How Poor Sleep and Glucose Control Are Connected in Real Time
Most people understand that diet affects blood sugar. Very few understand that the night before a meal matters as much as the meal itself. Poor sleep does not simply leave the body tired — it reconfigures the entire metabolic response to food for the following 24 hours. The same breakfast eaten after seven hours of restorative sleep and after five hours of fragmented sleep produces measurably different glucose curves. The food has not changed. The metabolic context has.
The connection between poor sleep and glucose control is direct, real-time, and bidirectional. Poor sleep worsens glucose handling. Unstable glucose worsens sleep quality. The patient caught in that loop is often the one whose metabolic markers refuse to stabilize despite genuine dietary effort — because neither side of the loop is being addressed with the same seriousness as the diet.
This post focuses on glucose behavior specifically — not insulin resistance as an abstract mechanism, but what actually happens to blood sugar when sleep is insufficient, what that looks like on a continuous glucose monitor, and what to do on the day after poor sleep to minimize the damage.
What you will learn: How poor sleep alters the postprandial glucose curve | What CGM reveals about overnight glucose instability before fasting glucose becomes abnormal | The bidirectional loop between dysglycemia and poor sleep | Why glucose variability is a more sensitive marker of sleep disruption than fasting glucose | Practical dietary strategies for the day after poor sleep
What Poor Sleep Does to the Postprandial Glucose Curve
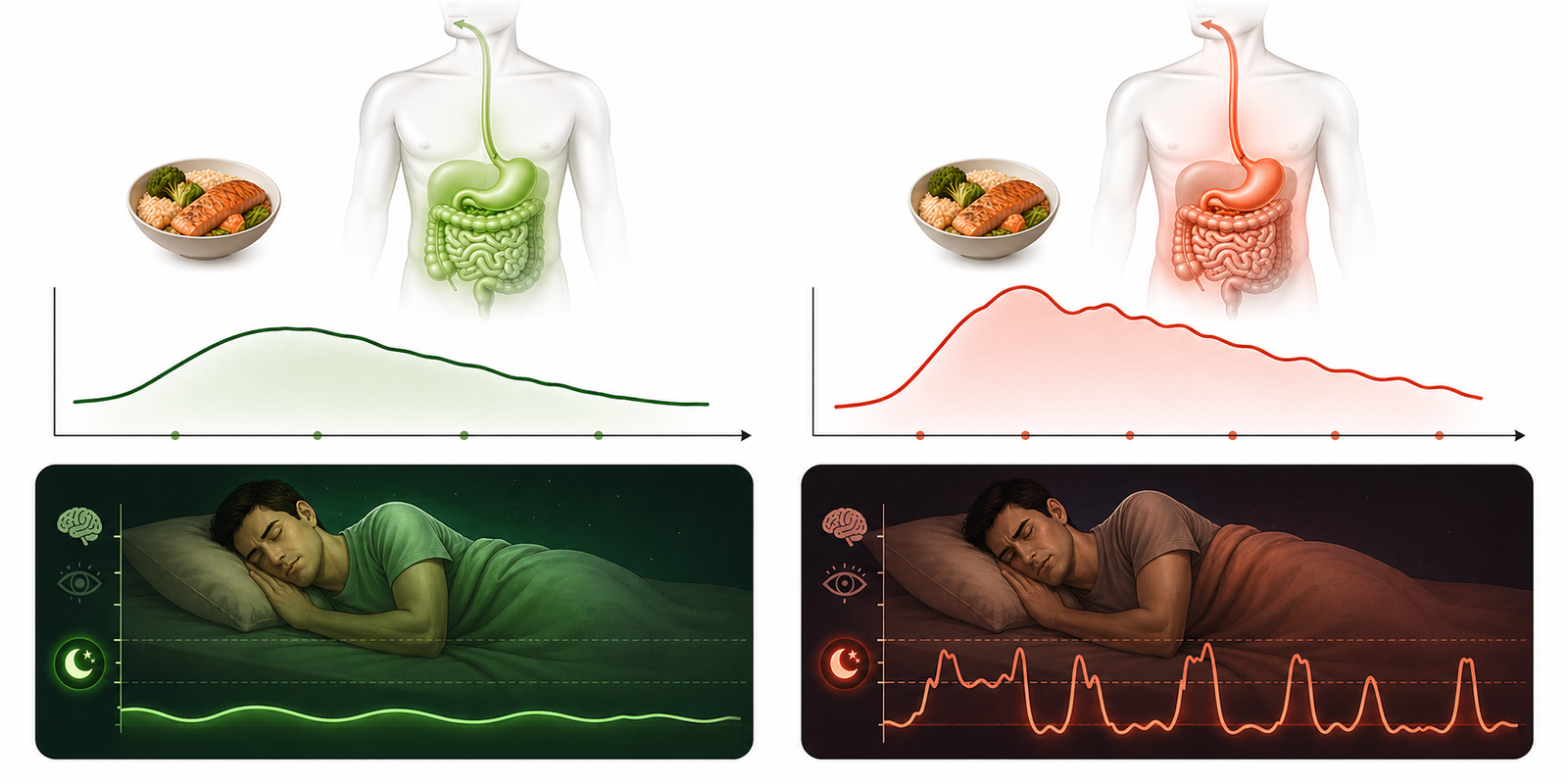
The most clinically immediate effect of poor sleep on insulin resistance is not the fasting glucose the next morning — it is what happens to glucose after the first meal of the day.
