Most of the conversation about fatty liver focuses on what is going into the liver — too much carbohydrate, too much fructose, too much free fatty acid spillover from insulin-resistant adipose tissue. Understanding the difference between visceral fat vs. subcutaneous fat can provide further context. The previous posts in this cluster have covered those mechanisms in depth. But there is a second dimension to fatty liver that receives almost no clinical attention — not what is going in, but what cannot get out.
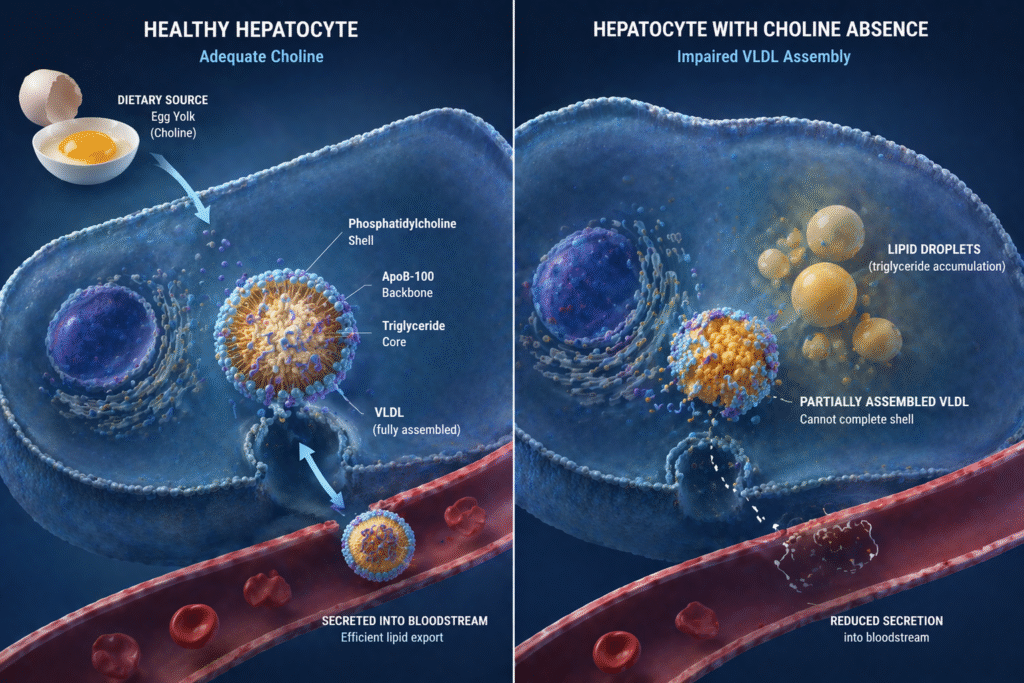
The liver has a dedicated export mechanism for the fat it accumulates: it packages triglycerides into VLDL particles and ships them into the circulation. This export process requires a specific structural molecule — phosphatidylcholine — to assemble the VLDL particle membrane. Phosphatidylcholine requires choline for its synthesis. And choline is a nutrient that a significant proportion of the population is not consuming in adequate amounts — in large part because the foods that supply it most abundantly have been the target of the most sustained and consequential dietary misinformation campaign in modern nutritional history.
Eggs. Liver. Meat. Fish. Dairy.
When choline is insufficient, the liver’s fat export machinery is compromised. Fat accumulates not only because it is being produced or delivered at elevated rates — but because it cannot be packaged and removed. The liver becomes a one-way street: fat enters through multiple pathways, but the exit is partially blocked by a nutrient deficiency that standard clinical practice does not test for, does not discuss, and in many cases actively creates by advising patients to avoid the foods that would correct it.
What you will learn: What choline is and why it is indispensable for hepatic fat export | The molecular pathway through which choline deficiency produces fatty liver — independent of insulin resistance and de novo lipogenesis | Why animal foods are the only reliable dietary source and what plant-based diets do to choline status | How to infer choline insufficiency from standard lab markers without a choline test | Why the clinical advice most patients receive about diet is doing the opposite of what their liver needs
Clinical Perspective: What I See in Practice
I will be direct: choline deficiency as a specific driver of fatty liver was not something I had placed at the center of my clinical framework until recently — until I began examining the mechanistic pathway more carefully and recognized how precisely it explained patterns I was already seeing in practice. The recognition came partly from the science and partly from something closer to home.
