Most people with high blood pressure have been given a story about their condition that is both incomplete and, in one specific detail, precisely backwards. The story goes like this: your blood pressure is elevated, the cause is primary hypertension — meaning there is no identifiable underlying cause — it is a lifelong condition, and you will take medication to manage it indefinitely. You should also reduce salt. Salt is the dietary villain. Reduce animal fat while you are at it.
This narrative is so thoroughly established in standard clinical practice that the majority of hypertensive patients accept it without question. They reduce their salt intake. They take the medication. They return for their check-up and receive either the same prescription or a stronger one. And the underlying mechanism producing their elevated blood pressure — which in the majority of metabolic hypertension cases is not salt and is not animal fat, but chronic hyperinsulinemia and the downstream vascular, renal, and inflammatory consequences it produces — continues operating, unaddressed, for years.
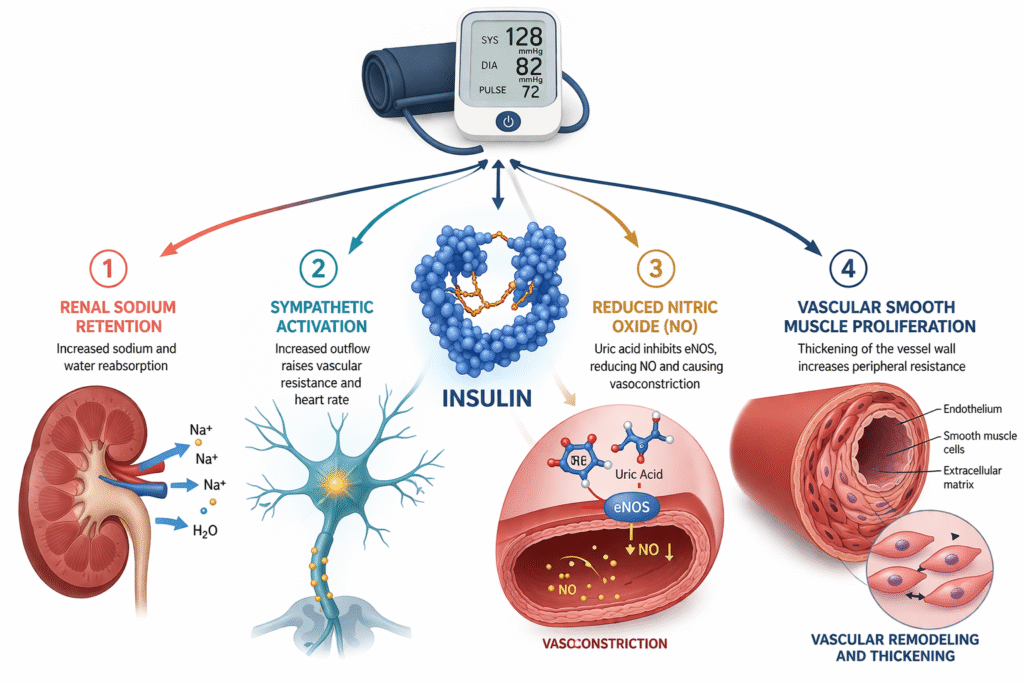
The metabolic science of hypertension tells a significantly different story from the one most patients receive. Insulin resistance and the compensatory hyperinsulinemia it produces affect blood pressure through at least four distinct and well-characterized mechanisms: renal sodium retention, sympathetic nervous system activation, endothelial dysfunction through nitric oxide suppression via uric acid elevation, and direct vascular smooth muscle proliferation. In a patient with metabolic syndrome — elevated fasting insulin, elevated TG/HDL ratio, visceral adiposity, low HDL — the blood pressure reading on the cuff is a downstream output of these upstream metabolic signals. Lower the insulin load, and the blood pressure moves. Sometimes to the amazement of the GP.
