, your TG/HDL ratio is above 2.0, your liver enzymes are above the functional concern threshold, and you have been told to reduce animal fat and increase plant-based foods — the mechanism described in this post is almost certainly operating in your case, and the advice you have received is addressing the wrong variable.
The practical starting point is measuring fasting insulin if it has not been done, calculating your TG/HDL ratio from your existing lipid panel, and applying the functional medicine thresholds rather than the conventional reference ranges to your ALT and GGT. Then direct the intervention at the insulin load — not the dietary fat.
The fat in your liver was not put there by the fat you ate. It was manufactured there, by your liver, from the carbohydrate substrate your insulin was signaling it to convert. That distinction is not academic. It is the difference between an intervention that works and one that doesn’t.
People Also Ask
What is de novo lipogenesis?
De novo lipogenesis is the biochemical process by which the liver converts excess carbohydrate substrate — including glucose and fructose — into fatty acids and ultimately into triglycerides. The word means literally new fat synthesis. In the context of chronic hyperinsulinemia and continuous carbohydrate loading, DNL becomes a major driver of hepatic fat accumulation, producing intrahepatic triglyceride independently of dietary fat intake.
Does dietary fat cause fatty liver?
In the majority of metabolic fatty liver cases, dietary fat is a minor contributor. Tracer studies in humans with established NAFLD demonstrate that approximately 15% of hepatic triglyceride derives from dietary fat, while approximately 60% derives from adipose tissue free fatty acid spillover and approximately 25% from hepatic de novo lipogenesis. The dominant drivers are insulin resistance and carbohydrate-driven fat synthesis — not dietary fat intake.
Why is fructose particularly bad for the liver?
Fructose is metabolized almost exclusively in the liver and is phosphorylated by fructokinase without the feedback inhibition that limits glucose-driven fat production. This allows fructose to deliver substrate to the lipogenic machinery in an unregulated manner, bypassing the primary glycolytic checkpoint. In a liver where glycogen storage is already saturated from continuous carbohydrate intake, fructose has nowhere to go except into fat synthesis. HFCS in ultra-processed foods and soft drinks delivers this substrate continuously throughout the day.
What does the lab panel look like when de novo lipogenesis is active?
The characteristic combination includes: elevated fasting triglycerides, suppressed HDL, TG/HDL ratio above 2.0 in mg/dL, ALT above the functional concern threshold of 35 U/L, GGT above 20–30 U/L, elevated hsCRP reflecting systemic inflammation, and elevated fasting insulin when measured. This combination is present in every standard panel — it is almost never interpreted in terms of the hepatic fat manufacturing process producing it.
What is selective insulin resistance in the liver?
Selective hepatic insulin resistance describes the pattern in which the liver becomes resistant to insulin’s glucose-suppressing effects while remaining sensitive to insulin’s fat-producing effects. The glucose-suppressing pathway — through IRS-1/IRS-2, PI3K, Akt, and FoxO1 — is impaired by the DAG-PKCε mechanism, allowing gluconeogenesis to continue despite elevated insulin. The lipogenic pathway — through mTORC1 and SREBP-1c — remains intact, allowing elevated insulin to continue driving fat synthesis. The result is simultaneous fasting glucose elevation and triglyceride elevation from the same hyperinsulinemia.
How do you reverse hepatic de novo lipogenesis?
The intervention addresses the upstream insulin signal rather than the downstream fat product. Eliminating ultra-processed foods, HFCS, and refined carbohydrates removes the primary DNL substrate. Reducing carbohydrate intake to approximately 50 grams per day reduces the chronic insulin elevation that activates SREBP-1c and the carbohydrate flux that activates ChREBP. Adequate animal-based protein supports lean mass and peripheral glucose disposal, reducing the hepatic carbohydrate burden. Structured fasting lowers insulin, suppresses lipogenesis, and allows the liver to shift into fat-clearing mode. Triglycerides typically begin to respond within 8 weeks of consistent implementation.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Donnelly KL, Smith CI, Schwarzenberg SJ, et al. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. Journal of Clinical Investigation. 2005;115(5):1343–1351. 🔗 https://pubmed.ncbi.nlm.nih.gov/15864352/
2. Stanhope KL, Schwarz JM, Keim NL, et al. Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids and decreases insulin sensitivity in overweight/obese humans. Journal of Clinical Investigation. 2009;119(5):1322–1334. 🔗 https://pubmed.ncbi.nlm.nih.gov/19381015/
3. Lim JS, Mietus-Snyder M, Valente A, Schwarz JM, Lustig RH. The role of fructose in the pathogenesis of NAFLD and the metabolic syndrome. Nature Reviews Gastroenterology and Hepatology. 2010;7(5):251–264. 🔗 https://pubmed.ncbi.nlm.nih.gov/20368739/
4. Brown MS, Goldstein JL. Selective versus total insulin resistance: a pathogenic paradox. Cell Metabolism. 2008;7(2):95–96. 🔗 https://pubmed.ncbi.nlm.nih.gov/18249166/
5. Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiological Reviews. 2018;98(4):2133–2223. 🔗 https://pubmed.ncbi.nlm.nih.gov/30067154/
6. Jornayvaz FR, Shulman GI. Diacylglycerol activation of protein kinase Cε and hepatic insulin resistance. Cell Metabolism. 2012;15(5):574–584. 🔗 https://pubmed.ncbi.nlm.nih.gov/22560219/
7. Luukkonen PK, Zhou Y, Sädevirta S, et al. Hepatic ceramides dissociate steatosis and insulin resistance in patients with non-alcoholic fatty liver disease. Journal of Hepatology. 2016;64(5):1167–1175. 🔗 https://pubmed.ncbi.nlm.nih.gov/26867493/
8. Softic S, Cohen DE, Kahn CR. Role of dietary fructose and hepatic de novo lipogenesis in fatty liver disease. Digestive Diseases and Sciences. 2016;61(5):1282–1293. 🔗 https://pubmed.ncbi.nlm.nih.gov/26856717/
9. Shimomura I, Matsuda M, Hammer RE, Bashmakov Y, Brown MS, Goldstein JL. Decreased IRS-2 and increased SREBP-1c lead to mixed insulin resistance and sensitivity in livers of lipodystrophic and ob/ob mice. Molecular Cell. 2000;6(1):77–86. 🔗 https://pubmed.ncbi.nlm.nih.gov/10949029/
10. Fabbrini E, Sullivan S, Klein S. Obesity and nonalcoholic fatty liver disease: biochemical, metabolic, and clinical implications. Hepatology. 2010;51(2):679–689. 🔗 https://pubmed.ncbi.nlm.nih.gov/20041406/
