How Sleep Apnea and Metabolic Disease Form a Vicious Cycle
Most patients with sleep apnea are told they have a breathing problem. What they are rarely told is that they have a metabolic problem — one that is being driven, sustained, and amplified every single night by repeated oxygen deprivation and stress hormone surges.
The connection between sleep apnea and metabolic disease is not incidental. It is mechanistic, bidirectional, and clinically significant. Insulin resistance promotes the anatomical conditions that cause airway obstruction. Sleep apnea then drives the hormonal environment that sustains and worsens insulin resistance. Each feeds the other — and the patient caught in that loop is often the one whose metabolic markers refuse to move despite genuine dietary effort.
This post explains the full mechanism, the clinical picture, and what actually changes when the airway problem is treated as a metabolic problem.
What you will learn:
How intermittent hypoxia drives insulin resistance independently of diet | Why sleep apnea behaves like a nightly metabolic stress event | The clinical profile of the undiagnosed sleep apnea patient | What the lab picture looks like before a diagnosis is made | Why treating sleep apnea allows the same diet and lifestyle to finally produce results | How to break the bidirectional cycle practically
What Sleep Apnea Actually Does to the Body Overnight
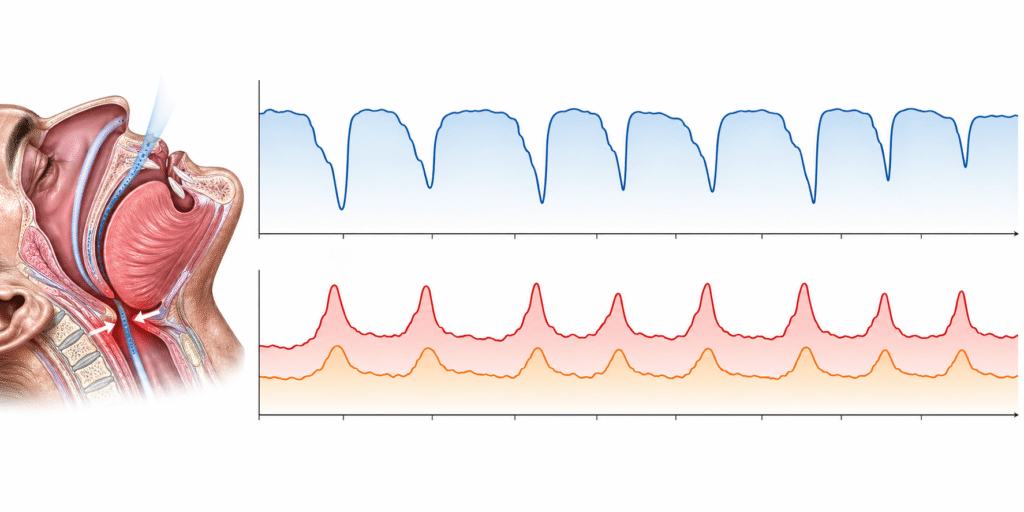
Obstructive sleep apnea is defined by repeated episodes of partial or complete upper airway obstruction during sleep. Each obstruction reduces or halts airflow, drops blood oxygen saturation, and triggers a micro-arousal that reopens the airway. In moderate to severe cases, this cycle repeats 20 to 50 or more times per hour — hundreds of times across a single night.
Each episode is not simply a breathing interruption. It is a physiological alarm event.
When blood oxygen falls, the brain activates the sympathetic nervous system immediately. Cortisol and catecholamines surge. Heart rate rises. Blood pressure spikes. The HPA axis activates as if the body is under acute physical threat. Oxygen is restored. The body settles — briefly — until the next obstruction, which arrives minutes later.
This cycle repeats across the entire night. The patient does not experience it as repeated awakenings. They experience it as unrefreshing sleep, morning fatigue, and a body that never properly recovered. What is actually happening metabolically is a nightly sequence of intermittent hypoxia, oxidative stress, inflammatory activation, and stress hormone surges that collectively produce a hormonal environment indistinguishable from chronic physiological stress.
The metabolic consequences of this environment are direct and well-characterized. Cortisol elevation at night increases hepatic glucose output and impairs insulin signaling in peripheral tissues. Catecholamine surges inhibit insulin-stimulated glucose uptake in skeletal muscle. Intermittent hypoxia activates NF-κB inflammatory signaling and generates reactive oxygen species that damage mitochondria and impair cellular insulin receptor function. Growth hormone secretion — which depends on intact slow-wave sleep — is suppressed. Each of these mechanisms operates independently. In a patient with moderate to severe sleep apnea, all of them operate simultaneously, every night.
