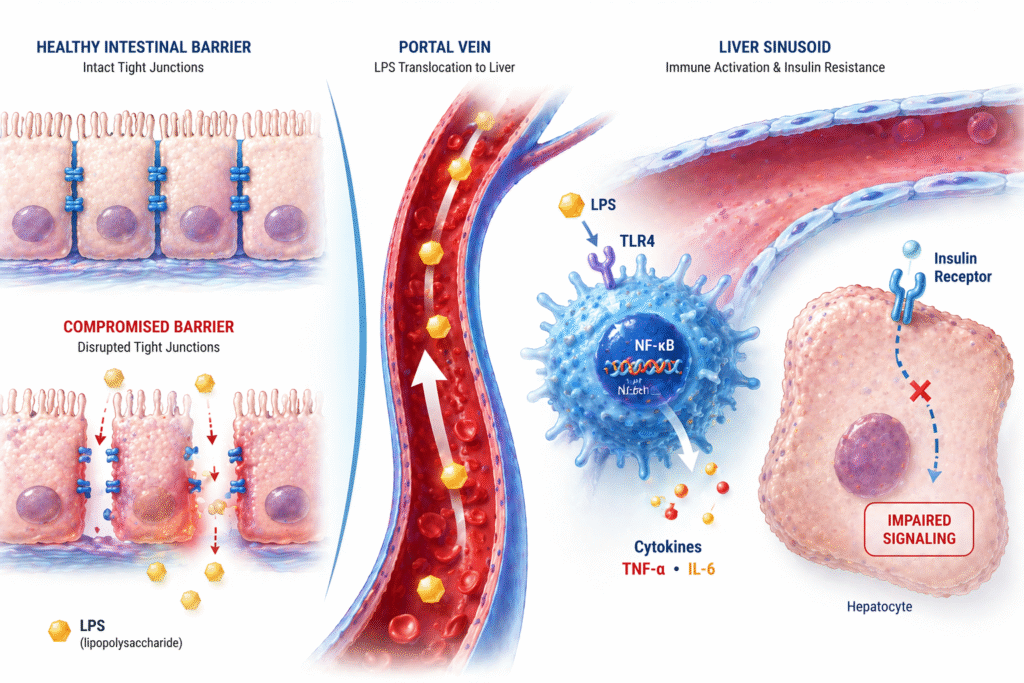
Leaky gut and insulin resistance are more directly connected than most patients — and most GPs — have ever been told. The gut wall is not just a digestive barrier. It is a metabolic gatekeeper. When it fails, the liver pays first.
There is a biological conversation happening continuously between your gut and your liver — one that standard medicine almost never discusses, that most patients have never heard of, and that is, in a significant proportion of cases, one of the primary upstream drivers of the insulin resistance and fatty liver that bring people to a functional medicine assessment after years of conventional management has failed to produce meaningful change.
The gut and the liver are anatomically and physiologically inseparable. Everything the intestine absorbs — every nutrient, every bacterial metabolite, every fragment of microbial cell wall — travels through the portal vein to the liver before it reaches anywhere else in the body. The liver is not a downstream organ in this relationship. It is the first organ. It receives the output of the gut directly, continuously, and at high concentration. When the gut is healthy — when the microbiome is diverse and balanced, the intestinal barrier is intact, and the immune activity at the gut wall is appropriately calibrated — the liver receives a largely benign input stream and processes it without difficulty.
When the gut is dysbiotic — when the microbial community has shifted toward gram-negative bacteria, when the intestinal barrier has been compromised by the dietary and pharmaceutical inputs of the modern environment, when lipopolysaccharide from bacterial outer membranes is crossing the gut wall and entering portal circulation — the liver receives a chronically inflammatory input stream that it was not designed to handle continuously. The consequence is hepatic inflammation, impaired insulin receptor signaling, elevated hepatic glucose output, and a self-reinforcing metabolic dysfunction that no amount of medication targeting downstream glucose numbers will resolve — because the upstream inflammatory driver remains unaddressed.