Under sleep restriction, the postprandial glucose curve becomes higher, longer, and less stable. The peak after the same meal is sharper. Glucose clearance is delayed. The return to baseline takes longer. Variability between meals increases. And meals that were previously well-tolerated — a moderate portion of oats, a piece of fruit, a bowl of rice — can produce exaggerated responses that the patient did not experience before.
The mechanism behind this shift operates through several pathways simultaneously. Sympathetic nervous system activation from the previous night’s poor sleep reduces GLUT4 translocation in skeletal muscle — the transporter that moves glucose from circulation into muscle cells following insulin stimulation. Elevated cortisol and catecholamines increase hepatic glucose output, adding to the circulating glucose load at the moment food is also arriving. Mitochondrial glucose oxidation is reduced, meaning cells are less efficient at processing the glucose that does enter. Incretin efficiency — the gut hormone response that amplifies insulin secretion in anticipation of food — is blunted under sleep restriction, reducing the speed and magnitude of the insulin response to a meal.
The combined effect is a body that receives the same glucose load from the same food but handles it with significantly reduced capacity. The glucose curve does not peak and resolve cleanly — it peaks higher, stays elevated longer, and produces a prolonged period of glucose exposure that drives oxidative stress, endothelial activation, and inflammatory signaling even in a patient who is eating entirely within their normal dietary pattern.
The clinical framing that makes this land for patients is direct: after poor sleep, the same meal behaves like a larger metabolic stressor. Interpreting postprandial glucose in isolation from the previous night’s sleep is clinically incomplete. The food is only one variable in the glucose equation.
What a CGM Reveals That Fasting Glucose Does Not
Fasting glucose is a single snapshot. It reflects the metabolic state at one moment — typically first thing in the morning, after an overnight fast. It is useful, but it is a blunt instrument for detecting the glucose dysregulation that poor sleep produces, because that dysregulation shows up most clearly not in the fasting state but in the overnight trace and the postprandial response.
Continuous glucose monitoring in poor sleepers reveals a pattern that fasting glucose entirely misses. During the night itself, instead of the stable flatline that characterizes healthy overnight glucose in a well-rested metabolically healthy individual, there are multiple small glucose rises corresponding to cortisol surges, sympathetic activation, micro-awakenings, and in patients with sleep-disordered breathing, hypoxia events. Glucose elevates around 2 to 5am — the window that corresponds to the cortisol and catecholamine activity of poor or fragmented sleep. The dawn phenomenon, which represents the pre-waking cortisol-driven rise in hepatic glucose output, is exaggerated. The overnight fasting period, which should be a metabolic rest and repair window, becomes a period of glucose instability.
The following day’s trace shows larger postprandial spikes, slower return to baseline, more variability between meals, stronger glucose responses to stress and caffeine, and reactive hunger and cravings that drive further eating and further glucose instability.
This CGM pattern frequently appears in patients whose fasting glucose and HbA1c still look acceptable on paper. Fasting glucose is sitting at 95 or 100 mg/dL — not flagged by any reference range. HbA1c is 5.4% — entirely normal. But the CGM is showing larger swings, prolonged postprandial excursions, overnight instability, and reduced time in range. The glucose variability is the early signal that the metabolic system is already under strain — and sleep is the variable driving it.