What you will learn:
The four specific mechanisms through which hyperinsulinemia elevates blood pressure | Why the salt narrative is physiologically incomplete — and why reducing salt without addressing insulin may actually keep insulin elevated | The specific metabolic marker cluster that tells you hyperinsulinemia is the primary driver of a patient’s hypertension | Why some antihypertensive medications worsen the metabolic condition producing the hypertension | What the intervention looks like and what the GP sees when it works
Clinical Perspective: What I See in Practice
When a hypertensive patient arrives in this practice and I ask them what their GP has told them about the cause of their elevated blood pressure, the answer is almost always the same. Primary hypertension. Lifelong. Take the medication. Reduce salt. Avoid animal fat. That is the sum total of the clinical explanation they have received, in some cases after years of increasingly complex antihypertensive medication regimens.
They have never heard of hyperinsulinemia. They have never been told that insulin acts on the kidney to retain sodium — that the very mechanism driving their blood pressure upward is the same mechanism that, when you reduce salt intake without addressing the insulin load, increases insulin secretion further to compensate for the sodium deficit through renal retention. Eat less salt, and insulin rises to make the kidneys hold onto the salt that remains. The blood pressure may or may not fall. The hyperinsulinemia worsens. The upstream driver of the problem becomes more entrenched.
I eat 9 grams of salt per day. High quality, unprocessed, 250-million-year-old Alpine salt from the mountains of southern Bavaria — the kind of salt that has been in this landscape since before the Alps were lifted to their current height. My blood pressure is normal. My metabolic markers are optimal. Salt is not inflammatory. Salt, consumed at physiological quantities from an unprocessed, mineral-rich source, in the context of an otherwise insulin-sensitive metabolic state, does not drive hypertension in a metabolically healthy individual.
What drives hypertension is the hyperinsulinemia that most patients with elevated blood pressure carry — visible in their elevated fasting insulin when it has been measured, in their TG/HDL ratio above 2.0, in their visceral adiposity, in their low vitamin D, their low magnesium, their elevated homocysteine, their elevated CRP. The same metabolic cluster. Different downstream number.
The clinical picture of the patients who arrive with insulin resistance-driven hypertension also consistently includes low vitamin K2 — a nutrient required for vascular calcification regulation — and a sedentary lifestyle combined with active sun avoidance, because mainstream messaging has for decades told people that sun exposure causes cancer while ignoring its role in vitamin D synthesis, nitric oxide production in skin, and vascular tone regulation.
Some patients are on anti-pregnancy pills — oral contraceptives that elevate renin-angiotensin-aldosterone system activity and compound the sodium retention and blood pressure elevation that hyperinsulinemia is already driving. Chronic stress and elevated cortisol add a further upstream driver — cortisol independently elevates blood pressure through glucocorticoid receptor activation in vascular smooth muscle and through its stimulation of hepatic glucose output that compounds the hyperinsulinemia.
The most memorable patient presentations in this area are those who had been seeing their GP three times a day because they were afraid their blood pressure might be too high. Every visit, the reading was elevated — partly because of the anxiety the visit itself was producing — and the GP’s response was to prescribe stronger or additional medication. These patients arrived trapped in a loop: hypertension anxiety driving sympathetic activation driving blood pressure elevation driving more medication driving more anxiety.
When I explained the mechanism, gave them a clear dietary and lifestyle protocol addressing the upstream insulin load, and they saw their blood pressure numbers move, the anxiety about blood pressure began to lift. They stopped seeing their GP three times a day. Reduced medication — to the GP’s amazement. In some cases the GP called and asked what they had done. What they had done was address the biology producing the number instead of suppressing the number while the biology continued.
How Hyperinsulinemia Elevates Blood Pressure: The Four Mechanisms
The causal chain from insulin resistance to hypertension is not a single pathway. It is a convergence of four independent mechanisms, each of which elevates blood pressure through a distinct biological route, and each of which is downstream of the same upstream driver: compensatory hyperinsulinemia produced by the pancreatic response to progressive insulin resistance in liver, muscle, and adipose tissue.