What you will learn:
What microbiome dysbiosis is and how it develops in the modern food and pharmaceutical environment | The specific mechanism through which gram-negative LPS crosses the gut wall and reaches the liver | How LPS activates Kupffer cells via TLR4, triggers NF-κB signaling, and directly impairs hepatic insulin receptor function | Why GGT is the most sensitive standard lab marker for gut-derived hepatic inflammatory load | The specific dietary, pharmaceutical, and environmental drivers that are producing this picture at scale | What the intervention looks like in practice — and why it requires more than fiber and probiotics
Clinical Perspective: What I See in Practice
When a patient arrives and I am trying to understand whether gut-liver axis disruption is a primary driver in their case, I cannot directly measure microbiome composition, intestinal permeability, or LPS translocation from a standard panel. These are not markers that exist in routine clinical practice. What I have instead is the clinical interview, the dietary and medication history, and the metabolic marker pattern — and in combination, these tell a coherent story that points specifically toward this mechanism when it is operating.
The marker pattern that raises my index of suspicion for gut-liver axis disruption as a primary driver is: liver enzymes above functional concern thresholds — particularly GGT, which is the most sensitive indicator of gut-derived hepatic oxidative and inflammatory load — combined with a TG/HDL ratio above 2.0, an elevated hsCRP, and a metabolic syndrome picture that does not fully resolve with dietary quality improvement alone. When fasting insulin is available and elevated, and when the patient’s history shows years of ultra-processed food consumption, high HFCS intake, regular antibiotic use, or a polypharmacy load — that combination makes me certain that the gut is a major upstream driver.
The polypharmacy picture in particular deserves to be named directly. Many of the patients who arrive in this practice are taking between 8 and 15 medications simultaneously. Not 2 or 3. Eight to fifteen. One drug to suppress a number. Another to suppress the side effect of the first. A third to manage the consequence of the second. The cumulative effect of this pharmaceutical load on gut microbiome diversity, intestinal barrier integrity, and hepatic processing capacity is not something that standard medicine addresses — in fact, it is not something that standard medicine even conceptualizes as a unified problem.
These patients are not being treated. They are being managed — downstream — while the upstream environment that is producing their symptoms becomes progressively more compromised with every additional prescription.
When I explain the gut-liver axis mechanism to patients — that gram-negative bacteria in a dysbiotic gut release LPS as a normal metabolic byproduct, that LPS can cross a compromised intestinal barrier and enter the portal vein, that it reaches the liver and activates Kupffer cells via TLR4 receptors, that this produces the hepatic inflammatory cascade that directly impairs insulin receptor signaling — the reaction is consistently: they have never heard any of this.
They have heard something vague about gut health and fiber. Some have taken a probiotic at the recommendation of a wellness influencer. But the mechanistic connection between their microbiome, their gut barrier, the bacterial endotoxin crossing into their portal circulation, and the insulin resistance in their liver that is producing their elevated fasting glucose — this causal chain is entirely absent from every clinical conversation they have had.
The GPs — and I say this without blame, because the structural constraints of a 90-second appointment leave no room for systems biology — are not discussing this. In many cases I am genuinely uncertain whether they are aware of this connection themselves. The university training in metabolic medicine that I encountered in Germany did not address it. The standard prescribing approach does not account for it. And the dietary advice that most patients receive — which includes multiple medications that further compromise gut barrier integrity while recommending ultra-processed plant-based alternatives that are among the most significant gut disruptors in the modern food supply — actively worsens the upstream condition it is supposed to be managing.
The Gut Microbiome: What It Is and What Dysbiosis Means
The human gut microbiome is a community of approximately 38 trillion microorganisms — bacteria, archaea, fungi, and viruses — residing primarily in the large intestine. In a state of health, this community is diverse, with hundreds of different species occupying different metabolic niches, producing beneficial metabolites including short-chain fatty acids, vitamins, and bile acid transformations, and maintaining a relationship with the intestinal immune system that keeps the barrier between the gut lumen and the portal circulation appropriately calibrated.
Dysbiosis is not a single defined state — it is a shift in the composition and functional capacity of this community away from the diversity and balance that characterize metabolic health. The specific features of dysbiosis that are most relevant to the gut-liver axis and insulin resistance are: a reduction in the abundance of beneficial bacteria — particularly Lactobacillus, Bifidobacterium, and short-chain fatty acid-producing Firmicutes — and a relative increase in gram-negative bacteria, whose outer membranes contain lipopolysaccharide as a structural component.