This is why glucose variability — measured as standard deviation, time in range, or the magnitude and frequency of excursions — is a more sensitive marker of sleep-driven metabolic disruption than either fasting glucose or HbA1c. Both of those markers average out the instability that variability directly quantifies. The HOMA-IR calculation captures the fasting insulin resistance picture, but it cannot capture the postprandial and overnight variability that CGM makes visible.
The visual impact of showing a patient their own CGM trace alongside their sleep quality data is one of the most powerful clinical communication tools available. When a patient sees their glucose behaving as if their body was under stress all night — the rises, the exaggerated morning peak, the prolonged postprandial curves — the connection between sleep and metabolic control becomes undeniable in a way that no verbal explanation fully replicates.
The Bidirectional Loop: How Poor Glucose Control Worsens Sleep
The relationship between poor sleep and glucose control is not unidirectional. It runs in both directions — and understanding this loop is essential for the patient who is trying to fix one without addressing the other.
When glucose remains elevated overnight — from a late high-carbohydrate meal, from the hepatic glucose overproduction of nighttime cortisol dysregulation, or from the insulin resistance that impairs glucose clearance — the elevated glucose itself disrupts sleep through several mechanisms. Osmotic fluid shifts from elevated blood glucose increase urinary frequency. Nocturia fragments sleep repeatedly through the night. Glucose variability activates sympathetic pathways — the drop from a postprandial peak produces a mild stress response that raises heart rate and reduces the depth of slow-wave sleep. Patients who eat a large high-carbohydrate meal in the evening and experience a reactive glucose pattern overnight often describe nighttime overheating, restlessness, early morning awakening, and the inability to return to deep sleep after waking.
The day following this pattern then shows worse insulin sensitivity, larger postprandial spikes, increased cravings, and elevated cortisol — all of which make the following evening’s glucose pattern more likely to be unstable again. The loop tightens.
As described in how poor sleep causes insulin resistance, even a single night of poor sleep reduces whole-body insulin sensitivity by 20 to 30 percent the following day. When that impaired insulin sensitivity is combined with the dietary choices that fatigue and cravings drive — larger portions, more refined carbohydrates, reward eating from exhaustion — the postprandial glucose load on an already glucose-intolerant system is compounded. Many patients in this loop improve only when both sides are addressed simultaneously: glucose stability and sleep restoration running as parallel interventions, not sequential ones.
The melatonin dimension of this loop is also clinically relevant. Elevated evening glucose suppresses melatonin onset — the body does not transition cleanly into its fasting and repair phase when glucose and insulin are still elevated. Delayed melatonin onset extends the biological day, delays sleep onset, reduces sleep depth, and perpetuates the overnight glucose instability that started the loop. Correcting evening meal timing is therefore not just a circadian intervention — it is a direct intervention on the glucose-sleep cycle.
Clinical Perspective: What I See in Practice
The patient in the poor-sleep glucose loop rarely presents with both problems clearly identified. They present with one — usually the metabolic problem — and the sleep component emerges only when the clinical questioning goes deeper.
The pattern I see consistently is a patient with glucose variability that is disproportionate to their dietary choices. They are eating reasonably well. Their fasting glucose is borderline — 100 to 110 mg/dL. Their HbA1c is acceptable. But their postprandial responses are unpredictable — a meal they handled well on Monday produces a larger response on Thursday with no obvious dietary explanation. Their energy is inconsistent. Cravings fluctuate without pattern. They describe being reactive to foods and stress in ways that vary day to day without any change in what they are eating.
When I ask about sleep, the picture clarifies immediately. The nights before the worst glucose days are consistently the nights of poorest sleep — fragmented, short, late, or disrupted by the 2 to 4am awakening pattern described in the nighttime cortisol post. The nights before the better glucose days correspond to earlier sleep onset, fewer awakenings, and more restorative rest.
This day-to-day variability in glucose response to the same food — with sleep quality as the differentiating variable — is one of the clearest clinical demonstrations of the sleep-glucose connection. Once patients see their own data organized this way, the motivation to prioritize sleep as a glucose management tool shifts immediately.
What I explain to patients in this situation is direct: your glucose is not just responding to what you eat — it is responding to how well you slept the night before. The food is constant. The metabolic context is not. And the metabolic context is largely determined by sleep.