I eat five eggs per day, always with the yolk. My wife — 38 years old, from Southeast Asia, living in Bavaria — eats three to five eggs every day and has done so for eleven years. She eats rice daily, meat, fruit, salad. She cooks exclusively with grass-fed butter, ghee, and coconut oil. Raw milk, raw cheese, raw milk kefir, raw wild honey. No ultra-processed food of any kind. Natural movement throughout the day, no formal resistance training, no significant stress. In February of this year — less than two months ago — her labs came back. Shared here with her permission and all identifying information removed.
| Marker | Result | Unit | Functional Medicine Assessment | Functional Medicine Target |
|---|---|---|---|---|
| Fasting Glucose | 81.0 | mg/dL | Optimal | below 85 |
| HbA1c | 4.9% | % | Optimal | below 5.2 |
| AST | 22.2 | U/L | Optimal | below 25 |
| ALT | 20.9 | U/L | Optimal | below 35 |
| GGT | 10.5 | U/L | Optimal | between 20-30 |
| HDL | 56.4 | mg/dL | Optimal | above 50 |
| Triglycerides | 68.7 | mg/dL | Optimal | below 100 |
| TG/HDL Ratio | 1.2 | – | Optimal | below 2 |
| hsCRP | 0.04 | mg/L | Exceptional | below 1 |
Every single metabolic marker sits not just within the conventional normal range but at the optimal end of the functional medicine range. The TG/HDL ratio is 1.2 — well below even the most conservative functional concern threshold. The liver enzymes are in the optimal zone. The inflammatory marker is essentially zero.
Her GP’s response to these results? A red flag on total cholesterol at 228 mg/dL and LDL at 168 mg/dL. The advice: reduce meat and fat. Eat muesli for breakfast. Every two hours, small portions of wholegrain bread, carrot cake, or fruit and vegetables. This GP has such a reputation in upper Bavaria that patients wait three hours in the corridor to see him.
He looked at two numbers in isolation, flagged them as dangerous, and recommended a dietary pattern that would raise her triglycerides, lower her HDL, increase her postprandial glucose excursions, and likely — over time — begin moving her perfect metabolic markers in the wrong direction. He did not discuss the fasting glucose of 81. This omission is critical because understanding the link between NAFLD and diabetes is essential for preventing long-term metabolic decline. He did not discuss the GGT of 10.5. He did not discuss the hsCRP of 0.04 or the TG/HDL ratio of 1.2. None of those numbers existed for him clinically.
This is what the dietary misinformation around eggs and animal fat produces at the clinical coalface. A patient with an objectively exceptional metabolic panel, built over eleven years of eating five eggs a day and cooking with butter, being told by a respected physician that she needs to stop eating fat and start eating carrot cake every two hours.
The patients who arrive in this practice from the other direction — the ones who have followed the advice — present with the predictable consequence. Egg yolks discarded, only the white consumed. Animal fat replaced with oat milk and industrial seed oil. Meat reduced or eliminated. And alongside these dietary changes, a metabolic picture that standard care misinterprets: cholesterol numbers that may look acceptable, and liver enzymes that may sit within conventional reference ranges while the liver is quietly accumulating fat it cannot export because the choline that would enable that export has been eliminated from the diet.
The homocysteine elevation I see in these patients is the most consistent indirect signal. A homocysteine above the functional optimal — above 7–8 µmol/L, and particularly above 10 — in the context of low or borderline B12, low folate, and the dietary history of animal food avoidance, tells me that one-carbon metabolism is impaired. And impaired one-carbon metabolism means impaired phosphatidylcholine synthesis through the PEMT pathway — the pathway that depends on methyl groups to convert phosphatidylethanolamine into phosphatidylcholine. The fat export machinery in the liver is dependent on a metabolic pathway that a low-animal-food diet quietly dismantles.
When I explain this mechanism — that the liver cannot export fat without phosphatidylcholine, that phosphatidylcholine requires choline, that choline comes primarily from eggs and organ meat, and that avoiding these foods based on cholesterol fear is one of the reasons the liver is struggling — the reaction is consistent. The patient has never heard any of this. Their GP told them eggs cause fatty liver. The mechanism I am describing is the opposite of what they have been told, and they find it almost impossible to believe at first.