This shift matters because the gram-negative bacterial population is the source of LPS. In a diverse, balanced microbiome with an intact intestinal barrier, the LPS produced by gram-negative bacteria in the gut lumen is largely contained within the gut — handled by secretory IgA, degraded by intestinal alkaline phosphatase, and prevented from crossing the epithelial barrier in significant quantities. When dysbiosis shifts the gram-negative population upward and the barrier is simultaneously compromised, the containment system fails and LPS enters the portal circulation.
The Intestinal Barrier: How It Fails and Why It Matters
The intestinal barrier is not a passive physical wall. It is a dynamic, biologically active structure consisting of a single layer of epithelial cells connected by tight junction protein complexes — claudins, occludin, and zonula occludens proteins — that regulate the permeability of the paracellular space between cells. The mucus layer above the epithelium, produced by goblet cells, provides a physical buffer between the gut lumen and the epithelial surface. The secretory IgA layer within the mucus provides an immunological buffer. The enteric immune system below the epithelium provides a surveillance and response layer.
This barrier is maintained and regulated by the microbiome itself — specifically by the short-chain fatty acids produced by Firmicutes fermentation of dietary fiber, particularly butyrate. Butyrate is the primary energy source for colonocytes — the epithelial cells of the colon — and it is the primary signal that maintains tight junction protein expression and mucus layer thickness. When Firmicutes populations decline due to dysbiosis, butyrate production falls, colonocyte energy supply is impaired, tight junction proteins are underexpressed, and paracellular permeability increases. The barrier becomes selectively permeable — or, in clinical shorthand, leaky.
The term “leaky gut” is frequently misused in popular health media as a general explanation for everything. The mechanistic reality is more specific and more clinically important than the popular framing: it describes a measurable increase in intestinal paracellular permeability driven by specific molecular changes in tight junction architecture, produced by specific dysbiotic conditions and specific dietary and pharmaceutical insults, with specific and traceable downstream consequences for hepatic immune activation and insulin signaling. It is not a metaphor. It is a cellular and molecular event with a traceable causal chain.
The Dietary and Pharmaceutical Drivers
The modern food and pharmaceutical environment contains a specific set of inputs that disrupt the microbiome and compromise the intestinal barrier with particular efficiency. Understanding which inputs are responsible is the foundation of the intervention.
Industrial seed oils — refined omega-6-rich oils including sunflower, soybean, corn, and canola oil — are among the most significant gut barrier disruptors in the modern food supply. Their high omega-6 content, when consumed chronically in excess relative to omega-3, shifts the intestinal immune environment toward a pro-inflammatory state that degrades tight junction integrity. Their oxidation products — aldehydes, lipid peroxides, and 4-hydroxynonenal — are directly cytotoxic to colonocytes and compromise the mucus layer. They are present in virtually every ultra-processed food, every restaurant-prepared meal that does not specifically use alternative fats, and every supposedly health-promoting plant-based alternative product on the market.
High-fructose corn syrup feeds the fructose-metabolizing bacteria in the gut preferentially, disrupts the metabolic balance of the microbial community, and delivers substrate to the liver’s de novo lipogenesis pathway simultaneously — compounding gut-liver axis disruption with direct hepatic fat accumulation as covered in the previous post in this cluster.
Emulsifiers — including lecithin, carboxymethylcellulose, polysorbate-80, and carrageenan — are present in most commercially processed foods and have been shown in multiple animal and human studies to disrupt the mucus layer, alter microbiome composition toward pro-inflammatory species, and increase intestinal permeability. They are in protein bars, gym drinks, plant-based meat alternatives, keto snacks, and every other category of processed “health food” that people consume believing they are making beneficial choices.
Antibiotics are the most acutely disruptive pharmaceutical input. A single course of broad-spectrum antibiotics can reduce microbiome diversity by 30–50% and shift the community composition in ways that persist for months to years in susceptible individuals. Repeated antibiotic courses — common in patients with recurrent infections — produce cumulative dysbiosis with progressively greater barriers to microbiome recovery. The gram-negative overgrowth that follows antibiotic disruption of gram-positive populations is the direct mechanism through which antibiotic use increases LPS load in the portal circulation.