The most consistent lab picture in these patients includes fasting insulin elevated above the functional optimal, triglycerides in the mildly elevated range, and a TG/HDL ratio above 2.0 despite appropriate dietary management. Where CGM data is available, standard deviation above 15 to 20 mg/dL and time in range below 80 to 85 percent in a non-diabetic patient are consistent findings. These are not dietary failures. They are glucose stability failures driven by sleep instability.
Glucose Variability: The Marker That Fasting Glucose Misses
Metabolic health is not just about average glucose — it is about stability. This distinction matters because fasting glucose and HbA1c both reflect average glucose status. Neither captures the amplitude, frequency, or duration of glucose excursions that define the metabolic experience of a poor sleeper.
Glucose variability — the degree to which glucose fluctuates above and below its mean across the day and night — is independently associated with oxidative stress, endothelial dysfunction, and inflammatory signaling. Repeated glucose excursions, even when the average remains acceptable, generate oxidative bursts at the cellular level that drive vascular damage, impair mitochondrial function, and activate inflammatory pathways. The harm is not in the average — it is in the swings.
Poor sleep increases every measurable component of glucose variability. It raises the standard deviation of glucose across the day. It increases the frequency and magnitude of postprandial excursions. It reduces time in range. It amplifies the overnight glucose instability that corresponds to cortisol and sympathetic activity during fragmented sleep. And it does all of this before fasting glucose becomes conventionally abnormal — making variability the earlier and more sensitive signal.
This is clinically important because it means the patient with normal fasting glucose and normal HbA1c is not necessarily metabolically protected. If their sleep is consistently poor and their glucose variability is elevated, the metabolic damage is accumulating at the cellular level while the standard screening panel continues to flag nothing. The circadian rhythm disruption that underlies most poor sleep patterns compounds this further — the peripheral clocks governing glucose metabolism lose their amplitude under chronic sleep disruption, and the timed precision of insulin sensitivity across the day becomes less defined, producing more variable and less predictable glucose responses independent of dietary composition.
Practical Dietary Strategy for the Day After Poor Sleep
After a night of poor sleep, the metabolic system is temporarily more insulin resistant and stress-reactive. The practical goal for the following day is to reduce metabolic volatility — not to compensate for lost sleep with aggressive dietary restriction, but to eat in a way that minimizes the glucose instability that impaired insulin sensitivity is already primed to produce.
The most protective strategy for a poor-sleep day begins with the first meal. Prioritizing protein early — eggs, meat, fish, Greek yogurt — blunts the postprandial glucose curve through multiple mechanisms: protein stimulates glucagon-like peptide 1, slows gastric emptying, and provides substrate for gluconeogenesis without contributing to glycemic load. Keeping carbohydrates lower or moderate after a bad night — and avoiding large refined-carbohydrate meals that would be manageable under rested conditions — prevents the exaggerated postprandial spikes that impaired insulin sensitivity is primed to produce.
The worst combination in clinical practice is sleep deprivation combined with ultra-processed carbohydrates and a sedentary day. This combination produces exaggerated glucose spikes, prolonged postprandial elevation, reactive cravings, and further cortisol activation — all of which compound the insulin resistance from the poor sleep and make the following night’s sleep more likely to be disrupted.
Post-meal walking is one of the most effective glucose management tools available on a poor-sleep day. Even ten minutes of walking after a meal significantly improves postprandial glucose clearance by activating insulin-independent glucose uptake in skeletal muscle through AMPK pathways. On a day when insulin sensitivity is reduced, this non-insulin-dependent glucose disposal pathway is proportionally more valuable.
Evening meal timing on a poor-sleep day deserves specific attention. Eating earlier in the evening — closing the last meal by 19:00 to 20:00 — prevents the overnight glucose elevation that would otherwise fragment the following night’s sleep and perpetuate the loop. Stabilizing meal timing throughout the day, hydrating adequately, and increasing electrolyte and mineral intake — both of which are depleted by the stress response of poor sleep — reduces the glucose volatility that fatigue-driven dietary choices would otherwise amplify.
The framing that shifts patient behavior on poor-sleep days is this: after poor sleep, your metabolism behaves more fragile — eat more predictably, not more emotionally. Fatigue drives reward eating. Reward eating on a glucose-intolerant day drives variability. Variability drives further sleep disruption. Breaking the loop requires eating with more deliberate structure on the days when the temptation to eat impulsively is highest.
