Gestational diabetes is one of the most consequential metabolic diagnoses a woman can receive — and one of the most consistently mismanaged. Not because the condition itself is poorly understood biochemically, but because the clinical framework applied to it is the same downstream, symptom-suppression framework that fails metabolic patients everywhere else: measure a number, prescribe a drug to lower it, and treat the resulting pharmaceutical dependency as an acceptable long-term outcome.
The biological reality of insulin resistance during pregnancy is far more nuanced — and in its early stages, far more purposeful — than the standard clinical narrative acknowledges. Pregnancy requires a specific hormonal metabolic shift that makes glucose more available to the fetus. This shift involves a physiological degree of maternal insulin resistance that is not pathological. It is evolutionary.
It is the biological mechanism through which the maternal metabolic system ensures that the developing fetus has a reliable glucose supply even during periods of maternal energy scarcity. What becomes pathological is when a woman enters pregnancy with pre-existing insulin resistance, when the physiological insulin resistance of pregnancy amplifies an already compromised metabolic baseline, and when the clinical response to the resulting glucose elevation is to add pharmaceutical glucose management on top of a dietary pattern that is continuously driving the problem.
The woman who is told to eat berries every two hours and take metformin has not been given a metabolic intervention. She has been given a protocol that manages the downstream glucose number while the upstream insulin resistance producing it — present before conception, amplified by pregnancy, and left entirely unaddressed — continues to deteriorate. And in too many cases, the insulin that was prescribed as a temporary gestational measure becomes a permanent fixture of her metabolic management, not because her condition required it, but because nobody ever addressed the mechanism that produced the condition in the first place.
What you will learn: The evolutionary and physiological basis of insulin resistance during pregnancy — and why some degree of it is normal | The difference between physiological gestational insulin resistance and the pathological escalation that gestational diabetes represents | What the early warning markers are and why they are almost never measured | The clinical management approach that addresses the mechanism rather than suppressing the downstream number | The long-term consequences for mother and child when gestational insulin resistance is not addressed at the root
Clinical Perspective: What I See in Practice
Women who arrive in this practice with gestational diabetes or pregnancy-related insulin resistance come with a consistent combination of confusion and distress. They left their GP’s office — or their obstetric consultation — uninformed about what is actually happening in their metabolism. The condition was named. A medication was prescribed, or a dietary instruction was given. The underlying mechanism was not explained. And now they are frightened, uncertain whether what they are experiencing is dangerous, and looking for someone who will explain it to them rather than manage them through it.
I had a patient just last week — a bright, informed German woman who had been doing her own research on nutrition and understood macronutrients and micronutrients at a level well above the average patient. Her GP’s advice for her gestational insulin resistance: eat berries every two hours. That was the full clinical intervention.
I was genuinely relieved that he had not prescribed metformin or exogenous insulin — both of which I have seen prescribed for gestational glucose elevation in conditions that, with proper dietary and lifestyle intervention, would not have required pharmaceutical management at all. This woman understood the mechanism quickly when I explained it, implemented the protocol willingly and with discipline, and is managing her pregnancy metabolically without medication. She is an exception — not because her condition was exceptional, but because she had the knowledge and motivation to seek a different approach.
The majority of women in this situation do not arrive with that background. They trust their GPs, they follow the instructions given, and those instructions — in the cases I see regularly — consist of a high-carbohydrate, frequent-eating protocol combined with pharmaceutical glucose suppression. Eat seven times a day. Fruits and wholegrain bread at every eating occasion. And then take a drug to lower the blood sugar that the dietary pattern is producing.
The internal contradiction of this approach — driving glucose upward with continuous carbohydrate loading and then suppressing it pharmacologically — is not apparent to the patient because nobody has explained the mechanism. The glucose number is being managed. The insulin resistance producing the glucose elevation continues, undisturbed, throughout the pregnancy and beyond.