Mechanism 1: Renal Sodium and Water Retention
Insulin acts directly on the renal tubule — specifically the proximal tubule and the thick ascending limb of the loop of Henle — to stimulate sodium reabsorption via the Na-K-ATPase pump and the sodium-hydrogen exchanger (NHE3). In a metabolically healthy individual, the postprandial insulin rise produces a modest, temporary increase in sodium retention that is self-limiting as insulin falls between meals. In chronic hyperinsulinemia — the state produced by progressive insulin resistance — this renal sodium retention is persistent, continuous, and cumulative. The kidneys are retaining more sodium than physiological needs require, expanding extracellular fluid volume, and raising blood pressure through this volume expansion mechanism.
This is why the standard advice to reduce salt intake, while not entirely without merit in some contexts, misidentifies the primary driver. When dietary sodium is reduced without addressing the insulin load driving renal retention, the kidney compensates by increasing its reabsorptive efficiency — and insulin assists this compensation. The sodium balance may not meaningfully change, but the insulin level required to maintain it has increased. The upstream driver worsens while the downstream number may or may not improve.
Mechanism 2: Sympathetic Nervous System Activation
Insulin stimulates the sympathetic nervous system through central mechanisms — specifically through insulin receptor signaling in the hypothalamus, which increases sympathetic outflow to the vasculature, the heart, and the kidneys. Elevated sympathetic activity raises peripheral vascular resistance, increases heart rate and cardiac output, and stimulates renin release from the juxtaglomerular apparatus — activating the renin-angiotensin-aldosterone system (RAAS) and producing a further layer of sodium retention and vasoconstriction on top of the direct renal mechanism.
Chronic hyperinsulinemia therefore produces a state of tonically elevated sympathetic drive — a persistent increase in the vascular and cardiac tone that the nervous system would normally modulate in response to transient stimuli. This mechanism connects the metabolic and cardiovascular manifestations of insulin resistance: the same hyperinsulinemia that is driving hepatic glucose overproduction and hepatic fat accumulation is simultaneously maintaining an elevated sympathetic baseline that raises blood pressure through neural and hormonal pathways.
Mechanism 3: The Insulin-Uric Acid-Nitric Oxide Pathway
Chronically elevated insulin raises serum uric acid through two converging mechanisms: it stimulates renal urate reabsorption via URAT1 transporters, and it increases uric acid synthesis through fructose-driven purine metabolism in the liver — particularly relevant given the high fructose content of the ultra-processed food environment in which most metabolic patients are eating. Elevated uric acid then inhibits endothelial nitric oxide synthase (eNOS), reducing the production of nitric oxide — the primary vasodilatory signal in the vascular endothelium.
Nitric oxide maintains vascular smooth muscle relaxation, inhibits platelet aggregation, and prevents the leukocyte adhesion and vascular inflammation that initiates and accelerates atherosclerosis. When nitric oxide production is chronically suppressed by uric acid-mediated eNOS inhibition, vascular tone rises, endothelial function deteriorates, and blood pressure increases through a mechanism that operates entirely independently of dietary sodium intake. This is the mechanistic pathway through which hyperinsulinemia produces endothelial dysfunction and hypertension even in the absence of obesity, and it explains why the TOFI patient — normal weight, apparently healthy, but hyperinsulinemic — presents with elevated blood pressure that standard medicine frames as idiopathic.
Sunlight exposure directly stimulates nitric oxide production in the skin through UVA-mediated photolysis of skin nitrite stores, independent of vitamin D synthesis. This is one of the physiological reasons that sun avoidance — recommended with such confidence by mainstream medicine — may independently worsen vascular tone and contribute to the hypertension epidemic in populations with chronically reduced sun exposure.
Mechanism 4: Vascular Smooth Muscle Proliferation and Stiffness
Insulin is a growth factor — it shares downstream signaling pathways with the insulin-like growth factor-1 (IGF-1) receptor — and at chronically elevated levels, it stimulates vascular smooth muscle cell proliferation through the MAPK/ERK pathway. This proliferative effect increases the medial thickness of arterial walls, reduces their compliance, and raises pulse wave velocity — elevating systolic blood pressure and pulse pressure through a structural mechanism that reflects years of hyperinsulinemia rather than a transient functional change. Arterial stiffness is both a consequence of chronic hyperinsulinemia and an independent risk factor for cardiac events — and it is not meaningfully addressed by antihypertensive medication that lowers the pressure reading without correcting the hyperinsulinemia that has been driving the structural change.
