Why treating estrogen while ignoring insulin is failing millions of women — and what the evidence actually says.
Most women who arrive with estrogen dominance symptoms have already been through the standard clinical route. They have had their estradiol checked — often at a random point in their cycle — been told their levels are normal, and been offered either the pill, a hormonal IUD, or an SSRI for the mood component. Some have been told it is simply perimenopause. Some have been told it is stress. Almost none have had their fasting insulin measured. Almost none have had their SHBG interpreted in this context. And the metabolic environment producing their symptoms has continued, unaddressed, for years.
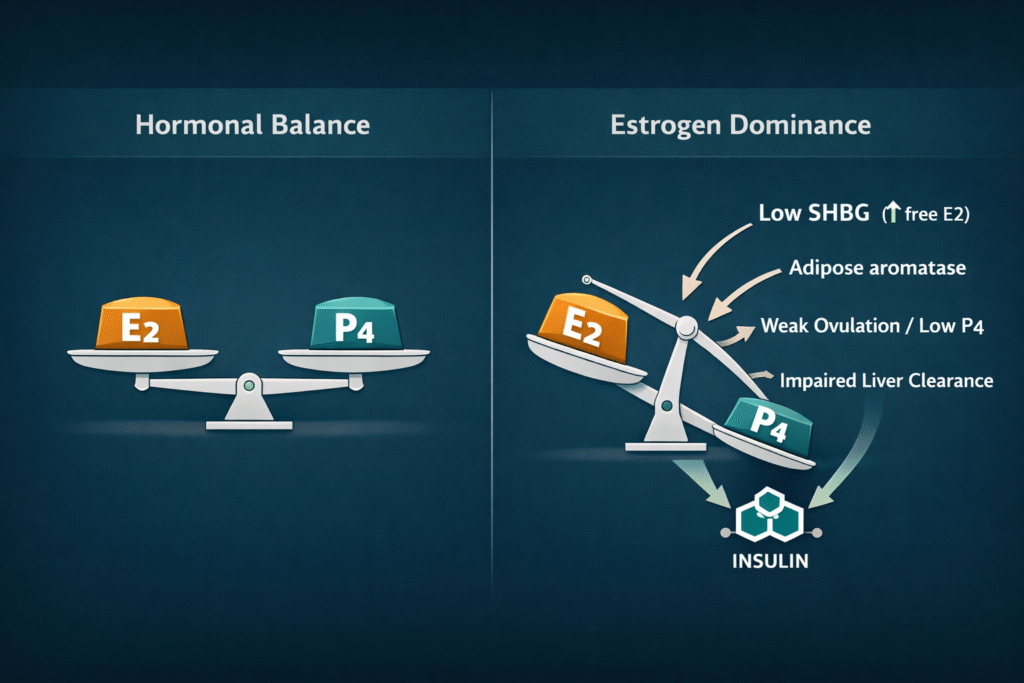
Estrogen dominance is one of the most common hormonal complaints in clinical practice and one of the most consistently misframed. The conventional approach treats it as a problem of too much estrogen — a hormone story with a hormone solution. The metabolic evidence tells a more precise and more actionable story: in the majority of women presenting with this symptom cluster, the issue is not simply estrogen excess. It is a combination of
- relative progesterone insufficiency
- reduced estrogen clearance driven by impaired liver and gut function
- and — most critically — the downstream hormonal consequences of hyperinsulinemia and insulin resistance acting on the same biological systems that govern estrogen activity.
Correct the metabolic environment, and the hormonal picture changes. In clinical practice, it changes with a predictability that the hormone-only treatment approach rarely achieves.