Polypharmacy extends this disruption far beyond antibiotics alone. Proton pump inhibitors alter the gastric acid environment that normally prevents bacterial overgrowth in the upper gastrointestinal tract. NSAIDs compromise the prostaglandin-dependent mechanisms that maintain intestinal barrier integrity. Metformin — the most commonly prescribed diabetes medication — alters microbiome composition significantly, a fact that is rarely communicated to patients. The patient who arrives taking 8 to 15 medications simultaneously is carrying a pharmaceutical load that, across multiple mechanisms, is continuously undermining the gut barrier that their metabolic recovery depends on.
Gluten, lectins, phytic acid, and other plant defense compounds contribute additional barrier disruption in susceptible individuals — particularly in the context of existing dysbiosis, where the barrier’s resilience is already reduced. In a healthy gut with a diverse microbiome and adequate butyrate production, these compounds are tolerated. In a dysbiotic gut with compromised tight junctions, they add to the permeability load. Reducing the diversity and processing complexity of the diet — eating simple, whole, single-ingredient foods — reduces this load meaningfully.
Environmental toxins — microplastics, heavy metals, endocrine-disrupting chemicals in cosmetics and personal care products — add a further layer of barrier disruption that is rarely accounted for in clinical practice but which accumulates continuously in the modern environment.
How LPS Activates the Liver’s Inflammatory Machinery
When intestinal barrier integrity is compromised and LPS enters the portal circulation, it arrives at the liver in concentrations that overwhelm the normal hepatic clearance mechanisms. LPS binds to Toll-like receptor 4 (TLR4) on Kupffer cells — the liver’s resident macrophages, which constitute approximately 80–90% of the body’s total macrophage population and are positioned strategically in the hepatic sinusoids to intercept portal blood.
TLR4 binding activates the MyD88-dependent signaling pathway, which leads to IκB kinase (IKK) activation, IκB degradation, and the nuclear translocation of NF-κB — the master transcription factor of the inflammatory response. NF-κB drives the expression of pro-inflammatory cytokines including TNF-alpha, IL-6, and IL-1beta, as well as inducible nitric oxide synthase (iNOS) and cyclooxygenase-2 (COX-2), producing a hepatic inflammatory environment that compounds the metabolic dysfunction already present from FFA spillover and de novo lipogenesis.
The specific mechanism through which this inflammatory activation impairs insulin signaling is the same one that operates in adipose tissue and skeletal muscle: TNF-alpha and IL-6 activate JNK (c-Jun N-terminal kinase) and IKKβ, which phosphorylate IRS-1 at serine residues rather than tyrosine residues. Serine phosphorylation of IRS-1 converts it from a signal activator to a signal inhibitor — it can no longer effectively transmit the downstream PI3K-Akt signaling that mediates insulin’s effects on glucose production suppression. The liver becomes insulin resistant through an inflammatory mechanism that is entirely independent of the lipid-mediated DAG-PKCε pathway described in the insulin resistance drives fatty liver post — two distinct molecular pathways converging on the same clinical outcome.
The consequence of this TLR4-NF-κB-mediated hepatic insulin resistance is the same as from the lipid-mediated pathway: the liver continues producing glucose autonomously despite elevated insulin, fasting glucose rises, compensatory hyperinsulinemia increases, and the downstream metabolic cascade accelerates. The upstream driver, however, is not fat accumulation. It is gut-derived bacterial endotoxin. And an intervention that addresses lipid metabolism without addressing gut barrier integrity will produce incomplete results — because the inflammatory input from the gut will continue to impair insulin receptor signaling regardless of what the lipid environment inside the hepatocyte looks like.
Why GGT Is the Most Sensitive Standard Marker for This Pathway
Of all the standard lab markers, GGT — gamma-glutamyl transferase — is the one that most directly reflects gut-derived hepatic inflammatory and oxidative load. As described in the post on ALT, AST, and GGT in the clinical interpretation cluster, GGT is not primarily a damage marker like ALT. It is a stress and adaptation marker — it rises in response to oxidative stress, hepatic metabolic burden, and the induction of the glutathione system that the liver activates when it is under sustained chemical or inflammatory pressure.