The long-term consequence of this management approach is what I find most clinically troubling. I have had multiple women in this practice who began exogenous insulin during pregnancy and were still using it years after delivery. Not because their pancreatic function was insufficient — but because the insulin resistance that gestational management failed to address became permanent metabolic dysfunction, and the insulin that was initiated as a temporary measure became a structural fixture of a care pathway that never revisited whether it was still necessary. The woman who needed insulin at 28 weeks of pregnancy did not necessarily need it at three years postpartum. But no one had the conversation.
The evolutionary framing matters enormously in these consultations because it transforms the patient’s relationship to their condition. When I explain that a degree of insulin resistance during pregnancy is not a failure or a disease — that it is the biological mechanism through which the maternal metabolic system ensures fetal glucose supply even under conditions of maternal energy constraint — the relief in the room is palpable. They are not broken.
Their body is not malfunctioning. The mechanism is physiologically appropriate. What has gone wrong is that the mechanism is amplified beyond the physiological range by a pre-existing metabolic baseline that was already compromised before conception, and that the dietary and lifestyle environment of modern pregnancy — continuous carbohydrate loading, frequent eating, physical inactivity, sleep disruption — is driving it further in the wrong direction. That is a different clinical problem from the one they walked in believing they had. And it has a different — and more complete — solution.
Laleh’s story — which she has shared publicly and with her full consent — is the clearest illustration of what this approach produces. A 45-year-old nurse from England, already managing an autoimmune condition (thrombocytopenia), 16 weeks pregnant. Her British GP told her she needed insulin because her blood sugar was too high. She sought a different path.
With a personalized nutrition protocol addressing the upstream insulin load rather than the downstream glucose number, her blood sugar normalized without medication. She carried her pregnancy without insulin. She describes the experience as remarkable — not because the intervention was exotic, but because it was the first time someone had explained what was actually happening and designed a response to the mechanism rather than the marker.
The Evolutionary Biology of Gestational Insulin Resistance
To understand gestational diabetes as a pathological amplification of a normal process, the normal process must first be understood.
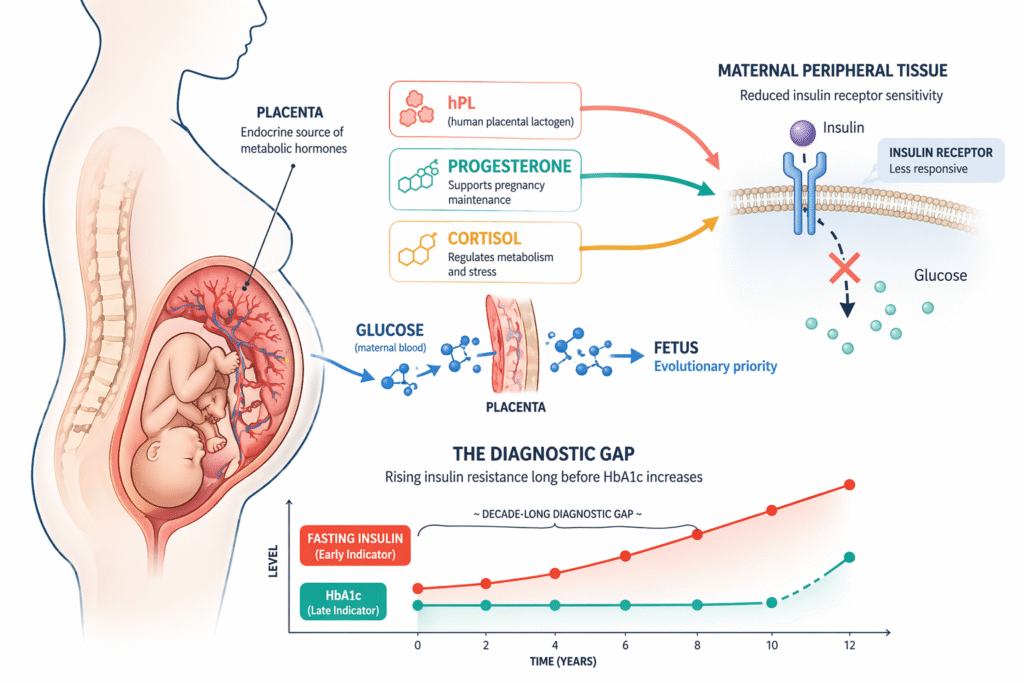
During pregnancy, the placenta produces a specific set of hormones — including human placental lactogen (hPL), progesterone, cortisol, and placental growth hormone — whose combined effect is to progressively increase maternal peripheral insulin resistance across the second and third trimesters. This is not a malfunction. It is a precisely orchestrated hormonal shift that serves a specific evolutionary purpose: by reducing maternal insulin sensitivity in skeletal muscle and adipose tissue, the placental hormones redirect glucose away from maternal peripheral tissue and toward the placenta and fetus, which receive glucose through facilitated diffusion independent of insulin. The fetus is guaranteed a glucose supply regardless of maternal nutritional state.