Some patients require weeks or months before the fear of cholesterol loosens its grip — not because they distrust me, but because the conditioning is deep, it is socially reinforced, and it is broadcast from every mainstream media platform they encounter. My role in these consultations is not to fight that conditioning directly. It is to explain the mechanism clearly, show them the data, and let the biology make the argument.
What Choline Is and Why the Liver Needs It
Choline is an essential nutrient — classified as such by the Institute of Medicine in 1998, yet still absent from most clinical conversations about metabolic health. It is a water-soluble compound that serves as a precursor to several critical biological molecules: phosphatidylcholine, sphingomyelin, acetylcholine, and betaine. Of these, phosphatidylcholine is the most directly relevant to hepatic fat metabolism, and its synthesis is the primary reason choline deficiency produces fatty liver with such predictable consistency.
Phosphatidylcholine is the dominant phospholipid in cell membranes throughout the body — it constitutes approximately 65–75% of the phospholipid content of most biological membranes. In the liver, it has a specific and indispensable function beyond membrane structure: it is the critical structural component of VLDL particles, the lipoproteins that the liver assembles to package and export triglycerides into the circulation.
A VLDL particle is essentially a fat transport vessel — a spherical structure with a hydrophobic core of triglycerides and cholesterol esters surrounded by a hydrophilic shell composed primarily of phospholipids, of which phosphatidylcholine is the most abundant. Apolipoprotein B-100 (ApoB-100) provides the structural backbone of the particle and the ligand for receptor-mediated clearance downstream. But the particle cannot be assembled, cannot achieve the surface properties required for stability in aqueous circulation, and cannot be secreted from the hepatocyte without an adequate supply of phosphatidylcholine.
When choline is insufficient, phosphatidylcholine synthesis is limited. VLDL particle assembly is impaired. The triglycerides that the liver has synthesized through de novo lipogenesis, or received as free fatty acids from adipose tissue, cannot be packaged for export. They remain inside the hepatocyte. Fat accumulates — not because production is elevated, but because the export mechanism is broken.
Every other mechanism discussed in this cluster — FFA spillover from adipose tissue, de novo lipogenesis driven by fructose and hyperinsulinemia — increases the fat load entering the liver. To understand how insulin resistance drives fatty liver, read our dedicated post. Choline deficiency does something different. It blocks the door through which fat leaves. Choline deficiency does something different. It blocks the door through which fat leaves.
The Two Pathways of Phosphatidylcholine Synthesis
The liver synthesizes phosphatidylcholine through two pathways, and understanding both clarifies why choline deficiency is particularly consequential and why animal foods are uniquely important for maintaining hepatic choline status.
The CDP-choline pathway (Kennedy pathway) is the primary route and accounts for approximately 70% of hepatic phosphatidylcholine synthesis. It begins with choline itself — absorbed from the diet or released from membrane phospholipid turnover — which is phosphorylated to phosphocholine, then converted to CDP-choline, and finally combined with diacylglycerol to produce phosphatidylcholine. This pathway is directly and stoichiometrically dependent on dietary choline availability. When dietary choline falls below the threshold required to maintain hepatic CDP-choline flux, VLDL assembly is impaired proportionally.
The PEMT pathway (phosphatidylethanolamine N-methyltransferase) accounts for approximately 30% of hepatic phosphatidylcholine synthesis. It converts phosphatidylethanolamine to phosphatidylcholine through three successive methylation reactions, each requiring a methyl group donated by S-adenosylmethionine (SAM). SAM is produced from methionine through the one-carbon metabolic cycle, which depends on adequate folate and vitamin B12 for the regeneration of homocysteine back to methionine. The PEMT pathway is therefore dependent not on dietary choline directly, but on the methylation capacity of one-carbon metabolism — which is itself dependent on B12, folate, and methionine availability from animal protein.
The clinical consequence of this dual dependency is important. A patient following a low-animal-food diet is simultaneously depleting both pathways: the CDP-choline pathway is impaired by insufficient dietary choline from eggs, liver, and meat; and the PEMT pathway is impaired by declining B12 and folate status — both of which are most bioavailable from animal foods — reducing SAM availability for the methylation reactions required to produce phosphatidylcholine endogenously. The two pathways support each other, and a dietary pattern that restricts animal foods undermines both simultaneously.
