How Nighttime Cortisol Dysregulation Silently Drives Insulin Resistance and Visceral Fat
Most patients think of cortisol as a daytime hormone. They associate it with stress, with the morning rush, with the fight-or-flight response that activates when demands pile up. What they do not know is that cortisol’s behavior at night is equally important metabolically — and that when cortisol fails to fall properly after dark, the metabolic consequences are specific, measurable, and directly connected to insulin resistance — largely invisible to standard medical screening.
Nighttime cortisol dysregulation is not the same as general stress or poor sleep. It is a specific failure of the HPA axis to complete its daily downregulation cycle — a state in which the body remains partially activated through the hours that should be dedicated to repair, fat oxidation, growth hormone secretion, and glucose stabilization. The patient is technically asleep. Metabolically, the body is behaving as if it is under chronic overnight pressure.
This post explains the mechanism, the clinical profile, the lab picture, and what changes when nighttime cortisol is specifically addressed — not just as a side effect of fixing sleep, but as a primary metabolic target in its own right.
What you will learn: How the HPA axis is supposed to downregulate at night and why it fails | What chronic nighttime cortisol elevation does to glucose, visceral fat, muscle, and growth hormone | The clinical profile of the patient whose HPA axis never fully switches off | How to recognize the pattern before lab work confirms it | What testing reveals that a single morning cortisol misses entirely | What intervention actually looks like in practice
The Normal Cortisol Rhythm and Why Nighttime Matters Most
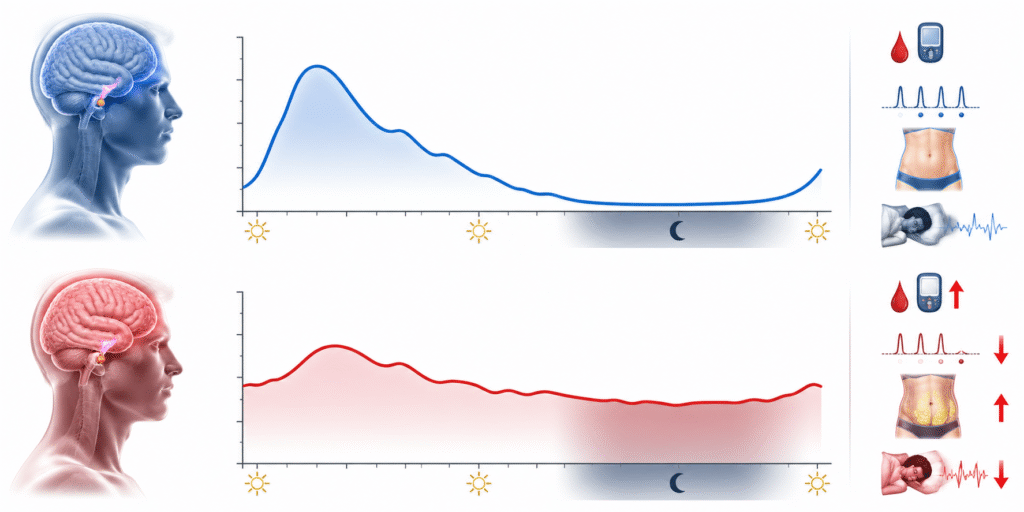
Cortisol follows a precise 24-hour arc. It reaches its nadir — its lowest point — between midnight and 2am. It begins rising in the pre-dawn hours. It peaks sharply within 30 to 45 minutes of waking in what is known as the cortisol awakening response. It then declines progressively through the morning and afternoon, reaching low levels again by evening.
This arc is not incidental. It is the primary hormonal scaffold around which the body organizes its metabolic day and night. The morning peak mobilizes glucose, activates gluconeogenesis transiently, prepares skeletal muscle for insulin-stimulated uptake, and synchronizes the peripheral clocks in the liver and pancreas — as covered in detail in the circadian rhythm and metabolism post. The nighttime nadir is equally critical, and it is the half of the rhythm that medicine almost entirely ignores.