The result is a metabolic state that no dietary protocol can fully correct — because the hormonal disruption is not originating from food. It is originating from the airway.
Intermittent Hypoxia: The Mechanism That Makes Sleep Apnea a Metabolic Driver
Of all the mechanisms through which sleep apnea drives metabolic dysfunction, intermittent hypoxia is the most clinically important and the least understood by patients.
Intermittent hypoxia — the repeated cycling of low and normal oxygen levels — is distinct from sustained hypoxia in a critical way. Where sustained hypoxia is damaging primarily through oxygen depletion, intermittent hypoxia is damaging primarily through the reperfusion injury that occurs when oxygen is restored. Each reoxygenation event generates a burst of reactive oxygen species. In a patient experiencing 30 apnea events per hour across seven hours of sleep, that represents hundreds of oxidative bursts per night — a level of mitochondrial and vascular oxidative stress that accumulates over months and years.
The metabolic consequences of chronic intermittent hypoxia are specific and measurable. In adipose tissue, hypoxia-inducible factor 1-alpha (HIF-1α) activation promotes inflammatory cytokine production and impairs adiponectin secretion. Reduced adiponectin directly worsens insulin sensitivity in the liver and skeletal muscle. In the liver, intermittent hypoxia promotes lipid accumulation, impairs beta-oxidation, and activates inflammatory pathways that worsen hepatic insulin resistance. In skeletal muscle, oxidative stress from repeated hypoxia-reoxygenation cycles impairs mitochondrial function and reduces the capacity for insulin-stimulated glucose uptake.
These effects are independent of body weight, dietary composition, and physical activity. A lean, metabolically conscious patient with untreated moderate sleep apnea is experiencing nightly mitochondrial stress and inflammatory activation that their diet cannot counteract. This is why sleep apnea should be understood as a metabolic disorder with an airway component — not a snoring problem with some downstream health consequences.
The Bidirectional Loop: How Insulin Resistance and Sleep Apnea Sustain Each Other
The relationship between sleep apnea and metabolic disease is not unidirectional. It runs in both directions simultaneously, and understanding this loop is essential for clinical management.
Insulin resistance drives visceral fat accumulation. Visceral fat deposits in the neck, tongue base, and pharyngeal walls reduce airway diameter and increase collapsibility. Hyperinsulinemia promotes fluid retention and upper airway soft tissue hypertrophy. The result is an anatomical environment in which airway obstruction during sleep is more likely, more severe, and more persistent.
Sleep apnea then sustains the insulin resistance through the overnight hormonal cascade described above. Elevated nocturnal cortisol and catecholamines maintain a state of peripheral insulin resistance and hepatic glucose overproduction. Suppressed growth hormone impairs fat oxidation and muscle recovery. Chronic intermittent hypoxia drives systemic inflammation. Fasting insulin remains elevated. Visceral fat continues to accumulate. Airway anatomy worsens.
Each half of the loop reinforces the other. The patient who is trying to address insulin resistance through diet and exercise is swimming against a biological current generated by the airway problem they do not yet know they have. And the standard approach of treating the apnea with CPAP while leaving the insulin resistance and visceral fat unaddressed means the anatomical driver of the obstruction remains intact.
Breaking this loop requires intervening on both sides simultaneously — not waiting to fix one before addressing the other.
Clinical Perspective: What I See in Practice
The sleep apnea patient rarely arrives with a sleep complaint. They arrive with a metabolic complaint.
The typical presentation is insulin resistance or stubborn type 2 diabetes, central obesity, resistant hypertension, chronic fatigue despite normal labs, and brain fog that has become so normalized they no longer mention it. They wake unrefreshed. They depend heavily on caffeine. Many report nocturia — waking two or three times per night to urinate, which they attribute to fluid intake rather than sympathetic activation. In men, low testosterone and reduced libido are frequent accompanying complaints. The body presentation includes a thick neck, mouth breathing at rest, and the characteristic facial appearance of chronic poor sleep.
The lab picture that raises suspicion before sleep is even discussed is equally consistent: fasting insulin elevated, triglycerides high, HDL low, hsCRP elevated, ferritin above the functional optimal, GGT and ALT mildly elevated, and hematocrit at the high end of normal or above it. That last finding — elevated hematocrit — is a physiological adaptation to chronic intermittent hypoxia. The body increases red blood cell production to carry more oxygen in response to repeated overnight oxygen deprivation. It is not flagged as abnormal. It is almost never connected to sleep. But in the right clinical context it is a direct readout of nocturnal hypoxia.