What you will learn:
What estrogen dominance actually means physiologically — and why the lab number alone misses it | The four metabolic mechanisms driving the symptom cluster | How insulin resistance raises free estrogen activity without changing total estradiol | The role of the liver and gut in estrogen clearance — and where that system breaks down | What changes when the metabolic root is addressed, and on what timeline
The Conventional Story — and Where It Falls Short
Estrogen dominance as a clinical concept describes a state in which estrogen activity is excessive relative to progesterone opposition — not necessarily because estradiol is elevated in absolute terms, but because the hormonal balance between the two has shifted in estrogen’s favour. This distinction matters enormously, and it is almost universally missed in standard clinical assessment.
The conventional workup, when it happens at all, typically measures serum estradiol — often on a random cycle day without reference to the luteal phase — and reports it as normal. What it does not assess is progesterone status at the appropriate luteal phase window, the estradiol-to-progesterone ratio that determines net hormonal effect, sex hormone-binding globulin levels that govern how much of that estradiol is biologically active, or the metabolic drivers that are shifting that ratio in the first place.
Treatments then follow from the incomplete assessment: oral contraceptives to suppress the cycle and reduce bleeding, progesterone cream based on a single saliva result, SSRIs for mood symptoms that are actually rooted in hormonal and metabolic disruption, and in some cases nothing at all — because the labs look normal and the symptoms are attributed to stress or aging. The upstream metabolic driver continues operating. The symptoms return the moment any symptomatic intervention is withdrawn.
Treating estrogen dominance without addressing insulin resistance is treating the alarm without investigating the fire.
Clinical Perspective: What I See in Practice
When a woman arrives with the classic estrogen dominance picture — weight accumulating around the hips and thighs, breast tenderness in the week before her period, mood instability that tracks the luteal phase, heavy or clotty bleeding, fatigue, and low libido — the first thing I do is look at the full hormonal and metabolic context, not just the estradiol number.
What I see consistently is this: estradiol is often normal to high-normal. Progesterone is low — particularly in the luteal phase — and the estradiol-to-progesterone ratio is shifted toward estrogen effect. This is not a story of dramatic hormone excess. It is a story of relative imbalance: enough estrogen activity, and insufficient progesterone opposition to balance it. The luteal progesterone deficit often traces back to anovulatory cycles, weak ovulation, chronic stress suppressing the HPG axis, or PCOS-spectrum patterns where ovulation is irregular and unreliable.
But what stands out metabolically — and what almost no conventional workup captures — is the insulin picture. Fasting insulin is elevated. HOMA-IR is elevated. Fasting glucose may still look entirely normal. Triglycerides are creeping upward, HDL is trending down, and the TG/HDL ratio is beginning to shift. The woman in front of me has been told her labs are fine.
What the labs are not showing is that chronically elevated insulin has suppressed her hepatic SHBG production. Her total estradiol may be within range, but her SHBG is low — and low SHBG means more free, biologically active estradiol at the tissue level. This is one of the most important and most consistently missed mechanisms in this presentation: you can have normal total estrogen and low SHBG and still be experiencing the full clinical picture of estrogen dominance, because what matters is not the total amount but how much is unbound and active. Fasting insulin and SHBG together are the pair I reach for first.
I also see, consistently: ferritin depletion from heavy menstrual bleeding — iron lost month after month without replacement, presenting as fatigue that gets attributed to stress rather than investigated. I see signs of impaired liver clearance in women with fatty liver or elevated GGT and ALT. I see gut history: constipation, bloating, a pattern of dysbiosis that fits elevated beta-glucuronidase activity reactivating estrogen in the gut. And I see the cortisol picture — chronic stress suppressing ovulation, weakening the luteal phase, reducing progesterone, and compounding every hormonal imbalance already in motion.
None of these systems are being interrogated in the standard GP or gynaecology consultation. The symptoms are real, the biology is coherent, and the upstream drivers are identifiable and addressable.
How Insulin Resistance Drives Estrogen Dominance: The Four Mechanisms
The metabolic pathway from hyperinsulinemia to estrogen dominance symptoms is not a single channel. It converges through four mechanisms, each operating independently, each downstream of the same upstream driver: chronically elevated insulin produced by progressive insulin resistance in liver, muscle, and adipose tissue.