When LPS activates TLR4 on Kupffer cells and triggers the NF-κB inflammatory cascade, the resulting production of reactive oxygen species — from iNOS-derived nitric oxide, from activated NADPH oxidase, and from the mitochondrial electron transport chain under inflammatory stress — creates an oxidative burden that the liver responds to by upregulating glutathione synthesis. GGT is central to glutathione metabolism and rises proportionally to the oxidative and inflammatory load. A GGT above the functional concern threshold of 20–30 U/L — sitting within the conventional normal range of below 55–60 U/L — is a sensitive early signal that the gut-liver inflammatory axis is under strain before ALT begins to rise and before structural hepatocyte damage has occurred.
This is why, in practice, the combination of elevated GGT with elevated hsCRP — without a correspondingly elevated ALT — points specifically toward gut-derived inflammatory load as the primary hepatic stressor. The gut barrier is generating the oxidative and inflammatory signal that GGT is measuring. The structural hepatocyte damage that would elevate ALT has not yet occurred. This combination is the intervention window — the stage at which the gut-liver axis disruption is measurable, the liver has not yet been structurally damaged, and the intervention produces the most complete and durable results.
The Intervention: What Actually Restores Gut-Liver Axis Integrity
Restoring gut-liver axis integrity requires removing the inputs that are disrupting it and providing the conditions in which the microbiome can rebalance and the intestinal barrier can repair. This is not achieved by adding a probiotic supplement to an otherwise unchanged diet and lifestyle. The supplement cannot counteract the industrial seed oils, emulsifiers, HFCS, and pharmaceutical load that are continuously disrupting the system it is supposed to support.
Remove everything disrupting the barrier. This means eliminating ultra-processed foods, industrial seed oils, HFCS, emulsifiers, alcohol, packaged and microwave foods, and the entire category of processed health foods — protein bars, gym drinks, keto snacks, beyond-meat products with 40-plus ingredients — that are marketed as beneficial and are, in terms of their effect on gut barrier integrity, equivalent to or worse than the junk food they are intended to replace.
Eat whole fresh foods consisting of one ingredient. This principle — simple, local, seasonal, organic where possible — is the most powerful single dietary intervention for microbiome restoration. A diet built around meat, eggs, fish, vegetables, and fruit — each in their whole, unprocessed form — reduces the emulsifier, seed oil, additive, and HFCS load to essentially zero and provides the substrate diversity that supports microbiome balance.
Reduce restaurant eating. Restaurants — even high-quality ones — routinely cook with industrial seed oils because they are cheap and stable at high temperatures. The patient who eats breakfast at home and lunch and dinner in restaurants is receiving industrial seed oil exposure at two of three meals regardless of their home dietary choices.
Introduce natural pre- and probiotics through fermented foods. Raw kefir, raw dairy, naturally fermented pickles, and similar traditionally fermented foods provide live microbial populations and prebiotic substrates in a matrix that the gut has evolved to process. These are not equivalent to probiotic supplements — they provide microbial diversity in a food context that supports engraftment in ways that encapsulated probiotics frequently do not.
Structured irregular fasting. Fasting allows the migrating motor complex — the intestinal cleaning wave that sweeps the gut between meals — to operate effectively. Continuous eating suppresses this mechanism, promoting bacterial overgrowth in the small intestine and the fermentation-driven gas production and barrier disruption that accompanies it. Irregular intermittent fasting restores the gut’s between-meal housekeeping while simultaneously reducing the insulin load that compounds hepatic insulin resistance.
Address polypharmacy through root cause correction. This is the most clinically challenging component — and the most consequential. The patient taking 12 medications cannot repair their gut barrier while that pharmaceutical load continues to disrupt it. The path to medication reduction runs through root cause identification and correction — restoring metabolic function sufficiently that the downstream numbers the medications are managing normalize independently. This requires GP collaboration and is a process measured in months, not weeks — but it is the only pathway that addresses the problem rather than managing its consequences.
Reduce plant antinutrient load where barrier compromise is significant. In a patient with established dysbiosis and increased intestinal permeability, gluten, lectins, phytic acid, and other plant defense compounds add to the barrier disruption even when consumed in foods that are conventionally considered healthy. Reducing their intake — not permanently and not to zero, but appropriately for the clinical picture — reduces the barrier insult while the repair process proceeds.