Sleep Apnea as a Specific Driver of Overnight Glucose Instability
Among the causes of overnight glucose instability, sleep-disordered breathing deserves specific attention because its effects on overnight glucose are direct, measurable, and independent of diet and lifestyle.
As covered in detail in the sleep apnea post, each obstructive apnea event produces a cortisol and catecholamine surge that drives a transient rise in hepatic glucose output. In a patient with moderate to severe sleep apnea experiencing 30 events per hour, this means hundreds of micro glucose pulses across a single night. The CGM trace in an undiagnosed or untreated sleep apnea patient is recognizable: repeated small overnight glucose rises corresponding to apnea clusters, an exaggerated dawn phenomenon, and a morning fasting glucose that is higher than the diet would predict.
This overnight glucose instability from sleep apnea is not correctable through dietary management alone. The glucose rises are not from food — they are from the stress hormone response to airway obstruction. Treating the airway — through CPAP, positional therapy, or other appropriate means — is the intervention that removes the overnight glucose driver. In patients where sleep apnea is suspected based on the clinical picture and the CGM pattern, formal sleep testing is the appropriate next step before attributing the overnight glucose instability to diet or lifestyle.
The CGM glucose signature described here is one window into a larger system, and the full clinical framework that integrates sleep duration, circadian timing, hormonal rhythm, and the patient profile is laid out in the cornerstone post on sleep, the body clock, and metabolism.
A Note on Uncertainty
The relationship between sleep restriction and impaired postprandial glucose control is among the most consistently replicated findings in sleep and metabolic research. Controlled studies of acute sleep restriction show measurable increases in postprandial glucose and insulin in healthy subjects within one to two nights of restricted sleep. The mechanistic pathways — sympathetic activation, cortisol elevation, GLUT4 suppression, incretin blunting — are well-characterized.
The clinical use of CGM data to demonstrate sleep-glucose relationships in non-diabetic patients is an emerging area. The specific thresholds for glucose variability metrics — standard deviation, time in range, coefficient of variation — that define metabolic risk in non-diabetic populations are not yet standardized across guidelines. The interpretations presented in this post reflect functional medicine practice patterns and emerging research rather than established diagnostic criteria.
The dietary strategies for poor-sleep days described here are grounded in the established mechanisms of postprandial glucose management and are consistent with low-risk nutritional principles. They are practical recommendations, not clinical prescriptions. Patients with diabetes, those on glucose-lowering medications, or those using CGM devices should interpret their data and make dietary adjustments in collaboration with their treating physician.
People Also Ask
How does poor sleep affect blood sugar levels?
Poor sleep impairs insulin sensitivity through sympathetic nervous system activation, elevated cortisol, suppressed GLUT4 translocation in skeletal muscle, and reduced incretin efficiency. The combined effect is a higher postprandial glucose peak after the same meal, slower glucose clearance, and prolonged glucose elevation throughout the following day. Even a single night of poor sleep produces measurable deterioration in glucose handling within 24 hours.
What does a CGM show in people who sleep poorly?
Continuous glucose monitoring in poor sleepers typically shows multiple small overnight glucose rises corresponding to cortisol surges and micro-awakenings, an exaggerated dawn phenomenon, loss of the stable overnight flatline, larger postprandial spikes the following day, slower return to baseline, and reduced time in range. This pattern often appears before fasting glucose or HbA1c become abnormal — making CGM a more sensitive early indicator of sleep-driven glucose dysregulation.
Can poor sleep cause high fasting glucose in the morning?
Yes. Poor sleep elevates overnight cortisol and activates hepatic gluconeogenesis — the liver produces more glucose during the sleep period than it should. The result is a higher fasting glucose reading the next morning that is not driven by the previous day’s diet but by the overnight hormonal response to insufficient or fragmented sleep. This is the physiological basis of the exaggerated dawn phenomenon seen in poor sleepers.
Is glucose variability more important than fasting glucose?
For detecting the early metabolic effects of poor sleep, glucose variability is a more sensitive marker than fasting glucose. Fasting glucose reflects a single point in time and averages out the instability that poor sleep produces. Glucose variability — measured through standard deviation, time in range, and the magnitude of postprandial excursions — captures the full picture of glucose instability across the day and night, including the patterns that poor sleep specifically generates.
What should you eat after a bad night of sleep?