Mechanism 1: SHBG Suppression and Rising Free Estrogen Activity
Sex hormone-binding globulin is produced by the liver, and its synthesis is directly inhibited by insulin. When insulin is chronically elevated, hepatic SHBG production falls. Lower SHBG means a higher proportion of circulating estradiol is free and biologically active — available to bind estrogen receptors in breast tissue, the uterus, the brain, and adipose tissue.
The total estradiol number on the lab report does not change. The free fraction driving symptoms rises significantly. This mechanism explains why a woman can present with a full estrogen dominance picture while her total estradiol sits comfortably within the reference range. It also explains why the conventional lab interpretation — estradiol normal, therefore not estrogen dominance — is insufficient without SHBG in the picture.
Mechanism 2: Adipose Aromatase and Estrogen Amplification
Adipose tissue — particularly visceral and peripheral fat — contains the enzyme aromatase, which converts androgens into estrogens. In the presence of hyperinsulinemia, ovarian androgen production is increased via theca cell stimulation, providing more androgen substrate for peripheral aromatization. The result is a dual amplification: more androgens being produced upstream, more of those androgens being converted to estrogens in fat tissue. Women with central or peripheral adiposity are running a continuous low-grade estrogen-generating system in their fat cells — one that is directly fuelled by the same hyperinsulinemia driving their insulin resistance.
Mechanism 3: Anovulation and Progesterone Deficit
Hyperinsulinemia disrupts the hypothalamic-pituitary-gonadal axis and interferes with normal follicular development and ovulation. As described in the PCOS post, insulin excess causes follicles to respond to LH prematurely, arresting development before a dominant follicle can complete the maturation required for ovulation. When ovulation is absent or weak, the corpus luteum — the structure that produces progesterone in the luteal phase — either does not form or functions inadequately.
Progesterone falls. The estrogen-to-progesterone ratio shifts. The breast tenderness, the mood instability, the heavy bleeding, the fluid retention in the luteal phase — all of these are the downstream consequences of insufficient progesterone opposition to whatever estrogen activity remains. This is why the estradiol-to-progesterone ratio, measured at the appropriate luteal phase timing, tells a story that total estradiol alone cannot.
Mechanism 4: Impaired Liver Clearance and Gut Recirculation
The liver is the primary site of estrogen metabolism. Through phase I and phase II detoxification pathways — particularly glucuronidation and sulfation — the liver converts active estrogens into water-soluble conjugates that are then excreted via bile into the gut and via urine. When liver function is compromised — by insulin resistance and fatty liver, chronic inflammation, impaired methylation, or sluggish bile flow — this clearance pathway slows. Estrogen that should be eliminated is instead recirculated. The clinical picture is not one of excess estrogen production. It is one of reduced estrogen elimination — a distinction that entirely changes the therapeutic approach.
The gut adds a second layer of complexity through the estrobolome — the collection of gut bacteria that produce beta-glucuronidase, an enzyme that deconjugates processed estrogens in the gut, removing the glucuronic acid tag that marked them for elimination and allowing them to become active again and be reabsorbed through enterohepatic circulation. Dysbiosis with elevated beta-glucuronidase activity can significantly increase estrogen recirculation regardless of how well the liver processed it initially.
Constipation compounds this further: slower bowel transit means more time for reabsorption of conjugated estrogens before they can be excreted, even without severe dysbiosis. Gut barrier dysfunction — increased intestinal permeability — operates as an inflammatory amplifier rather than a direct estrogen driver, raising LPS exposure, systemic inflammation, insulin resistance, and cortisol dysregulation, all of which worsen aromatase activity and ovulatory quality downstream.
The Marker Cluster That Identifies the Metabolic Driver
Standard workups for estrogen dominance symptoms — when they occur at all — typically include a single estradiol measurement and perhaps a thyroid screen. The metabolic and hormonal context that would identify the upstream driver is almost never assessed.