The pattern that makes sleep apnea move to the top of my differential before any sleep history is taken is this combination: sympathetic overdrive, stubborn insulin resistance, morning fatigue, central obesity, inflammatory labs, and hypertension that is disproportionate to what the lifestyle would predict. These patients are often already doing the right things nutritionally. They are eating low carbohydrate, exercising, genuinely trying. And the HOMA-IR is not moving, the blood pressure is not coming down, and the abdominal fat is not responding.
The explanation I give when I raise sleep apnea with these patients is direct: sleep apnea is not poor sleep — it is repeated metabolic stress all night long. The analogy that consistently lands is this: imagine someone partially cutting off your oxygen and shaking you awake 30 times per hour, every hour, all night. Your brain reads that as danger. It releases stress hormones, raises blood sugar, raises blood pressure, and keeps the entire system in survival mode rather than repair mode. You can eat perfectly — but if your body spends the night in physiological alarm mode, metabolism cannot heal.
The cases where treatment produces the clearest clinical demonstration of this mechanism are those where a patient with genuinely good dietary habits and stubborn metabolic markers begins CPAP therapy or addresses their airway through other means. Within a few weeks: morning energy improves, cravings reduce, glucose readings stabilize, resting heart rate falls, nocturia resolves. Within a few months: fasting glucose and insulin improve, blood pressure decreases, triglycerides fall, inflammatory markers drop, abdominal fat begins to move more easily. In men, testosterone and recovery often improve significantly.
The observation I hear consistently from these patients at the two to three month mark is the same: for the first time, my body responds normally to healthy habits. Before the airway was addressed, physiology was working against them despite genuine effort. Once nighttime oxygenation and sleep architecture are restored, the same diet and lifestyle that was producing no movement begins producing results — because the hormonal environment it was operating against no longer exists.
Why Sleep Apnea Is Missed in Metabolic Patients
The standard screening question for sleep apnea is whether the patient snores. This misses a substantial portion of the metabolically relevant population.
Women with sleep apnea present differently than men. Their obstructions are more frequently partial — hypopneas rather than full apneas — and their symptoms lean toward insomnia, fatigue, mood disruption, and morning headaches rather than loud snoring and witnessed apneas. Postmenopausal women in particular represent a significantly under-screened group, in whom the loss of progesterone — which has a protective effect on upper airway muscle tone — substantially increases apnea risk at precisely the time when metabolic decline is already accelerating. Their sleep apnea is frequently attributed to menopause, anxiety, or simply aging.
Lean patients with sleep apnea are missed because the condition is conceptually associated with obesity. A lean patient with a recessed jaw, crowded airway, or structural nasal obstruction can have severe sleep apnea with a normal BMI and normal neck circumference. Their metabolic dysfunction — elevated fasting insulin, persistent fatigue, resistant glucose control — is attributed to other causes while the airway problem goes uninvestigated.
The physical examination findings that increase clinical suspicion are specific and accessible: thick neck circumference above 40cm in women and 43cm in men, retrognathia or micrognathia, tongue scalloping from chronic lateral pressure against the teeth, Mallampati score of 3 or 4 indicating a crowded oropharynx, and evidence of chronic mouth breathing. These findings do not confirm sleep apnea, but in the metabolic patient who matches the clinical picture described above, they make formal sleep investigation a clinical priority rather than an optional add-on.
The Lab Constellation That Points Toward Sleep Apnea
Standard metabolic panels do not include oxygen saturation monitoring or apnea indices. The diagnosis requires sleep testing. But the lab picture in undiagnosed sleep apnea is recognizable and clinically useful as a prompt to investigate.
The consistent pattern includes elevated fasting insulin disproportionate to dietary carbohydrate intake, elevated triglycerides with low HDL, mildly elevated hsCRP and ferritin indicating chronic low-grade inflammation, mildly elevated GGT and ALT reflecting hepatic stress from intermittent hypoxia, and elevated hematocrit as the polycythemic adaptation to nocturnal oxygen deprivation. Testosterone is frequently in the low-normal to low range in men, driven partly by the suppression of LH pulsatility from sleep architecture disruption and partly from the inflammatory burden.
The dawn phenomenon — a specific pattern of fasting glucose rising between 3am and waking despite appropriate evening glucose — is a clinical clue that deserves attention in this context. It reflects overnight cortisol and catecholamine surges driving hepatic glucose output during the early morning hours. While the dawn phenomenon has multiple causes, its presence in a patient with the physical and lab picture described above should prompt sleep apnea investigation. The nocturnal stress hormone surges from apnea events are a direct mechanistic driver of this pattern.