When cortisol reaches its overnight nadir, a cascade of repair processes activates. Growth hormone secretion peaks in the first slow-wave sleep cycle. Muscle protein synthesis runs at its highest rate. Fat oxidation is prioritized. Hepatic glucose output drops to its lowest point. Insulin secretion falls. The immune system performs anti-inflammatory housekeeping. The brain consolidates memory and clears metabolic waste through the glymphatic system.
Every one of these processes requires the low-cortisol environment that the healthy overnight nadir provides. Cortisol and growth hormone are physiological antagonists — when one is high, the other is suppressed. Cortisol and insulin operate in chronic opposition — when cortisol rises at night, hepatic glucose output rises with it, creating fasting glucose elevation that appears on the morning panel without any dietary explanation. Cortisol and fat oxidation are inversely related in visceral fat depots — elevated nighttime glucocorticoid signaling specifically promotes lipid accumulation in the abdomen through glucocorticoid receptor activation in visceral adipocytes.
When the HPA axis fails to complete its nightly downregulation, none of these repair processes can run at full capacity. The patient sleeps. The body does not recover.
Why the HPA Axis Fails to Downregulate at Night
The HPA axis is regulated by a negative feedback loop. Cortisol produced by the adrenal glands circulates back to the hypothalamus and pituitary, where it suppresses further CRH and ACTH release. Under healthy conditions, this feedback loop is tight and responsive — cortisol rises when demanded, and falls when the signal resolves.
Chronic stress — particularly cognitive, psychological, and emotional stress that does not have a physical resolution — progressively impairs this feedback loop. The hypothalamus remains partially activated. CRH secretion does not fully suppress. The adrenal glands continue receiving low-level stimulation throughout the night. The result is a cortisol curve that does not reach its proper nadir — it stays elevated at a low but metabolically significant level through the hours when it should be near zero.
This is distinct from the acute cortisol surges described in the sleep apnea post, where each apnea event triggers a sharp catecholamine and cortisol spike. Nighttime cortisol dysregulation from HPA axis impairment is a chronic, low-amplitude elevation — not dramatic, not symptomatic in any obvious way, but sufficient to suppress growth hormone, elevate hepatic glucose output, promote visceral fat deposition, and prevent the deep sleep stages that restorative recovery requires.
The impaired feedback loop also affects the morning cortisol awakening response. In the early stages of HPA dysregulation, the morning peak is exaggerated — the system overcompensates for poor overnight suppression with an overshooting morning rise. In more advanced or chronic cases, the CAR becomes blunted — the system is exhausted, the morning peak fails to fire cleanly, and the patient wakes with the characteristic combination of low morning cortisol and high evening cortisol that is the signature of a fully inverted rhythm. This is often described clinically as burnout physiology.
The triggers that sustain this impaired feedback loop are specific and recognizable: unresolved cognitive load that follows the patient into the evening, chronic emotional stress without adequate discharge, hypervigilance from past trauma, competitive or perfectionist personality patterns that keep the prefrontal cortex activated at night, and the physiological drivers — chronic pain, sleep-disordered breathing, blood sugar instability, and overtraining — that maintain sympathetic tone independently of psychological stress.
What Elevated Nighttime Cortisol Does to Metabolism
The metabolic consequences of chronically elevated nighttime cortisol operate through five distinct but interconnected pathways.
Hepatic glucose overproduction. Cortisol stimulates gluconeogenesis in the liver through multiple mechanisms — it activates PEPCK and G6Pase, the key enzymes of gluconeogenesis, and it promotes the release of gluconeogenic substrates from peripheral tissues. When nighttime cortisol is elevated, the liver produces more glucose during the overnight fasting period than it should. The result is fasting glucose that is elevated in the morning not because of dietary carbohydrate intake but because the liver has been producing glucose through the night. This is the physiological basis of the dawn phenomenon — the pattern of rising fasting glucose between 3am and waking that is one of the most consistent lab findings in patients with nighttime cortisol dysregulation.
This mechanism also contributes directly to hepatic fat accumulation. For the full picture of how insulin resistance and hepatic lipid dysregulation interact, see Fatty Liver and Metabolic Dysfunction.