The Metabolic Marker Cluster That Identifies Hyperinsulinemia as the Driver
Standard hypertension management involves blood pressure measurement, a basic metabolic panel, and a prescription. The metabolic marker cluster that specifically identifies hyperinsulinemia as the primary driver of the blood pressure elevation is not routinely interpreted — and in many cases is not ordered.
The combination that makes the case is this: a fasting insulin above the functional optimal of 5 µIU/mL — and particularly above 10 µIU/mL, which in my clinical assessment consistently correlates with hyperinsulinemia severe enough to drive vascular dysfunction. Alongside this, a TG/HDL ratio above 2.0 reflects hepatic insulin resistance and compensatory lipogenesis, while an increased abdominal circumference further supports the presence of central metabolic dysfunction. An hsCRP above 1.0 mg/L indicates a systemic inflammatory environment that both drives and results from chronically elevated insulin levels.
In addition, low vitamin D (below 50 ng/mL) and low magnesium — a key mineral for vascular smooth muscle relaxation, often depleted by antihypertensive medications such as thiazide diuretics — further compound the issue. Elevated homocysteine above 10 µmol/L reflects disruption in one-carbon metabolism, adding another layer of vascular stress. Finally, this entire pattern is typically reinforced by a dietary history centered on refined carbohydrates, high-fructose corn syrup, ultra-processed foods, and insufficient intake of animal protein.
This combination is consistently present in the hypertensive patients who arrive having been told their condition is primary, idiopathic, and lifelong. When these markers are interpreted together — not in isolation, not through the conventional reference ranges that report them as normal — the picture is not idiopathic. It is coherent. It has an upstream driver. And that upstream driver can be addressed.
The Problem With Some Antihypertensive Medications in a Metabolically Dysregulated Patient
The irony of the standard antihypertensive medication approach in insulin-resistant patients is that several of the most commonly prescribed drug classes actively worsen the metabolic condition producing the hypertension they are managing.
Thiazide diuretics — first-line antihypertensives — increase fasting plasma glucose by reducing pancreatic insulin secretion through hypokalemia-mediated beta cell impairment. A meta-analysis of 26 randomized trials found that thiazide diuretics significantly increased fasting plasma glucose compared with non-thiazide agents or placebo. In a patient whose blood pressure is being driven by hyperinsulinemia and insulin resistance, a medication that impairs insulin secretion and worsens glucose dysregulation may suppress the blood pressure number while compounding the metabolic dysfunction producing it. The downstream marker moves. The upstream problem worsens.
Certain beta-blockers reduce insulin sensitivity in skeletal muscle by inhibiting the glucose uptake mechanisms that normal sympathetic-parasympathetic cycling facilitates. In patients with existing insulin resistance, this effect is clinically meaningful. Calcium channel blockers produce peripheral edema through arteriolar dilation and intracapillary hypertension — a side effect requiring additional medication management in a significant proportion of long-term users, adding to the polypharmacy burden that further compromises gut barrier integrity and metabolic function as described in the gut-liver axis post.
ACE inhibitors carry a well-characterized adverse effect profile including dry cough, hypotension, hyperkalemia, and in some patients angioedema — effects that are well-documented in the research literature. The Albasri et al. 2021 BMJ systematic review found that antihypertensives as a class are associated with increased risks of acute kidney injury, hyperkalaemia, hypotension, and syncope. The Tinetti et al. JAMA Internal Medicine 2014 analysis found that antihypertensive medications are associated with an increased risk of serious fall injuries — particularly in older adults with multiple chronic conditions, for whom the blood pressure reduction that was the treatment goal may itself produce the hypotension that causes the fall.
These are not arguments against antihypertensive medication. They are arguments for understanding which patients need medication as a temporary bridge, which need it indefinitely, and what the cost-benefit calculation looks like when the metabolic condition producing the hypertension is being actively addressed alongside the pharmaceutical management of its downstream output.