The combination that clarifies the picture is:
fasting insulin, HOMA-IR, and SHBG as the core trio. Fasting insulin above 10 µIU/mL — and particularly above the functional optimal of 5 µIU/mL — combined with low SHBG, establishes the hyperinsulinemia-driven free estrogen mechanism directly. The TG/HDL ratio above 2.0 confirms hepatic insulin resistance and provides an accessible proxy where fasting insulin has not been ordered. Luteal phase progesterone — measured at day 21 in a 28-day cycle, or 7 days before the anticipated period in irregular cycles — is essential for establishing the estradiol-to-progesterone ratio that determines net hormonal effect.
Beyond this core cluster: ALT, AST, and GGT assess liver metabolic burden and can flag early fatty liver-associated clearance impairment even before overt NAFLD is present. Ferritin is essential — heavy periods deplete iron month after month, and the resulting low ferritin manifests as the fatigue and low energy that gets attributed to the hormonal picture but is actually a downstream consequence of it. hsCRP above 1.0 mg/L reflects the chronic inflammatory environment that amplifies both aromatase activity and insulin resistance. Cortisol patterns and thyroid function — particularly TSH, free T3, and thyroid antibodies — complete the picture, since hypothyroidism and Hashimoto’s frequently present alongside estrogen dominance symptoms and share insulin resistance as a common driver.
This is not a complex or expensive panel. It is the standard markers, interpreted through a systems lens, at the functional medicine thresholds rather than the conventional reference ranges that were derived from population averages rather than optimal health.
How elevated TBG from estrogen excess reduces free thyroid hormone availability — and the five functional patterns this produces — is covered in full in the thyroid metabolism framework.
The Problem With Treating Estrogen Without Addressing Metabolism
The most common management approach — oral contraceptives to regulate the cycle and reduce bleeding, progesterone supplementation based on incomplete testing, or SSRIs for mood symptoms — produces symptomatic relief that is real but incomplete. It addresses the downstream manifestation while leaving the metabolic driver in place.
Oral contraceptives suppress ovarian function and reduce endogenous estrogen and progesterone production. They can significantly reduce heavy bleeding and breast tenderness. But they do not lower fasting insulin, improve SHBG, reduce visceral fat aromatase activity, restore hepatic estrogen clearance capacity, or address the gut dysbiosis amplifying estrogen recirculation. When contraception is discontinued — for pregnancy, for personal preference, or for side effects — the underlying metabolic environment is unchanged, and the symptom picture returns.
Progesterone supplementation, similarly, can restore the estrogen-to-progesterone ratio directly. It is a legitimate intervention and often clinically useful as a bridge. But progesterone given to a woman with ongoing hyperinsulinemia, low SHBG, impaired liver clearance, and elevated beta-glucuronidase activity is correcting one variable in a system that is still producing the imbalance from multiple directions. The result is ongoing dependence on supplementation rather than genuine restoration of the system producing the imbalance.
The metabolic intervention does not replace these approaches where they are indicated. It creates the biological conditions in which the hormonal system can restore its own balance — and in which any hormonal support that remains necessary is working with a functional foundation rather than against a dysfunctional one.
The upstream stress mechanism producing luteal phase weakness and progesterone decline is mapped in detail in how chronic stress rewires the hormonal hierarchy.
The Intervention: What Addresses the Metabolic Root
The intervention that addresses estrogen dominance at its metabolic root is not designed to directly modulate estrogen. It is designed to correct the upstream conditions producing the imbalance.
Reduce the insulin load. Eliminating ultra-processed foods, refined carbohydrates, HFCS, and industrial seed oils removes the primary substrate driving chronic insulin elevation. This is the single intervention that simultaneously addresses SHBG suppression, ovarian androgen excess, adipose aromatase stimulation, and the inflammatory environment impairing liver clearance. Approximately 50 grams of carbohydrate per day from whole food sources is an appropriate target for women with active insulin resistance. Irregular intermittent fasting — not rigid daily same-time fasting, which causes metabolic adaptation — creates the between-meal insulin valleys that allow fasting insulin to fall toward the functional optimal below 5 µIU/mL.