In evolutionary context, this mechanism was essential. Pregnancy in a food-insecure environment — the norm for most of human history — required a biological guarantee that fetal development would not be compromised by periods of maternal food scarcity. The physiological insulin resistance of pregnancy is that guarantee. It is a maternal sacrifice of insulin sensitivity in the service of fetal glucose priority.
The system works as designed when the mother enters pregnancy with a metabolically healthy baseline — normal fasting insulin, normal hepatic insulin sensitivity, normal adipose tissue insulin sensitivity, normal pancreatic beta cell reserve. In this context, the placental hormones produce a modest, well-compensated degree of insulin resistance that the pancreas handles through a modest increase in insulin secretion, blood glucose remains within a narrow functional range, and the pregnancy proceeds without metabolic complication.
The system breaks down when the mother enters pregnancy with pre-existing insulin resistance — elevated fasting insulin, early hepatic insulin resistance, adipose tissue that is already dysregulated. In this context, the placental hormones amplify an already-compromised metabolic baseline. The pancreas, already secreting excess insulin to compensate for peripheral resistance, must increase its secretory output further. If beta cell reserve is insufficient to meet this demand — or if the hepatic and peripheral resistance is severe enough that even elevated insulin cannot maintain glucose control — fasting and postprandial glucose rise above the diagnostic threshold and gestational diabetes is identified.
What this framing clarifies is that gestational diabetes is not a new disease that begins in pregnancy. It is the unmasking of pre-existing metabolic dysfunction that was operating silently before conception — detectable through fasting insulin, the TG/HDL ratio, and the early insulin resistance markers that standard obstetric screening does not include — and that the metabolic demand of pregnancy has amplified beyond the threshold of compensation.
What Standard Screening Misses
Gestational diabetes is screened in most countries at 24–28 weeks of pregnancy using the oral glucose tolerance test. By 24 weeks, the placental hormonal shift is substantially advanced and the metabolic demand on the pancreas is near its peak. If insulin resistance was present before conception — as it is in the majority of women who develop gestational diabetes — the opportunity to identify and address it before the pregnancy created the conditions for its escalation has already passed.
The markers that would have identified pre-existing insulin resistance before conception, or early in the first trimester when intervention would have been most impactful, are not included in standard obstetric panels. Fasting insulin — the most direct measure of compensatory hyperinsulinemia and the primary upstream driver of the metabolic dysfunction that gestational diabetes represents — is almost never ordered in standard obstetric care in Germany.
The TG/HDL ratio, which provides the lipid fingerprint of hepatic insulin resistance and compensatory hyperinsulinemia, is in every standard lipid panel and is almost never interpreted in this context. GGT above the functional concern threshold signals hepatic metabolic stress before glucose becomes abnormal. A history of PCOS — itself a manifestation of hyperinsulinemia — is one of the strongest predictors of gestational diabetes and should trigger early metabolic assessment.