What the MRC Trial Actually Showed
The Medical Research Council 1985 trial of treatment of mild hypertension — one of the most cited foundations of antihypertensive prescribing policy — randomized 17,354 patients and found that drug treatment reduced stroke rates but had no significant effect on coronary events or overall mortality in mild hypertension. To prevent one stroke, 850 patients needed to be treated for one year. Coronary events were not significantly reduced.
The Cochrane systematic review by Arguedas et al. 2020 — analyzing 11 randomized controlled trials with 38,688 participants — found that treating to blood pressure targets lower than 135/85 mmHg does not reduce mortality or serious adverse events compared with standard targets, and requires an average of one additional antihypertensive medication to achieve.
These are not fringe findings. They are the evidence base that the standard of care is built on — and they suggest that pharmaceutical management of mild to moderate hypertension produces more modest benefits in outcomes terms than the lifetime prescription model implies, particularly when the metabolic mechanism driving the blood pressure elevation is left unaddressed.
The 2017 ACC/AHA Guideline for the Management of High Blood Pressure in Adults — the most comprehensive modern hypertension guideline — does specifically recommend lifestyle modification including diet, physical activity, and weight management as first-line interventions. The gap between the guideline recommendation and the clinical reality in most GP practices — where lifestyle modification is mentioned briefly and the prescription is written at the same appointment — is where the majority of hypertensive patients spend their clinical lives.
The Intervention: What Addresses Hyperinsulinemia-Driven Hypertension
The intervention that addresses hyperinsulinemia-driven hypertension is not designed to lower blood pressure directly. It is designed to lower the insulin load that is driving the blood pressure upward through the four mechanisms described above.
Eliminate refined carbohydrates, HFCS, and ultra-processed foods. This is the primary intervention — not because carbohydrate per se causes hypertension, but because the chronic insulin elevation that continuous carbohydrate loading produces activates all four of the hypertensive mechanisms simultaneously. Removing the substrate that keeps insulin chronically elevated removes the driver of renal sodium retention, sympathetic activation, uric acid-mediated nitric oxide suppression, and vascular smooth muscle proliferation at their source.
Prioritize animal-based protein at adequate intake. Adequate protein supports muscle mass — the primary peripheral glucose sink — and exerts a significantly lower insulinogenic effect than carbohydrate. A diet that replaces carbohydrate with protein reduces the postprandial insulin requirement and lowers the chronic insulin baseline that is driving the vascular dysfunction.
Restore magnesium adequacy. Magnesium is required for vascular smooth muscle relaxation and for the eNOS function that produces nitric oxide. Its deficiency is both a consequence of the modern dietary pattern and a compounding factor in hypertension. Magnesium-rich foods — dark leafy greens, nuts, seeds, and quality animal products — or supplementation where dietary intake is insufficient, directly support vascular function independent of insulin reduction.
Restore vitamin D adequacy. Vitamin D receptor activation in vascular smooth muscle inhibits the renin-angiotensin-aldosterone system and directly reduces vascular resistance. Target 50–70 ng/mL with D3 supplementation combined with K2 to direct calcium appropriately. Regular sun exposure provides both vitamin D and the direct skin-mediated nitric oxide production that UV-A drives.
Address cortisol and stress physiology. Chronic cortisol elevation independently activates RAAS, stimulates sodium retention, and compounds the sympathetic nervous system activation that hyperinsulinemia is producing. Sleep optimization, circadian alignment, and stress reduction are not peripheral recommendations — they are mechanistically relevant interventions for a condition that has a significant neuroendocrine component in most patients.
Structured irregular fasting. As described in the fasting post, intermittent and irregular fasting reduces chronic insulin levels by creating the between-meal periods that allow insulin to fall toward baseline. Lower insulin means less renal sodium retention, lower sympathetic drive, lower uric acid from improved renal clearance, and reduced stimulus for vascular smooth muscle proliferation — a comprehensive multi-mechanism effect on blood pressure through a single physiological lever.