The TG/HDL ratio is a reliable proxy for the systemic insulin resistance that sleep apnea sustains. In a patient with this ratio above 2.0 despite appropriate dietary management, and whose physical presentation and symptom cluster match the sleep apnea phenotype, the ratio is not just reflecting dietary carbohydrate load — it is reflecting the round-the-clock insulin resistance that the overnight hormonal disruption is maintaining.
Sleep Apnea, Fatty Liver, and the Hypoxic Liver
The relationship between sleep apnea and fatty liver deserves specific attention because it is clinically important and mechanistically distinct from the general insulin resistance pathway.
Intermittent hypoxia directly affects hepatic lipid metabolism. Repeated cycles of oxygen deprivation activate HIF-1α in hepatocytes, which upregulates genes involved in lipid synthesis and downregulates genes involved in fatty acid beta-oxidation. The liver produces more fat and oxidizes less of it — a combination that promotes steatosis independently of dietary fat or carbohydrate intake.
The portal vein delivers not only nutrients but also hypoxia-related inflammatory signals to the liver. In patients with sleep apnea and gut permeability — a combination that is common given the shared role of visceral adiposity and insulin resistance in both conditions — the hepatic inflammatory burden from both gut-derived endotoxin and hypoxia-driven oxidative stress is additive. This is why the combination of sleep apnea and fatty liver is so clinically consistent, and why patients with MASLD and elevated liver enzymes warrant specific sleep apnea screening even when their BMI does not flag them as obvious candidates.
Treating Sleep Apnea as a Metabolic Intervention
The conventional management of sleep apnea focuses on airway mechanics: CPAP therapy as first-line, mandibular advancement devices for mild to moderate cases, surgical airway procedures where anatomy warrants it. These are appropriate and effective for reducing apnea-hypopnea index. What they do not address is the metabolic driver of the anatomical problem — the visceral fat, hyperinsulinemia, and airway soft tissue hypertrophy that the insulin resistance is maintaining.
Effective clinical management requires both sides of the loop to be addressed simultaneously.
CPAP therapy restores overnight oxygenation and removes the nightly cortisol and catecholamine surges. This is the fastest route to reducing the overnight hormonal suppression of insulin sensitivity and allowing sleep architecture — particularly slow-wave sleep and growth hormone secretion — to normalize. For patients with significant apnea, CPAP is the most powerful single intervention for breaking the nighttime metabolic stress cycle.
Simultaneously, the metabolic work reduces visceral fat, lowers insulin, and begins reversing the anatomical drivers of airway obstruction. Dietary carbohydrate reduction and insulin-lowering nutrition reduce the hyperinsulinemia that promotes upper airway soft tissue hypertrophy and fluid retention. Resistance training improves glucose disposal and supports the muscle mass that protects airway tone. Alcohol reduction — particularly in the evening — removes a significant contributor to upper airway muscle relaxation and apnea severity.
Positional therapy is underutilized in clinical practice. A meaningful proportion of patients have predominantly positional sleep apnea — obstructions that occur primarily or exclusively in the supine position. For these patients, lateral sleep positioning alone can reduce AHI substantially. It is low-cost, free of side effects, and can be implemented immediately while other investigations and interventions are being organized.
Nasal breathing is both a treatment adjunct and a clinical signal. Chronic mouth breathing increases upper airway resistance, reduces nasal nitric oxide production — which has vasodilatory and antimicrobial properties — and promotes the retroposition of the tongue that contributes to airway collapse. Addressing chronic nasal obstruction through whatever means is appropriate — medical, structural, or behavioral — is part of the airway management picture that CPAP alone does not cover.
Circadian Disruption, Sleep Deprivation, and Sleep Apnea: The Cluster
In practice, sleep apnea rarely exists in isolation. The majority of patients with obstructive sleep apnea also have circadian rhythm disruption from the sleep fragmentation and overnight sympathetic activation that apnea produces. Many also carry a significant component of sleep deprivation — both because the architectural disruption of apnea prevents restorative sleep stages regardless of time in bed, and because the fatigue that untreated apnea produces drives behavioral patterns, including late nights and irregular schedules, that worsen circadian alignment.