The combination of prior PCOS, central adiposity, TG/HDL ratio above 2.0, fasting insulin above 10 µIU/mL, and GGT above functional concern thresholds in a woman of reproductive age is a coherent early insulin resistance picture that predicts gestational diabetes risk with considerable reliability. None of these markers are assessed in standard preconception or early antenatal care. The first clinical intervention in this trajectory, in most cases, is the glucose tolerance test at 24 weeks — by which point the window for the most complete and durable intervention has substantially narrowed.
The Downstream Consequences: Mother and Child
The consequences of unaddressed gestational insulin resistance extend significantly beyond the pregnancy itself — for both the mother and the child.
For the mother, gestational diabetes that is managed pharmacologically without addressing the underlying insulin resistance produces a specific and predictable long-term trajectory. The insulin resistance that was present before pregnancy and amplified by it does not resolve after delivery unless the upstream drivers are corrected. Women with gestational diabetes have a 50–70% lifetime risk of developing type 2 diabetes — a risk that reflects not the gestational event itself, but the pre-existing metabolic dysfunction that gestational diabetes identified.
If that dysfunction is not corrected postpartum, the risk is actualized progressively over the subsequent years. The woman who was told her gestational diabetes resolved after delivery because her postpartum glucose test came back normal has not been told that her fasting insulin may still be elevated, her TG/HDL ratio may still be above 2.0, and the metabolic trajectory that gestational diabetes identified is continuing.
For the child, the intrauterine environment of hyperglycemia and hyperinsulinemia during fetal development has specific and measurable consequences. Fetal hyperinsulinemia — the fetal pancreatic response to elevated maternal glucose crossing the placenta — drives excess fetal fat deposition, producing the large-for-gestational-age birth pattern associated with gestational diabetes.
Beyond birth weight, the intrauterine metabolic environment programs the child’s metabolic physiology through epigenetic mechanisms — altering gene expression patterns in metabolic tissues that increase the offspring’s lifetime risk of obesity, insulin resistance, and type 2 diabetes. The consequences of unaddressed gestational insulin resistance are not limited to the mother. They are transmitted to the next generation through a biological mechanism that is entirely preventable when the maternal insulin environment is addressed.
What the Intervention Actually Addresses
The clinical goal in gestational insulin resistance is not to lower glucose by adding medication on top of a high-carbohydrate dietary pattern. It is to reduce the insulin load that is driving the glucose elevation while maintaining adequate nutrition for fetal development. These are not competing goals. They are achievable simultaneously with the right dietary framework.
Carbohydrate quality and quantity. The continuous carbohydrate loading that characterizes the standard gestational dietary advice — frequent small meals, wholegrain bread, fruit at every eating occasion — maintains the insulin elevation that the placental hormones are already driving upward. A dietary pattern that reduces refined carbohydrates, eliminates HFCS and ultra-processed foods, and moderates total carbohydrate intake while prioritizing animal-based protein and fat significantly reduces the insulin demand placed on an already-stressed pancreas without compromising fetal nutrition. The fetus does not require maternal hyperglycemia. It requires an adequate glucose supply — which a controlled, stable maternal glucose environment provides more reliably than the glucose excursions produced by continuous carbohydrate loading.
Protein adequacy. Animal-based protein at adequate intake supports maternal lean mass, provides the amino acids essential for fetal tissue development, exerts a significantly lower insulinogenic effect than carbohydrate, and supports satiety — reducing the compensatory eating that the blood sugar roller coaster of high-carbohydrate frequent feeding produces. The concern that animal protein is unsafe during pregnancy is not supported by the evidence and represents an extension of the fat-phobia narrative into obstetric care.
Physical activity. Resistance training and walking after meals are the most evidence-supported non-dietary interventions for improving peripheral insulin sensitivity during pregnancy. Resistance training is safe and beneficial throughout uncomplicated pregnancy. Walking after meals reduces postprandial glucose excursions through muscle glucose disposal — the same mechanism that makes exercise a primary metabolic intervention in non-pregnant insulin resistance.