Prioritize animal-based protein at adequate intake. Protein at 1.6g per kg of ideal body weight supports muscle mass — the primary peripheral glucose sink — and provides the amino acid substrates, particularly methionine and glycine, required for hepatic phase II detoxification pathways. Eggs, specifically, provide choline — essential for hepatic phosphatidylcholine synthesis and VLDL export — which directly supports liver function and estrogen clearance capacity. The woman who has been avoiding eggs and animal fat out of cholesterol concern may be undermining the very organ system responsible for eliminating her excess estrogen.
Support liver clearance pathways. Beyond dietary quality, adequate B vitamins — particularly B6, B12, and folate — support methylation, one of the phase II pathways through which estrogen is processed for elimination. Magnesium supports glucuronidation. Cruciferous vegetables provide DIM (diindolylmethane) and indole-3-carbinol, which shift estrogen metabolism toward less active hydroxylation pathways. Reducing alcohol intake removes a direct competitive burden on hepatic phase I enzyme capacity. The liver’s role in estrogen metabolism — and how gut-liver axis dysfunction impairs clearance — is examined in the gut-liver connection and its metabolic consequences.
Restore gut transit and address dysbiosis. Adequate dietary fibre — from whole food sources rather than supplements where possible — supports bowel regularity and reduces the transit time available for estrogen reabsorption. Fermented foods and, where clinically indicated, targeted probiotic support can address the dysbiosis driving elevated beta-glucuronidase activity. Gut barrier integrity is supported by the same dietary foundations that reduce insulin resistance: eliminating ultra-processed emulsifiers and industrial seed oils, restoring adequate protein, and addressing sleep and cortisol.
Optimize vitamin D and address cortisol. Vitamin D receptor signalling modulates estrogen receptor activity and supports immune regulation in tissues with high estrogen sensitivity. Target 50–70 ng/mL with D3 and K2. Chronic cortisol elevation — from sleep disruption, psychological stress, or circadian misalignment — directly suppresses GnRH pulsatility, impairs ovulation, weakens the luteal phase, and reduces progesterone production. Sleep optimization and circadian alignment are not peripheral lifestyle recommendations. They are mechanistically relevant interventions for the progesterone deficit that sits at the centre of the estrogen dominance picture.
What Changes When the Metabolic Root Is Addressed — And When
In clinical practice, when the metabolic drivers are genuinely addressed, the estrogen dominance picture improves with a predictability that the symptomatic treatment approach rarely achieves.
The earliest shifts appear within two to six weeks: lower fasting insulin, reduced water retention and bloating, less breast tenderness in the premenstrual window, fewer and less severe mood swings. These are the direct downstream consequences of falling insulin — SHBG begins to rise, free estrogen activity begins to fall, and the inflammatory tone that was amplifying every symptom starts to reduce.
Between six and twelve weeks, the deeper hormonal picture begins to move: periods become lighter and less clotty, cycle timing becomes more predictable, luteal progesterone improves as ovulation normalises, libido begins to recover. The hip and thigh fat that is so characteristic of estrogen-dominant body composition begins to redistribute more slowly — this is the last to change, because it reflects months of accumulated adipose-driven aromatase activity unwinding. A five to ten percent reduction in body weight in women with insulin resistance-driven symptoms is often enough to significantly improve ovulatory function and restore a more favourable hormonal balance. Not dramatic weight loss. A metabolic shift that changes what the body’s fat tissue is doing hormonally.
These are not anecdotal observations disconnected from mechanism. They are the expected physiological consequences of removing the primary metabolic stressor — and they confirm, in real clinical time, that the hormonal imbalance was downstream of the metabolic one.