Visceral fat accumulation. Visceral adipose tissue expresses glucocorticoid receptors at higher density than subcutaneous fat. Cortisol binding to these receptors promotes lipoprotein lipase activity — the enzyme that captures circulating lipids and stores them in fat cells — while simultaneously impairing fat mobilization through hormone-sensitive lipase inhibition. The net effect is preferential fat deposition in the abdominal depot during periods of elevated cortisol. This is why the patient with chronic nighttime cortisol dysregulation accumulates central fat disproportionately to overall caloric intake, and why dietary intervention alone fails to move the abdominal circumference that cortisol is actively maintaining.
Growth hormone suppression. Growth hormone and cortisol are direct physiological antagonists. The primary GH secretory pulse occurs in slow-wave sleep, in the low-cortisol environment of the overnight nadir. Elevated nighttime cortisol suppresses this pulse through two mechanisms: it directly inhibits GHRH release from the hypothalamus, and it promotes somatostatin secretion, which blocks GH release from the pituitary. In practice, this means reduced overnight muscle protein synthesis, impaired fat oxidation during sleep, slower tissue repair, and a metabolic state that progressively loses lean mass while retaining and accumulating fat — independent of dietary protein intake.
Insulin resistance perpetuation. Cortisol impairs insulin signaling at multiple points — it reduces GLUT4 translocation in skeletal muscle, promotes hepatic insulin resistance through DAG accumulation, and drives compensatory hyperinsulinemia through its gluconeogenic and glycogenolytic effects. When this happens overnight, fasting insulin is elevated in the morning not from the previous day’s dietary load but from the overnight cortisol-driven glucose production that demanded insulin compensation. The HOMA-IR calculation in these patients consistently overestimates dietary insulin resistance — because the insulin resistance it is measuring is partly cortisol-driven and overnight in origin.
Sleep architecture destruction. Cortisol actively suppresses slow-wave sleep. Elevated nighttime cortisol shortens and fragments the deep sleep stages during which GH peaks, mitochondrial repair runs, and memory consolidation occurs. Less slow-wave sleep means less GH, less cellular repair, less cognitive recovery — and, critically, less of the overnight insulin sensitivity restoration that depends on adequate slow-wave sleep. This creates a self-reinforcing loop: elevated nighttime cortisol destroys the sleep architecture that would normally allow cortisol to fall, which sustains the elevated cortisol, which further destroys the sleep architecture. The patient wakes unrefreshed after adequate hours and cannot understand why.
Clinical Perspective: What I See in Practice
The patient with nighttime cortisol dysregulation is one of the most recognizable phenotypes in functional metabolic practice — and one of the most consistently missed in conventional medicine.
They do not present with a sleep complaint. They present with metabolic dysfunction that is disproportionate to their lifestyle. They eat well. They exercise. They are disciplined. And their abdominal fat will not move, their fasting glucose stays elevated, their energy is chronically poor, and nothing they do seems to produce the metabolic response it should.
The clinical picture before any testing is specific. They describe being exhausted all day and wired at night — a second wind that arrives around 9 to 11pm, at precisely the time cortisol should be at its lowest. They cannot fall asleep despite being physically tired because their brain will not stop — planning, problem-solving, reviewing, running through tomorrow’s demands. When they do fall asleep, they wake between 2 and 4am with a specific quality of alertness that is distinct from general insomnia: their mind is sharp, their thoughts are active, and returning to sleep takes an hour or longer. They wake in the morning feeling as if they never slept.
The behavioral clues are equally consistent. Evening cravings for carbohydrates, chocolate, alcohol, or screens — all cortisol-driven attempts to modulate a nervous system that will not downregulate. Teeth grinding or jaw tension on waking. Night sweats or a sensation of running hot. A resting heart rate that stays elevated through the night rather than dropping into the normal overnight range. Anxiety or irritability that peaks in the evening. Heavy caffeine dependence in the morning to compensate for non-restorative sleep.
The metabolic presentation includes elevated fasting glucose despite dietary compliance, central fat accumulation that is disproportionate to overall body composition, and a dawn phenomenon that produces the highest glucose readings of the day on an empty stomach before eating anything. These patients often say: “I eat well, but my abdominal fat and morning glucose won’t move.” That statement, in the right clinical context, is almost a direct description of chronic nighttime cortisol elevation driving visceral fat accumulation and hepatic glucose overproduction simultaneously.