This is why elevated homocysteine is such a useful clinical signal in this context. Homocysteine is an intermediate in the one-carbon cycle — it is remethylated back to methionine using B12 and folate as cofactors. When B12 and folate are insufficient, this remethylation is impaired, homocysteine accumulates in the blood, and SAM availability — the methyl donor for PEMT — falls. Elevated homocysteine is therefore not only an independent cardiovascular risk marker. It is a direct signal that the PEMT pathway is running below capacity, that phosphatidylcholine synthesis is impaired, and that VLDL export from the liver is compromised.
Why Animal Foods Are the Only Reliable Dietary Source
Choline is found in meaningful concentrations in a limited range of foods, and the pattern of its distribution in the food supply is one of the most clinically consequential facts in nutritional biochemistry — because it is essentially the inverse of the foods that mainstream dietary advice has been systematically discouraging for the past several decades.
Egg yolk is by far the richest dietary source of choline, containing approximately 147 mg per large egg yolk. A single egg provides approximately 25% of the adequate intake for choline. Five eggs per day — the quantity I consume myself — provide more than 100% of the daily adequate intake from this single food alone. Beef liver contains approximately 356 mg of choline per 100 grams, making it the most concentrated source per unit weight of any commonly available food. Chicken liver, beef, shrimp, salmon, and cod provide substantial amounts. Dairy contributes modest but meaningful quantities.
Plant foods contain choline in significantly lower concentrations. Soybeans, which are among the richest plant sources, provide approximately 107 mg per cup cooked — less than a single egg yolk per serving, and in a matrix with lower bioavailability. Cruciferous vegetables, legumes, and nuts contain small amounts. A dietary pattern that eliminates eggs, liver, and meat and replaces them with plant alternatives cannot reliably meet choline requirements through food alone.
The Adequate Intake for choline established by the Institute of Medicine is 425 mg per day for women and 550 mg per day for men. These thresholds were derived largely from liver fat accumulation studies — the level of intake below which hepatic fat begins to increase. National dietary surveys consistently show that a significant proportion of the population — and a substantially higher proportion of those following plant-based or low-animal-food diets — falls below these thresholds. The deficiency is not rare or edge-case. It is common, it is largely dietary in origin, and it is driven in part by the same nutritional messaging that has made egg yolks a symbol of cardiovascular risk.
The egg yolk specifically deserves attention — not only for its choline content but for the clinical absurdity of the advice to discard it. The yolk is where essentially all of the choline in an egg resides. It is also where the fat-soluble vitamins, the carotenoids, the omega-3 fatty acids, the lecithin, and the cholesterol are — the cholesterol that the body uses to synthesize steroid hormones, bile acids, and cell membranes. The instruction to eat only the white — which is primarily albumin protein — and discard the nutritionally dense yolk is metabolically equivalent to being told to eat the packaging and throw away the food.
The Relationship Between Choline Deficiency and Cholesterol
The cholesterol discussion cannot be avoided in the context of choline and fatty liver, because it is the fear of dietary cholesterol — concentrated almost exclusively in the same animal foods that provide choline — that is driving the dietary pattern that produces choline deficiency.
The relationship between dietary cholesterol and serum cholesterol is far more complex and far less linear than the model that has dominated medical education for fifty years. The liver is the primary regulator of circulating cholesterol — it produces approximately 75–80% of total body cholesterol endogenously, through the HMG-CoA reductase pathway, and adjusts its production in response to dietary intake through feedback regulation. When dietary cholesterol increases, hepatic cholesterol synthesis decreases proportionally in the majority of individuals. When dietary cholesterol decreases — by eliminating eggs and animal foods — hepatic synthesis increases to compensate. The serum cholesterol level is regulated primarily by the liver, not determined primarily by dietary intake.