This clustering matters clinically because treating the apnea alone — while leaving circadian disruption and sleep restriction unaddressed — produces incomplete metabolic recovery. The full picture requires restoring nighttime oxygenation, re-anchoring the circadian clock through morning light and consistent wake timing, and ensuring that total sleep opportunity is adequate once the apnea is controlled. These three interventions are not sequential. They are simultaneous, and their metabolic effects are additive.
The role of melatonin timing in this context deserves specific mention. Sleep apnea disrupts sleep architecture in a way that fragments the melatonin secretion profile — the repeated micro-arousals interrupt the continuous overnight melatonin rise that governs the transition from active metabolism to repair mode. Restoring nighttime oxygenation through CPAP allows melatonin secretion to normalize structurally. Supporting melatonin onset through evening darkness and light correction accelerates that normalization. The two interventions act on different parts of the same circadian-metabolic signaling system.
Sleep apnea is one mechanism within a larger system of sleep- and circadian-driven metabolic damage; for the full clinical framework that places airway obstruction alongside sleep deprivation, melatonin disruption, nighttime cortisol dysregulation, and shift work, see the cornerstone on sleep and the body clock as the master regulators of metabolism.
A Note on Uncertainty
The association between obstructive sleep apnea and insulin resistance, type 2 diabetes, hypertension, and cardiovascular disease is among the most consistently replicated findings in sleep medicine. The mechanistic pathways — intermittent hypoxia, oxidative stress, sympathetic activation, inflammatory signaling — are well-characterized in both human and experimental data.
What carries more uncertainty is the magnitude and reversibility of metabolic benefit from CPAP treatment specifically. Randomized controlled trials of CPAP therapy in metabolic patients have shown inconsistent results across different metabolic outcomes — some show significant improvements in insulin sensitivity, blood pressure, and inflammatory markers; others show modest or non-significant effects. This inconsistency likely reflects heterogeneity in patient selection, CPAP adherence, the degree to which metabolic interventions were co-applied, and the duration of follow-up.
The clinical pattern described in this post — metabolic recovery that becomes possible once the airway is treated and the bidirectional loop is broken — is consistent with the mechanistic literature and with what is observed repeatedly in practice. It does not mean CPAP alone corrects insulin resistance. It means that CPAP, as part of a comprehensive metabolic intervention, removes a significant biological obstacle that dietary and lifestyle changes alone cannot overcome.
Formal sleep testing — polysomnography or validated home sleep testing — is the appropriate diagnostic step where sleep apnea is clinically suspected. Management decisions, particularly regarding CPAP initiation and medical comorbidities, should involve the treating physician and where appropriate a sleep medicine specialist.
People Also Ask
How does sleep apnea cause insulin resistance?
Sleep apnea produces repeated episodes of intermittent hypoxia, cortisol and catecholamine surges, and NF-κB inflammatory activation throughout the night. These mechanisms impair insulin signaling in the liver, skeletal muscle, and adipose tissue independently of dietary intake. The result is insulin resistance that is sustained around the clock by the overnight hormonal disruption — and that cannot be fully corrected by dietary intervention while the apnea remains untreated.
Can treating sleep apnea improve blood sugar and metabolic markers?
Yes, particularly in patients where the sleep apnea is a significant contributor to the metabolic picture. Restoring nighttime oxygenation reduces nocturnal cortisol and catecholamine surges, allows growth hormone secretion and slow-wave sleep to normalize, and removes the chronic inflammatory burden of intermittent hypoxia. In patients with well-constructed diets whose markers have plateaued, CPAP therapy combined with metabolic intervention frequently produces improvements in fasting glucose, fasting insulin, triglycerides, blood pressure, and inflammatory markers within weeks to months.
What are the signs that sleep apnea may be driving metabolic dysfunction?
The clinical pattern includes insulin resistance or elevated fasting glucose disproportionate to dietary carbohydrate intake, central obesity with resistant hypertension, chronic fatigue despite normal labs, morning unrefreshingness, nocturia, heavy caffeine dependence, and a lab picture showing elevated fasting insulin, high triglycerides, low HDL, elevated hsCRP and ferritin, mildly elevated liver enzymes, and hematocrit at the high end of normal. The pattern that most strongly suggests sleep apnea is metabolic dysfunction that is disproportionate to the quality of the lifestyle the patient is already living.
Is sleep apnea more common in people with insulin resistance?
Yes, and the relationship is bidirectional. Insulin resistance promotes visceral fat accumulation in the neck and pharyngeal walls, narrows the airway, and increases obstructive risk. Sleep apnea then sustains insulin resistance through its overnight hormonal effects. The two conditions share overlapping pathophysiology and co-occur at rates significantly higher than chance. In clinical populations with metabolic syndrome, the prevalence of obstructive sleep apnea is substantially higher than in the general population.