The lab constellation that confirms the clinical suspicion includes elevated fasting insulin disproportionate to carbohydrate intake, elevated triglycerides, mildly elevated hsCRP and ferritin, mildly elevated GGT and ALT, and sometimes low testosterone in men or reduced progesterone resilience in women — both of which are suppressed by chronic cortisol excess through its inhibitory effect on the HPG axis. A standard morning serum cortisol is almost entirely useless in this context. The problem is not morning cortisol — it is the failure of cortisol to fall at night.
A morning cortisol can be perfectly normal while midnight cortisol is significantly elevated. The test that captures this is a multi-point salivary cortisol curve or DUTCH testing with a diurnal cortisol rhythm — measurements at waking, mid-morning, afternoon, evening, and bedtime. The pattern that confirms nighttime dysregulation is elevated evening or midnight cortisol with a flattened overall rhythm and, in more chronic cases, a blunted morning cortisol awakening response.
The interventions that shift this picture are not primarily dietary. They target the total nighttime stress signal to the nervous system. Strict sleep and wake timing. Earlier meals — blood glucose instability in the evening is an independent HPA axis activator, and a destabilizing late dinner can maintain cortisol through mechanisms entirely separate from psychological stress. Reducing evening cognitive load — no work screens, no problem-solving, no high-stimulation content in the two hours before sleep. Active parasympathetic downshifting through breathwork, slow walking, or other nervous system deactivation practices. Correcting sleep-disordered breathing where present. Reducing or eliminating evening caffeine and alcohol — both of which raise cortisol and fragment sleep architecture despite their subjective calming effects.
What shifts first is consistently the 2 to 4am awakening pattern — it reduces within the first one to two weeks of intervention, which is both clinically meaningful and motivating for the patient. Morning energy improves. Evening cravings reduce without dietary effort. Then the metabolic markers begin to move: fasting glucose drops, dawn phenomenon decreases, abdominal fat starts to respond, blood pressure lowers, TG/HDL ratio improves gradually.
The patient statement that appears consistently at the four to six week mark is this: “For the first time, I feel like my body is recovering overnight instead of fighting all night.” That shift — from overnight stress to overnight repair — is what allows the existing metabolic work to finally produce results.
The Patient Profile Where This Is Always Part of the Picture
There is a highly consistent phenotype where nighttime cortisol dysregulation is not a possibility to investigate — it is an expectation.
The most consistent profile is the high-performing, hyper-responsible individual who cannot mentally switch off. Executives and entrepreneurs under chronic cognitive load. Chronic caregivers — parents, medical professionals, anyone in a sustained role of responsibility for others. Overtrained fitness-oriented patients who use physical training as a stress outlet but whose training load itself becomes an additional physiological stressor.
Patients with unresolved trauma or persistent hypervigilance, whose nervous system remains in a low-level threat-detection state regardless of current circumstances. Perimenopausal and postmenopausal women under chronic stress load, in whom the loss of progesterone — which has direct GABAergic and calming effects — removes a significant buffer against HPA axis hyperactivation.
The common denominator across all of these profiles is sustained sympathetic activation without genuine recovery. These patients are not simply stressed — they have lost the physiological capacity to downregulate. Their nervous system has habituated to a state of partial activation, and the transition from day to night no longer produces the cortisol suppression that biology requires.
In this group, assessing nighttime cortisol regulation is not optional. It is part of the standard metabolic evaluation — as standard as checking fasting insulin or the TG/HDL ratio.
The Cortisol-Sleep Loop: Why This Becomes Self-Perpetuating
One of the most clinically important features of nighttime cortisol dysregulation is that it is self-reinforcing. Once established, it tends to perpetuate itself through a loop that does not resolve without deliberate intervention.
Elevated nighttime cortisol suppresses slow-wave sleep. Insufficient slow-wave sleep means the HPA axis does not receive the downregulation signal it needs — deep sleep itself is one of the inputs that suppresses CRH and cortisol secretion overnight. Less deep sleep therefore means less HPA suppression, which means higher cortisol, which means less deep sleep. The loop tightens progressively.