A Note on Uncertainty
The mechanisms described in this post — SHBG suppression by insulin, adipose aromatase amplification, anovulatory progesterone deficit, and impaired hepatic and gut estrogen clearance — are individually well-established in the research literature. What is less clearly quantified is the relative contribution of each mechanism in any given patient, and the proportion of women with estrogen dominance symptoms in whom metabolic dysfunction is the dominant rather than a contributing driver.
In women with primary ovarian insufficiency, genetic aromatase excess syndrome, or structural uterine pathology, the mechanisms described above may be compounding factors rather than primary causes. Clinical assessment should always include gynaecological evaluation alongside metabolic assessment, and medication decisions — including hormonal contraception, progesterone supplementation, and any other interventions — should be made in collaboration with the prescribing physician. The functional medicine protocol does not replace this. It creates the metabolic foundation on which any necessary hormonal support can work more effectively.
Practical Implications
If you have been told your estrogen levels are normal but you are still experiencing breast tenderness, heavy periods, mood instability in the luteal phase, weight gain around the hips and thighs, and low libido — and if your fasting insulin has never been measured, your SHBG has never been interpreted in this context, and your estradiol-to-progesterone ratio has never been assessed at the appropriate luteal phase timing — then the metabolic environment producing your symptoms has never been properly evaluated.
The practical starting point is measuring fasting insulin and SHBG alongside a luteal phase progesterone. If fasting insulin is above 10 µIU/mL and SHBG is low, the mechanism is identifiable. If the TG/HDL ratio is above 2.0 and ALT is trending above 25 U/L, the liver clearance pathway is under metabolic stress. These are not exotic tests. They are the standard panel, read through the right lens.
The hormonal symptoms you are experiencing are downstream outputs. They are measuring the tissue-level consequences of a metabolic signal that your standard panel has been ignoring. Correct the signal — and the downstream outputs move.
Estrogen dominance rarely exists in isolation — it sits within a larger pattern of hormonal imbalance driven by metabolic dysfunction, where insulin resistance, fatty liver, gut health, and stress physiology together shape the female hormonal environment.
People Also Ask
What is estrogen dominance?
Estrogen dominance describes a state in which estrogen activity is excessive relative to progesterone opposition — not necessarily because estradiol is elevated in absolute terms, but because the hormonal balance between the two has shifted. This can occur when estradiol is genuinely elevated, when progesterone is insufficient (as in anovulatory cycles or weak luteal phases), when SHBG is low (increasing free biologically active estrogen), or when estrogen clearance is impaired by liver or gut dysfunction. The total estradiol number alone is an insufficient diagnostic criterion.
Can insulin resistance cause estrogen dominance?
Yes — through multiple converging mechanisms. Chronically elevated insulin suppresses hepatic SHBG production, increasing free estrogen activity. It stimulates ovarian androgen production, providing more substrate for peripheral aromatization in fat tissue. It disrupts ovulation, reducing luteal progesterone and shifting the estradiol-to-progesterone ratio. And it promotes the inflammatory and liver metabolic dysfunction that impairs estrogen clearance. In women with estrogen dominance symptoms, fasting insulin and SHBG are among the most informative markers to assess.
Why do I have estrogen dominance symptoms if my estrogen levels are normal?
Because total estradiol is not the same as free estradiol activity. When SHBG is low — which occurs with hyperinsulinemia — a higher proportion of total estradiol is unbound and biologically active at tissue level. Additionally, estrogen dominance symptoms can reflect a progesterone deficit rather than absolute estrogen excess: if progesterone is low due to anovulation or weak ovulation, even normal estrogen levels create a relative imbalance. The ratio matters more than the absolute number.
What is the role of the liver in estrogen dominance?
The liver processes estrogen through phase I and phase II detoxification pathways, converting active estrogens into water-soluble conjugates for excretion. When liver function is impaired — by insulin resistance and fatty liver, chronic inflammation, impaired methylation, or sluggish bile flow — estrogen clearance slows and estrogen is recirculated rather than eliminated. This means estrogen dominance can reflect reduced clearance rather than excess production — a distinction that changes the entire therapeutic approach.