Sleep and circadian alignment. Sleep disruption during pregnancy — common for obvious reasons — worsens insulin resistance independently of diet and activity. Supporting sleep quality, maintaining consistent sleep-wake timing, and managing the circadian disruption that late-night screen exposure and irregular schedules produce is a metabolically meaningful intervention, not a lifestyle recommendation.
GP collaboration is non-negotiable. Pregnancy is the clinical context in which GP and obstetric collaboration is most critical. Every intervention in this context operates under medical supervision. Medication decisions — including decisions about whether to initiate or discontinue insulin or metformin — belong exclusively to the attending physician. The role of the functional nutrition framework is to reduce the insulin load through diet and lifestyle in a way that makes the glucose numbers move without medication when that is clinically achievable — and to work transparently with the supervising physician when it is not.
A Note on Uncertainty
The evolutionary framing of physiological gestational insulin resistance — as a mechanism for redirecting glucose to the fetus under conditions of maternal energy constraint — is a well-supported hypothesis with substantial mechanistic and anthropological evidence. What is less precisely defined is the threshold at which physiological gestational insulin resistance becomes pathological gestational diabetes in any given individual — a threshold that is influenced by pre-existing metabolic baseline, pancreatic beta cell reserve, dietary environment, activity level, and genetic factors.
The diagnostic criteria for gestational diabetes vary between countries and clinical guidelines, and the evidence base for specific glucose thresholds in pregnancy is not as robust as the clinical confidence with which those thresholds are applied would suggest. The conservative clinical approach — identifying and addressing insulin resistance risk before and early in pregnancy, supporting metabolic health throughout, and calibrating medical intervention to individual clinical response — reflects this uncertainty appropriately.
Practical Implications
If you have a history of PCOS, prior gestational diabetes, central adiposity, or a TG/HDL ratio above 2.0 and you are planning a pregnancy or are in early pregnancy — the metabolic picture that predicts gestational diabetes risk can be assessed and addressed before 24 weeks. Request fasting insulin. Calculate your TG/HDL ratio from your existing lipid panel. If fasting insulin is above 10 µIU/mL, the upstream insulin resistance that standard obstetric screening will not identify until it has already escalated is almost certainly present.
If you are currently managing gestational diabetes and your clinical advice consists of frequent carbohydrate eating and pharmaceutical glucose suppression — ask about the dietary framework that reduces the insulin load producing the glucose elevation rather than adding medication on top of the dietary pattern driving it.
Gestational insulin resistance is not a disease that begins in pregnancy. It is a metabolic signal that began years earlier — and that pregnancy has made visible. The question is not how to lower the number. The question is what produced the number, and whether that can be corrected rather than suppressed.
People Also Ask
What causes insulin resistance during pregnancy?
A physiological degree of insulin resistance during pregnancy is caused by placental hormones — including human placental lactogen, progesterone, cortisol, and placental growth hormone — that reduce maternal peripheral insulin sensitivity to redirect glucose toward the fetus. This is a normal evolutionary mechanism. It becomes pathological gestational diabetes when pre-existing maternal insulin resistance amplifies the placental hormonal effect beyond the pancreas’s compensatory capacity.
Is gestational diabetes permanent?
Gestational diabetes as defined by standard diagnostic criteria typically resolves after delivery as placental hormones clear. However, the pre-existing insulin resistance that gestational diabetes identified does not resolve unless it is actively addressed. Women with gestational diabetes have a 50–70% lifetime risk of developing type 2 diabetes — reflecting the persistence of the underlying metabolic dysfunction, not the gestational event itself.
What is the connection between PCOS and gestational diabetes?
PCOS is itself a manifestation of hyperinsulinemia — the same upstream driver of gestational diabetes. Women with PCOS have chronically elevated fasting insulin, impaired ovarian insulin signaling that drives androgen excess, and a pre-existing metabolic baseline that places them at significantly elevated risk for gestational diabetes when the insulin demands of pregnancy are added to an already-compromised metabolic foundation.
What should I eat if I have gestational diabetes?