Address environmental toxin exposure. Microplastics, heavy metals, endocrine-disrupting chemicals in personal care products, and synthetic fragrances contribute to gut barrier disruption through mechanisms that are underappreciated in clinical practice. Replacing conventional personal care products with clean alternatives reduces one component of the continuous environmental toxic load.
Create the conditions for eating as a deliberate act. This point belongs in a clinical protocol as much as any biochemical intervention: taking time to cook, calming down before eating, looking at food with gratitude — these are not optional lifestyle recommendations. They are specific activators of the parasympathetic nervous system that governs optimal gut motility, digestive enzyme secretion, and the metabolic state in which nutrients are absorbed and processed rather than rushed through a stress-activated gastrointestinal system. Chronic sympathetic dominance is a gut barrier disruptor in its own right.
A Note on Uncertainty
The mechanistic account of LPS translocation, TLR4 activation, and NF-κB-mediated hepatic insulin resistance is well-established in the research literature — including both animal models and human studies measuring portal LPS concentrations, Kupffer cell activation, and downstream inflammatory marker profiles. What is less precisely quantifiable in routine clinical practice is the relative contribution of gut-liver axis disruption versus adipose tissue FFA spillover versus de novo lipogenesis in any given patient’s insulin resistance picture. In the majority of patients presenting with established metabolic syndrome, all three mechanisms are operating simultaneously and reinforcing each other.
The intervention described above addresses all three in parallel — because the dietary changes that restore gut barrier integrity also reduce the insulin load driving lipogenesis and improve adipose tissue insulin sensitivity.
Practical Implications
If your fasting glucose remains elevated despite dietary improvement, if your GGT is above the functional concern threshold while your ALT remains normal, if your hsCRP is elevated without an obvious acute cause, and if your history includes years of ultra-processed food consumption, repeated antibiotic use, or a significant medication load — the gut-liver axis mechanism described in this post is almost certainly operating in your case.
If you do not yet have a fasting insulin result, requesting that test is the single most informative addition you can make to your standard metabolic panel.
The practical starting point is not a probiotic. It is the removal of the inputs disrupting the gut barrier — ultra-processed foods, industrial seed oils, emulsifiers, HFCS — and their replacement with whole, single-ingredient foods. The microbiome rebalances when the environment that produced the dysbiosis is changed. The intestinal barrier repairs when the inputs disrupting it are removed and the butyrate-producing conditions that maintain it are restored. And the liver begins to receive a less inflammatory portal input stream — which means the TLR4-NF-κB activation driving hepatic insulin resistance begins to resolve, and the fasting glucose that no dietary intervention targeting carbohydrate alone has been able to normalize begins to move.
The gut does not stay in the gut. Everything it produces arrives at your liver first. Restore the gut environment, and the liver’s inflammatory burden decreases — and with it, the insulin resistance that the standard management approach has been trying to manage downstream for years.
People Also Ask
What is the gut-liver axis?
The gut-liver axis describes the bidirectional communication between the intestinal microbiome and the liver through the portal vein. Everything the intestine absorbs — nutrients, bacterial metabolites, and microbial products including LPS — travels to the liver first via portal blood. When gut barrier integrity is compromised and microbiome dysbiosis increases gram-negative bacterial populations, LPS enters the portal circulation and activates the liver’s resident macrophages — Kupffer cells — triggering an inflammatory cascade that directly impairs hepatic insulin receptor signaling.
What is microbiome dysbiosis?
Microbiome dysbiosis describes a shift in the gut microbial community away from the diversity and functional balance that characterize metabolic health — specifically a reduction in beneficial short-chain fatty acid-producing bacteria and a relative increase in gram-negative bacteria whose outer membranes contain LPS. Dysbiosis is produced by the same inputs that drive metabolic dysfunction: ultra-processed foods, industrial seed oils, HFCS, emulsifiers, repeated antibiotic use, and polypharmacy.
How does LPS cause insulin resistance?
LPS — lipopolysaccharide from gram-negative bacterial outer membranes — crosses a compromised intestinal barrier and enters portal circulation, where it binds to TLR4 receptors on Kupffer cells in the liver. This activates NF-κB signaling, producing TNF-alpha and IL-6. These cytokines activate JNK and IKKβ, which phosphorylate IRS-1 at serine residues — converting it from a signal activator to a signal inhibitor. The result is impaired downstream insulin signaling in the liver, reduced suppression of hepatic glucose output, and elevated fasting glucose — hepatic insulin resistance driven by a gut-derived inflammatory mechanism.