Can you have sleep apnea without being overweight?
Yes. While obesity increases risk significantly, sleep apnea occurs in lean individuals due to anatomical factors including retrognathia, crowded oropharynx, nasal obstruction, and low muscle tone. Lean patients with sleep apnea are frequently missed because the condition is conceptually associated with obesity. Their metabolic dysfunction — elevated fasting insulin, persistent fatigue, resistant glucose control — is often attributed to other causes while the airway problem goes uninvestigated.
What is the connection between sleep apnea and fatty liver?
Intermittent hypoxia directly impairs hepatic lipid metabolism by activating HIF-1α, which upregulates fat synthesis genes and suppresses fat oxidation. This promotes hepatic steatosis independently of dietary fat or carbohydrate intake. The combination of sleep apnea and fatty liver is clinically consistent, and patients with MASLD and elevated liver enzymes warrant sleep apnea screening even when their BMI does not flag them as obvious candidates.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
- Punjabi NM. The epidemiology of adult obstructive sleep apnea. Proceedings of the American Thoracic Society. 2008;5(2):136–143. 🔗 https://pubmed.ncbi.nlm.nih.gov/18250205/
- Punjabi NM, Sorkin JD, Katzel LI, et al. Sleep-disordered breathing and insulin resistance in middle-aged and overweight men. American Journal of Respiratory and Critical Care Medicine. 2002;165(5):677–682. 🔗 https://pubmed.ncbi.nlm.nih.gov/11874813/
- Aronsohn RS, Whitmore H, Van Cauter E, Tasali E. Impact of untreated obstructive sleep apnea on glucose control in type 2 diabetes. American Journal of Respiratory and Critical Care Medicine. 2010;181(5):507–513. 🔗 https://pubmed.ncbi.nlm.nih.gov/19926868/
- Drager LF, Togeiro SM, Polotsky VY, Lorenzi-Filho G. Obstructive sleep apnea: a cardiometabolic risk in obesity and the metabolic syndrome. Journal of the American College of Cardiology. 2013;62(7):569–576. 🔗 https://pubmed.ncbi.nlm.nih.gov/23770180/
- Polotsky VY, Li J, Punjabi NM, et al. Intermittent hypoxia increases insulin resistance in genetically obese mice. Journal of Physiology. 2003;552(1):253–264. 🔗 https://pubmed.ncbi.nlm.nih.gov/12878760/
- Drager LF, Polotsky VY, Lorenzi-Filho G. Obstructive sleep apnea: an emerging risk factor for atherosclerosis. Chest. 2011;140(2):534–542. 🔗 https://pubmed.ncbi.nlm.nih.gov/21813534/
- Ip MS, Lam B, Ng MM, et al. Obstructive sleep apnea is independently associated with insulin resistance. American Journal of Respiratory and Critical Care Medicine. 2002;165(5):670–676. 🔗 https://pubmed.ncbi.nlm.nih.gov/11874812/
- Tasali E, Mokhlesi B, Van Cauter E. Obstructive sleep apnea and type 2 diabetes: interacting epidemics. Chest. 2008;133(2):496–506. 🔗 https://pubmed.ncbi.nlm.nih.gov/18252913/
- Aron-Wisnewsky J, Minville C, Tordjman J, et al. Chronic intermittent hypoxia is a major trigger for non-alcoholic fatty liver disease in morbid obese patients. Journal of Hepatology. 2012;56(1):225–233. 🔗 https://pubmed.ncbi.nlm.nih.gov/21703181/
- Mokhlesi B, Ham SA, Gozal D. The effect of sex and age on the comorbidity burden of OSA: an observational analysis from a large nationwide US health claims database. European Respiratory Journal. 2016;47(4):1162–1169. 🔗 https://pubmed.ncbi.nlm.nih.gov/26917611/
- Kohler M, Stradling JR. Mechanisms of vascular damage in obstructive sleep apnea. Nature Reviews Cardiology. 2010;7(12):677–685. 🔗 https://pubmed.ncbi.nlm.nih.gov/20938455/
- Hoyos CM, Killick R, Yee BJ, Grunstein RR, Liu PY. Effects of testosterone therapy on sleep and breathing in obese men with severe obstructive sleep apnoea. European Journal of Endocrinology. 2012;167(4):549–557. 🔗 https://pubmed.ncbi.nlm.nih.gov/22865915/