The dietary approach that addresses the mechanism of gestational insulin resistance focuses on reducing refined carbohydrates, eliminating HFCS and ultra-processed foods, and prioritizing animal-based protein and fat that exert a lower insulinogenic effect than carbohydrate. This contrasts with the standard advice to eat frequent small carbohydrate-containing meals, which maintains the insulin elevation that placental hormones are already driving upward. All dietary changes during pregnancy should be implemented under medical supervision.
Can gestational diabetes be managed without medication?
In many cases, yes — particularly when the dietary pattern is changed to reduce the insulin load driving the glucose elevation rather than adding medication on top of a high-carbohydrate dietary pattern. The documented case of a 45-year-old nurse with gestational diabetes and thrombocytopenia who normalized her blood sugar through dietary intervention alone — without the insulin her GP had prescribed — illustrates what is achievable when the mechanism is addressed rather than suppressed. This requires medical supervision and individual clinical assessment.
What are the long-term consequences of gestational diabetes for the baby?
The intrauterine environment of hyperglycemia and hyperinsulinemia during fetal development drives excess fetal fat deposition through fetal hyperinsulinemia — the fetal pancreatic response to elevated maternal glucose crossing the placenta. Beyond birth weight, the maternal metabolic environment programs fetal metabolic physiology through epigenetic mechanisms, increasing the offspring’s lifetime risk of obesity, insulin resistance, and type 2 diabetes. These consequences are preventable when maternal insulin resistance is identified and addressed early.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Catalano PM, Huston L, Amini SB, Kalhan SC. Longitudinal changes in glucose metabolism during pregnancy in obese women with normal glucose tolerance and gestational diabetes mellitus. American Journal of Obstetrics and Gynecology. 1999;180(4):903–916. 🔗 https://pubmed.ncbi.nlm.nih.gov/10203659/
2. Kim C, Newton KM, Knopp RH. Gestational diabetes and the incidence of type 2 diabetes: a systematic review. Diabetes Care. 2002;25(10):1862–1868. 🔗 https://pubmed.ncbi.nlm.nih.gov/12351492/
3. Bellamy L, Casas JP, Hingorani AD, Williams D. Type 2 diabetes mellitus after gestational diabetes: a systematic review and meta-analysis. The Lancet. 2009;373(9677):1773–1779. 🔗 https://pubmed.ncbi.nlm.nih.gov/19465232/
4. Dabelea D, Hanson RL, Lindsay RS, et al. Intrauterine exposure to diabetes conveys risks for type 2 diabetes and obesity: a study of discordant sibships. Diabetes. 2000;49(12):2208–2211. 🔗 https://pubmed.ncbi.nlm.nih.gov/11118027/
5. Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiological Reviews. 2018;98(4):2133–2223. 🔗 https://pubmed.ncbi.nlm.nih.gov/30067154/
6. Lowe WL Jr, Scholtens DM, Lowe LP, et al. Association of gestational diabetes with maternal disorders of glucose metabolism and childhood adiposity. JAMA. 2018;320(10):1005–1016. 🔗 https://pubmed.ncbi.nlm.nih.gov/30209464/
7. Crowther CA, Hiller JE, Moss JR, et al. Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. New England Journal of Medicine. 2005;352(24):2477–2486. 🔗 https://pubmed.ncbi.nlm.nih.gov/15951574/
8. Tobias DK, Zhang C, van Dam RM, Bowers K, Hu FB. Physical activity before and during pregnancy and risk of gestational diabetes mellitus. Diabetes Care. 2011;34(1):223–229. 🔗 https://pubmed.ncbi.nlm.nih.gov/20876206/
9. Reaven GM. Role of insulin resistance in human disease. Diabetes. 1988;37(12):1595–1607. 🔗 https://pubmed.ncbi.nlm.nih.gov/3056758/
10. Feig DS, Zinman B, Wang X, Hux JE. Risk of development of diabetes mellitus after diagnosis of gestational diabetes. CMAJ. 2008;179(3):229–234. 🔗 https://pubmed.ncbi.nlm.nih.gov/18663202/