What does elevated GGT indicate about the gut-liver axis?
GGT — gamma-glutamyl transferase — is a stress and adaptation marker rather than a damage marker. It rises in response to oxidative stress and inflammatory load in the liver. When LPS from the gut activates Kupffer cells and triggers reactive oxygen species production, GGT rises as part of the glutathione system’s adaptive response. An elevated GGT above the functional concern threshold of 20–30 U/L — particularly in combination with elevated hsCRP and a normal ALT — is a specific and sensitive early signal that gut-derived hepatic inflammatory load is present and the intervention window is open.
What are the main dietary drivers of leaky gut?
The most significant dietary drivers of intestinal barrier disruption are: industrial seed oils (through oxidative colonocyte damage and pro-inflammatory omega-6 excess), HFCS (through dysbiotic microbiome shifts), emulsifiers including carboxymethylcellulose and polysorbate-80 (through direct mucus layer disruption), and the combination of refined carbohydrates and ultra-processed food ingredients that reduce the short-chain fatty acid-producing bacteria whose butyrate output maintains tight junction integrity. Repeated antibiotic use and polypharmacy compound this disruption through pharmaceutical mechanisms.
Can polypharmacy cause gut-liver axis disruption?
Yes — through multiple mechanisms. Antibiotics directly reduce microbiome diversity and shift composition toward gram-negative overgrowth. Proton pump inhibitors alter gastric acid, promoting bacterial overgrowth in the upper GI tract. NSAIDs compromise prostaglandin-dependent intestinal barrier maintenance. Metformin significantly alters microbiome composition. The patient taking 8 to 15 medications simultaneously is carrying a pharmaceutical load that, across these mechanisms, continuously undermines the gut barrier integrity that metabolic recovery depends on.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Cani PD, Amar J, Iglesias MA, et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56(7):1761–1772. 🔗 https://pubmed.ncbi.nlm.nih.gov/17456850/
2. Tilg H, Moschen AR. Evolution of inflammation in nonalcoholic fatty liver disease: the multiple parallel hits hypothesis. Hepatology. 2010;52(5):1836–1846. 🔗 https://pubmed.ncbi.nlm.nih.gov/21038418/
3. Ghoshal S, Witta J, Zhong J, de Villiers W, Eckhardt E. Chylomicrons promote intestinal absorption of LPS. Journal of Lipid Research. 2009;50(1):90–97. 🔗 https://pubmed.ncbi.nlm.nih.gov/18815435/
4. Hotamisligil GS. Inflammation, metaflammation and immunometabolic disorders. Nature. 2017;542(7640):177–185. 🔗 https://pubmed.ncbi.nlm.nih.gov/28179656/
5. Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiological Reviews. 2018;98(4):2133–2223. 🔗 https://pubmed.ncbi.nlm.nih.gov/30067154/
6. Chassaing B, Gewirtz AT. Gut microbiota, low-grade inflammation, and metabolic syndrome. Toxicologic Pathology. 2014;42(1):49–53. 🔗 https://pubmed.ncbi.nlm.nih.gov/24285672/
7. Sonnenburg JL, Bäckhed F. Diet-microbiota interactions as moderators of human metabolism. Nature. 2016;535(7610):56–64. 🔗 https://pubmed.ncbi.nlm.nih.gov/27383980/
8. de Kort S, Keszthelyi D, Masclee AA. Leaky gut and diabetes mellitus: what is the link? Obesity Reviews. 2011;12(6):449–458. 🔗 https://pubmed.ncbi.nlm.nih.gov/21029312/
9. Bischoff SC, Barbara G, Buurman W, et al. Intestinal permeability — a new target for disease prevention and therapy. BMC Gastroenterology. 2014;14:189. 🔗 https://pubmed.ncbi.nlm.nih.gov/25407511/
10. Schnabl B, Brenner DA. Interactions between the intestinal microbiome and liver diseases. Gastroenterology. 2014;146(6):1513–1524. 🔗 https://pubmed.ncbi.nlm.nih.gov/24440671/