As described in how poor sleep causes insulin resistance, insufficient slow-wave sleep also impairs insulin sensitivity the following day. The resulting metabolic stress — unstable glucose, elevated insulin, cravings, energy dysregulation — feeds back into the psychological and physiological stress load that activates the HPA axis the following night. The metabolic consequences of the cortisol dysregulation are themselves cortisol-activating.
This is why patients in this loop often feel as if the problem is getting gradually worse despite their best efforts. They are not imagining it. The loop is tightening. Breaking it requires targeting the cortisol side directly — not just improving sleep hygiene and waiting for cortisol to follow, but actively reducing the HPA activation load through the specific interventions that address the inputs maintaining it.
The connection to melatonin timing is also relevant here. Elevated nighttime cortisol and delayed or suppressed melatonin onset frequently co-occur — cortisol inhibits melatonin secretion through direct suppression of pineal gland activity. In patients with both patterns, addressing cortisol dysregulation without also correcting melatonin timing produces incomplete results. The two systems are not independent. They are part of the same circadian-hormonal network that needs to be realigned as a whole.
Nighttime cortisol elevation is one of the most clinically actionable signals in the broader pattern of sleep- and circadian-driven metabolic dysfunction; the unified framework, diagnostic sequence, and intervention hierarchy are detailed in the cornerstone on sleep, circadian rhythm, and metabolic health.
A Note on Uncertainty
The role of chronic cortisol elevation in visceral fat accumulation, insulin resistance, and sleep architecture disruption is well-established in both mechanistic and clinical research. The direct consequences of Cushing’s syndrome — pathological cortisol excess — provide a clear proof of concept for what chronic cortisol elevation does to body composition, glucose metabolism, and recovery capacity.
What carries more uncertainty is the precise threshold of subclinical nighttime cortisol elevation that produces clinically significant metabolic consequences. The transition from normal variation to pathological dysregulation is a continuum, not a threshold. Multi-point salivary cortisol and DUTCH testing provide more clinically useful information than single-point serum cortisol, but interpretation of these patterns requires clinical judgment rather than rigid reference ranges.
The interventions described in this post — sleep timing, evening stress reduction, parasympathetic activation, blood glucose stabilization — are low-risk behavioral modifications with broad evidence bases for their effects on HPA axis regulation and sleep quality. They are not replacements for clinical assessment. Where burnout, trauma, significant mood disorder, or adrenal pathology is suspected, collaboration with a physician or specialist is the appropriate next step.
People Also Ask
What is nighttime cortisol dysregulation?
Nighttime cortisol dysregulation is the failure of the HPA axis to complete its daily downregulation cycle. Instead of reaching its overnight nadir between midnight and 2am, cortisol remains elevated at a low but metabolically significant level through the sleep period. This suppresses growth hormone secretion, promotes hepatic glucose overproduction, drives visceral fat accumulation, and destroys the deep sleep stages required for metabolic recovery.
How does elevated nighttime cortisol cause insulin resistance?
Cortisol stimulates hepatic gluconeogenesis and impairs insulin signaling in skeletal muscle and the liver. When cortisol remains elevated overnight, the liver produces excess glucose during the fasting sleep period, demanding compensatory insulin secretion. The result is elevated fasting insulin and fasting glucose in the morning — an insulin resistance pattern that is cortisol-driven rather than diet-driven, and that dietary intervention alone cannot fully correct.
What are the signs of nighttime cortisol dysregulation?
The key clinical pattern includes a second wind in the late evening when cortisol should be falling, difficulty falling asleep despite exhaustion, waking between 2 and 4am with alertness and racing thoughts, non-restorative sleep despite adequate hours, elevated fasting glucose in the morning, central fat accumulation disproportionate to overall diet quality, and a dawn phenomenon where fasting glucose is highest before eating anything.
What is the dawn phenomenon and is it related to cortisol?
The dawn phenomenon is the pattern of rising fasting glucose between approximately 3am and waking, independent of food intake. It is directly driven by the cortisol and growth hormone rise in the pre-dawn hours, which stimulates hepatic glucose production in preparation for waking. In patients with nighttime cortisol dysregulation, this pre-dawn rise is exaggerated because cortisol has remained elevated throughout the night, and the liver has been producing glucose continuously rather than suppressing output during the early overnight hours.