How does gut health affect estrogen levels?
Gut bacteria produce an enzyme called beta-glucuronidase, which can deconjugate estrogen that the liver has processed for elimination, making it active again and allowing reabsorption through enterohepatic circulation. Dysbiosis with elevated beta-glucuronidase activity — as well as constipation increasing transit time — can significantly increase estrogen recirculation. Supporting bowel regularity and gut microbiome diversity is a meaningful component of addressing estrogen dominance from the metabolic root.
How long does it take for estrogen dominance symptoms to improve when insulin resistance is addressed?
In clinical practice, the earliest improvements — reduced breast tenderness, less water retention, improved mood stability — typically appear within two to six weeks of meaningfully reducing the insulin load. Over six to twelve weeks, periods become lighter and more regular, luteal progesterone often improves as ovulation normalizes, and libido begins to recover. A five to ten percent reduction in body weight in women with insulin resistance is often sufficient to significantly improve ovulatory function and hormonal balance. The adipose-driven body composition changes take longest — reflecting the gradual unwinding of months of fat-tissue estrogen amplification.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Plymate SR, Matej LA, Jones RE, Friedl KE. Inhibition of sex hormone-binding globulin production in the human hepatoma (Hep G2) cell line by insulin and prolactin. Journal of Clinical Endocrinology & Metabolism. 1988;67(3):460–464. 🔗 https://pubmed.ncbi.nlm.nih.gov/3403065/
2. Nestler JE, Powers LP, Matt DW, et al. A direct effect of hyperinsulinemia on serum sex hormone–binding globulin levels in obese women with the polycystic ovary syndrome. Journal of Clinical Endocrinology & Metabolism. 1991;72(1):83–89. 🔗 https://pubmed.ncbi.nlm.nih.gov/1986026/
3. Simpson ER. Sources of estrogen and their importance. Journal of Steroid Biochemistry and Molecular Biology. 2003;86(3–5):225–230. 🔗 https://pubmed.ncbi.nlm.nih.gov/14623515/
4. Bulun SE, Chen D, Moy I, Brooks DC, Zhao H. Aromatase, breast cancer and obesity: a complex interaction. Trends in Endocrinology & Metabolism. 2012;23(2):83–89. 🔗 https://pubmed.ncbi.nlm.nih.gov/22169755/
5. Plottel CS, Blaser MJ. Microbiome and malignancy. Cell Host & Microbe. 2011;10(4):324–335. 🔗 https://pubmed.ncbi.nlm.nih.gov/22018234/
6. Baker JM, Al-Nakkash L, Herbst-Kralovetz MM. Estrogen–gut microbiome axis: physiological and clinical implications. Maturitas. 2017;103:45–53. 🔗 https://pubmed.ncbi.nlm.nih.gov/28778332/
7. Dunaif A. Insulin resistance and the polycystic ovary syndrome: mechanism and implications for pathogenesis. Endocrine Reviews. 1997;18(6):774–800. 🔗 https://pubmed.ncbi.nlm.nih.gov/9408743/
8. Selva DM, Hogeveen KN, Innis SM, Hammond GL. Monosaccharide-induced lipogenesis regulates the human hepatic sex hormone–binding globulin gene. Journal of Clinical Investigation. 2007;117(12):3979–3987. 🔗 https://pubmed.ncbi.nlm.nih.gov/18060042/
9. Michnovicz JJ, Bradlow HL. Altered estrogen metabolism and excretion in humans following consumption of indole-3-carbinol. Nutrition and Cancer. 1991;16(1):59–66. 🔗 https://pubmed.ncbi.nlm.nih.gov/1656396/
10. Reed BG, Carr BR. The Normal Menstrual Cycle and the Control of Ovulation. In: Feingold KR, et al., eds. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2018. 🔗 https://www.ncbi.nlm.nih.gov/books/NBK279054/