GP collaboration throughout. Medication reduction as blood pressure normalizes must be supervised by the prescribing physician. The functional medicine protocol does not change medications — it creates the biological conditions in which the GP has the clinical grounds to reduce them. This has happened repeatedly in this practice. The GP is surprised. They ask what changed. The patient explains. In some cases the conversation begins to shift.
A Note on Uncertainty
The mechanistic account of hyperinsulinemia-driven hypertension presented in this post — renal sodium retention via NHE3, sympathetic activation via hypothalamic insulin receptors, uric acid-mediated eNOS inhibition, and vascular smooth muscle proliferation via MAPK/ERK — is well-established in mechanistic research. What is less clearly quantified in clinical practice is the relative contribution of each mechanism in any given patient, and the proportion of hypertensive patients in whom hyperinsulinemia is the dominant rather than a contributing driver.
In a patient with primary hypertension, vascular disease, or significant renal dysfunction, the mechanisms described above may be compounding factors rather than primary causes. The intervention addresses the insulin load regardless of its relative contribution, and the blood pressure response — measured and tracked in collaboration with the GP — provides the clinical feedback that determines how much of the hypertension was metabolically driven.
Practical Implications
If you have been diagnosed with primary hypertension — if your GP has told you it is lifelong, the cause is unknown, and the treatment is medication and salt reduction — and if your fasting insulin has never been measured, your TG/HDL ratio has never been interpreted in this context, and your metabolic marker cluster has never been read as a unified picture, then the upstream mechanism driving your blood pressure has never been assessed.
The practical starting point is measuring fasting insulin if it has not been done, calculating your TG/HDL ratio from your existing lipid panel, and applying the functional medicine thresholds rather than the conventional reference ranges to the result. If fasting insulin is above 10 µIU/mL and TG/HDL is above 2.0, the metabolic environment producing your blood pressure elevation is identifiable and addressable.
The blood pressure reading on your arm is a downstream output. It is measuring the vascular consequence of a hormonal signal that your standard panel has been ignoring for years. Correct the signal, and the downstream output moves — sometimes to the amazement of the person who had been adjusting the medication every time the number appeared.
People Also Ask
What is the connection between insulin resistance and high blood pressure?
Insulin resistance and the compensatory hyperinsulinemia it produces elevate blood pressure through four mechanisms: direct renal sodium retention via tubular Na-K-ATPase stimulation, sympathetic nervous system activation through hypothalamic insulin receptor signaling, uric acid-mediated suppression of endothelial nitric oxide synthase, and vascular smooth muscle proliferation through the MAPK/ERK pathway. All four mechanisms are downstream of the same upstream driver — chronic hyperinsulinemia produced by insulin resistance in liver, muscle, and adipose tissue.
Why does reducing salt not always lower blood pressure?
Because salt is frequently not the primary driver of the blood pressure elevation. In patients with hyperinsulinemia, the kidney is retaining sodium in response to elevated insulin — not elevated dietary sodium. When dietary sodium is reduced without addressing the insulin load, insulin secretion may increase further to maintain sodium retention through improved renal efficiency. The blood pressure may not meaningfully improve because the renal sodium handling is insulin-driven, not sodium-intake-driven.
What lab markers indicate that high blood pressure is driven by insulin resistance?
The combination that indicates hyperinsulinemia as a primary driver includes: fasting insulin above 10 µIU/mL, TG/HDL ratio above 2.0, abdominal adiposity, hsCRP above 1.0 mg/L, low vitamin D below 40 ng/mL, low magnesium, elevated homocysteine above 10 µmol/L, and a dietary history built around refined carbohydrates and ultra-processed foods. This combination is present in the majority of metabolic hypertension patients and is almost never interpreted in this context in standard clinical practice.
Do some blood pressure medications worsen insulin resistance?