What testing is most useful for nighttime cortisol dysregulation?
A single morning serum cortisol is insufficient — it can be entirely normal while nighttime cortisol is significantly elevated. Multi-point salivary cortisol testing, measuring cortisol at waking, mid-morning, afternoon, evening, and bedtime, provides a complete picture of the diurnal rhythm. DUTCH testing offers a similar diurnal cortisol profile alongside additional metabolic markers. The pattern to look for is elevated evening or midnight cortisol, a flattened overall rhythm, and in more advanced cases a blunted morning cortisol awakening response.
Can fixing nighttime cortisol improve metabolic markers without changing diet?
Yes, particularly in patients where cortisol dysregulation is the rate-limiting variable. Reducing the total nighttime stress signal — through earlier meals, evening light reduction, cognitive downshifting, and parasympathetic activation practices — consistently improves the 2 to 4am awakening pattern within one to two weeks, followed by improvements in fasting glucose, dawn phenomenon, abdominal fat responsiveness, and triglycerides over four to eight weeks. The diet does not change. The hormonal environment in which the diet operates does.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
- Tsigos C, Chrousos GP. Hypothalamic-pituitary-adrenal axis, neuroendocrine factors and stress. Journal of Psychosomatic Research. 2002;53(4):865–871. 🔗 https://pubmed.ncbi.nlm.nih.gov/12377295/
- Björntorp P, Rosmond R. Obesity and cortisol. Nutrition. 2000;16(10):924–936. 🔗 https://pubmed.ncbi.nlm.nih.gov/11054598/
- Leproult R, Copinschi G, Buxton O, Van Cauter E. Sleep loss results in an elevation of cortisol levels the next evening. Sleep. 1997;20(10):865–870. 🔗 https://pubmed.ncbi.nlm.nih.gov/9415946/
- Van Cauter E, Leproult R, Plat L. Age-related changes in slow wave sleep and REM sleep and relationship with growth hormone and cortisol levels in healthy men. JAMA. 2000;284(7):861–868. 🔗 https://pubmed.ncbi.nlm.nih.gov/10938176/
- Rosmond R, Dallman MF, Björntorp P. Stress-related cortisol secretion in men: relationships with abdominal obesity and endocrine, metabolic and hemodynamic abnormalities. Journal of Clinical Endocrinology & Metabolism. 1998;83(6):1853–1859. 🔗 https://pubmed.ncbi.nlm.nih.gov/9626108/
- Dallman MF, Pecoraro N, Akana SF, et al. Chronic stress and obesity: a new view of comfort food. PNAS. 2003;100(20):11696–11701. 🔗 https://pubmed.ncbi.nlm.nih.gov/12975492/
- Spiegel K, Leproult R, Van Cauter E. Impact of sleep debt on metabolic and endocrine function. The Lancet. 1999;354(9188):1435–1439. 🔗 https://pubmed.ncbi.nlm.nih.gov/10543671/
- Chrousos GP. Stress and disorders of the stress system. Nature Reviews Endocrinology. 2009;5(7):374–381. 🔗 https://pubmed.ncbi.nlm.nih.gov/19488073/
- Epel ES, McEwen B, Seeman T, et al. Stress and body shape: stress-induced cortisol secretion is consistently greater among women with central fat. Psychosomatic Medicine. 2000;62(5):623–632. 🔗 https://pubmed.ncbi.nlm.nih.gov/11020091/
- Plat L, Leproult R, L’Hermite-Baleriaux M, et al. Metabolic effects of short-term elevations of plasma cortisol are more pronounced in the evening than in the morning. Journal of Clinical Endocrinology & Metabolism. 1999;84(9):3082–3092. 🔗 https://pubmed.ncbi.nlm.nih.gov/10487666/
- Tasali E, Leproult R, Ehrmann DA, Van Cauter E. Slow-wave sleep and the risk of type 2 diabetes in humans. PNAS. 2008;105(3):1044–1049. 🔗 https://pubmed.ncbi.nlm.nih.gov/18172212/
- Adam TC, Epel ES. Stress, eating and the reward system. Physiology & Behavior. 2007;91(4):449–458. 🔗 https://pubmed.ncbi.nlm.nih.gov/17543357/