After poor sleep, insulin sensitivity is temporarily reduced and the metabolic system is more stress-reactive. The most protective dietary approach prioritizes protein early in the day — eggs, meat, fish, Greek yogurt — keeps carbohydrates lower or moderate, avoids large refined-carbohydrate meals, stabilizes meal timing, and closes the last meal by early evening. Post-meal walking significantly improves postprandial glucose clearance on days when insulin sensitivity is reduced. The goal is metabolic predictability rather than restriction.
What is the connection between sleep apnea and overnight blood sugar?
Obstructive sleep apnea produces repeated cortisol and catecholamine surges from each apnea event, each of which drives a transient rise in hepatic glucose output. In moderate to severe sleep apnea, this produces hundreds of micro glucose pulses overnight — resulting in a CGM pattern of repeated overnight glucose rises, an exaggerated dawn phenomenon, and a morning fasting glucose that is higher than the diet would predict. This overnight glucose instability is driven by the airway obstruction, not by food, and requires airway treatment rather than dietary adjustment.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
- Donga E, van Dijk M, van Dijk JG, et al. A single night of partial sleep deprivation induces insulin resistance in multiple metabolic pathways in healthy subjects. Journal of Clinical Endocrinology & Metabolism. 2010;95(6):2963–2968. 🔗 https://pubmed.ncbi.nlm.nih.gov/20371664/
- Spiegel K, Leproult R, Van Cauter E. Impact of sleep debt on metabolic and endocrine function. The Lancet. 1999;354(9188):1435–1439. 🔗 https://pubmed.ncbi.nlm.nih.gov/10543671/
- Buxton OM, Pavlova M, Reid EW, et al. Sleep restriction for 1 week reduces insulin sensitivity in healthy men. Diabetes. 2010;59(9):2126–2133. 🔗 https://pubmed.ncbi.nlm.nih.gov/20585000/
- Leproult R, Van Cauter E. Role of sleep and sleep loss in hormonal release and metabolism. Endocrine Development. 2010;17:11–21. 🔗 https://pubmed.ncbi.nlm.nih.gov/19955752/
- Van Cauter E, Spiegel K, Tasali E, Leproult R. Metabolic consequences of sleep and sleep loss. Sleep Medicine. 2008;9(Suppl 1):S23–S28. 🔗 https://pubmed.ncbi.nlm.nih.gov/18929315/
- Broussard JL, Ehrmann DA, Van Cauter E, Tasali E, Brady MJ. Impaired insulin signaling in human adipocytes after experimental sleep restriction. Annals of Internal Medicine. 2012;157(8):549–557. 🔗 https://pubmed.ncbi.nlm.nih.gov/23070488/
- Ceriello A, Esposito K, Piconi L, et al. Oscillating glucose is more deleterious to endothelial function and oxidative stress than mean glucose in normal and type 2 diabetic patients. Diabetes. 2008;57(5):1349–1354. 🔗 https://pubmed.ncbi.nlm.nih.gov/18299315/
- Monnier L, Mas E, Ginet C, et al. Activation of oxidative stress by acute glucose fluctuations compared with sustained chronic hyperglycemia in patients with type 2 diabetes. JAMA. 2006;295(14):1681–1687. 🔗 https://pubmed.ncbi.nlm.nih.gov/16609090/
- Tasali E, Mokhlesi B, Van Cauter E. Obstructive sleep apnea and type 2 diabetes: interacting epidemics. Chest. 2008;133(2):496–506. 🔗 https://pubmed.ncbi.nlm.nih.gov/18252913/
- Scheer FA, Hilton MF, Mantzoros CS, Shea SA. Adverse metabolic and cardiovascular consequences of circadian misalignment. PNAS. 2009;106(11):4453–4458. 🔗 https://pubmed.ncbi.nlm.nih.gov/19255424/
- Tremblay MS, Colley RC, Saunders TJ, Healy GN, Owen N. Physiological and health implications of a sedentary lifestyle. Applied Physiology, Nutrition, and Metabolism. 2010;35(6):725–740. 🔗 https://pubmed.ncbi.nlm.nih.gov/21164543/
- Knutson KL, Spiegel K, Penev P, Van Cauter E. The metabolic consequences of sleep deprivation. Sleep Medicine Reviews. 2007;11(3):163–178. 🔗 https://pubmed.ncbi.nlm.nih.gov/17442599/