Yes. Thiazide diuretics increase fasting plasma glucose through hypokalemia-mediated beta cell impairment, worsening the glucose dysregulation that co-occurs with the insulin resistance driving the hypertension. Certain beta-blockers reduce skeletal muscle insulin sensitivity. These medications manage the blood pressure number while the metabolic mechanism producing it continues and may worsen. This is not an argument against their use — it is an argument for addressing the metabolic condition alongside their use.
Can blood pressure normalize naturally when insulin resistance is addressed?
In patients where hyperinsulinemia is the primary driver, yes — and the timeline depends on the severity of the insulin resistance and the consistency of the intervention. Eliminating ultra-processed foods and refined carbohydrates, restoring adequate protein and magnesium, implementing irregular intermittent fasting, and addressing vitamin D and cortisol physiology create the conditions in which all four hyperinsulinemia-driven hypertensive mechanisms simultaneously reduce in intensity. Blood pressure normalization under these conditions has been observed consistently in clinical practice. All medication reductions must be supervised by the prescribing physician.
What is the role of nitric oxide in blood pressure regulation?
Nitric oxide, produced by endothelial cells through the action of endothelial nitric oxide synthase, is the primary vasodilatory signal in the arterial wall. It relaxes vascular smooth muscle, reduces peripheral resistance, and lowers blood pressure. In hyperinsulinemia-driven hypertension, elevated uric acid — itself a consequence of insulin-stimulated renal urate retention and fructose-driven hepatic purine synthesis — inhibits eNOS and suppresses nitric oxide production. Restoring eNOS activity through uric acid reduction — achieved by lowering the insulin and fructose load — is a specific mechanistic target for blood pressure normalization.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Reaven GM. Role of insulin resistance in human disease. Diabetes. 1988;37(12):1595–1607. 🔗 https://pubmed.ncbi.nlm.nih.gov/3056758/
2. Tinetti ME, Han L, Lee DSH, et al. Antihypertensive medications and serious fall injuries in a nationally representative sample of older adults. JAMA Internal Medicine. 2014;174(4):588–595. 🔗 https://pubmed.ncbi.nlm.nih.gov/24567036/
3. Makani H, Bangalore S, Romero J, et al. Peripheral edema associated with calcium channel blockers: incidence and withdrawal rate — a meta-analysis of randomized trials. Journal of Hypertension. 2011;29(7):1270–1280. 🔗 https://pubmed.ncbi.nlm.nih.gov/21558959/
4. Na Takuathung M, Sakuludomkan W, Khatsri R, et al. Adverse effects of angiotensin-converting enzyme inhibitors in humans: a systematic review and meta-analysis of 378 randomized controlled trials. International Journal of Environmental Research and Public Health. 2022;19(14):8373. 🔗 https://pubmed.ncbi.nlm.nih.gov/35886227/
5. Albasri A, Hattle M, Koshiaris C, et al. Association between antihypertensive treatment and adverse events: systematic review and meta-analysis. BMJ. 2021;372:n189. 🔗 https://pubmed.ncbi.nlm.nih.gov/33568342/
6. Zhang X, Zhao Q. Association of thiazide-type diuretics with glycemic changes in hypertensive patients: a systematic review and meta-analysis of randomized controlled clinical trials. Journal of Clinical Hypertension (Greenwich). 2016;18(4):342–351. 🔗 https://pubmed.ncbi.nlm.nih.gov/26395424/
7. Medical Research Council Working Party. MRC trial of treatment of mild hypertension: principal results. British Medical Journal (Clinical Research Edition). 1985;291(6488):97–104. 🔗 https://pubmed.ncbi.nlm.nih.gov/2861880/
8. Arguedas JA, Leiva V, Wright JM. Blood pressure targets in adults with hypertension. Cochrane Database of Systematic Reviews. 2020;12:CD004349. 🔗 https://pubmed.ncbi.nlm.nih.gov/33332584/
9. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in Adults. Journal of the American College of Cardiology. 2018;71(19):e127–e248. 🔗 https://pubmed.ncbi.nlm.nih.gov/29133356/
10. Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiological Reviews. 2018;98(4):2133–2223. 🔗 https://pubmed.ncbi.nlm.nih.gov/30067154/