The clinical consequence of this biology — regularly visible in practice — is the patient who has eliminated eggs and animal fat for years, whose total cholesterol and LDL sit at levels their GP considers acceptable, and whose metabolic picture tells a completely different story: triglycerides elevated, HDL suppressed, TG/HDL ratio above 2.0, fasting insulin above 10 µIU/mL, liver enzymes trending upward, and homocysteine elevated — the full fingerprint of choline-insufficient, insulin-resistant hepatic dysfunction — while the GP is congratulating them on their cholesterol number.
Compare this to the metabolic picture of someone eating five eggs per day with the yolk: fasting glucose of 81, HbA1c of 4.9, ALT of 20.9, GGT of 10.5, HDL of 56.4, triglycerides of 68.7, hsCRP of 0.04. This is what adequate choline intake, combined with the full nutritional package of animal foods — fat-soluble vitamins, B12, bioavailable protein, omega-3 fatty acids — does to the metabolic picture over years of consistent consumption. The total cholesterol and LDL may be above the conventional threshold. Every other marker tells you that the liver is functioning optimally, inflammation is essentially absent, and the metabolic machinery is operating exactly as it should.
Cholesterol is not blood fat. It is not the same as triglycerides. LDL is not cholesterol — it is a lipoprotein particle that carries cholesterol and other lipids through the circulation. These are distinctions that patients arriving in this practice frequently do not know, because their GPs have communicated all lipid markers as “cholesterol” or “blood fat” — a conflation that has produced decades of dietary decisions based on a fundamental misunderstanding of lipid physiology.
How to Infer Choline Insufficiency Without a Choline Test
Choline is not measured in standard clinical panels. There is no routine serum choline test in GP practice. The assessment of choline status therefore requires inference from the combination of dietary history, clinical picture, and indirect biochemical markers.
Dietary history is the most direct starting point. A patient who has eliminated or significantly reduced egg yolk, organ meat, red meat, and dairy from their diet over a period of months or years is at high likelihood of choline insufficiency. The specific pattern of being told to avoid these foods by a clinician — and following that advice — is a clinical red flag for choline deficiency that is far more informative than any biomarker.
Homocysteine is the most accessible indirect marker. Above the functional optimal of 7–8 µmol/L — and particularly above 10 µmol/L — homocysteine signals that one-carbon metabolism is under strain. In the context of low or borderline B12 and folate, this signals specifically that the PEMT pathway for phosphatidylcholine synthesis is impaired. Homocysteine above 12 µmol/L in a patient eating a low-animal-food diet is a strong combined signal of choline insufficiency, B12 deficiency, and impaired hepatic VLDL export capacity.
Vitamin B12 — both serum B12 and, where available, active B12 (holotranscobalamin) or methylmalonic acid — provides direct assessment of the B12 status that the PEMT pathway depends on. Serum B12 below 300 pmol/L warrants attention. Below 200 pmol/L is clinically significant deficiency by most functional medicine standards, even if it falls within the conventional laboratory reference range.
Folate supports the same one-carbon cycle that B12 depends on. Low folate compounds the impairment of PEMT-mediated phosphatidylcholine synthesis and amplifies the homocysteine elevation.
ALT and GGT above functional concern thresholds — in the context of the dietary history and homocysteine elevation described above — complete the picture. The liver is under metabolic stress. Fat export is impaired. The combination of low animal food intake, elevated homocysteine, low B12, and rising liver enzymes in the absence of significant metabolic syndrome markers points specifically toward choline insufficiency as a primary contributor rather than de novo lipogenesis or FFA spillover as the dominant mechanism.
Ferritin in the context of low B12 and low animal food intake adds another dimension — both are markers of the nutritional insufficiency that plant-predominant diets produce, and both compound the metabolic consequences of inadequate animal food intake.
The Intervention: What Restores Hepatic Choline Status
The primary intervention for choline insufficiency is the most direct one available: reintroduce the foods that provide it. Eggs — with the yolk — are the most practical, most accessible, and most cost-effective source of dietary choline available. Three to five eggs per day, consistently, provides a substantial and reliable choline intake that the CDP-choline pathway can use directly. Beef liver, consumed once or twice per week, provides the single most concentrated dietary choline source available alongside exceptional B12, retinol, iron, and folate content. Meat and fish eaten regularly contribute meaningfully to the cumulative daily intake.
The educational challenge in implementing this intervention is significant, and navigating it requires clinical patience rather than confrontation. The fear of dietary cholesterol is not a rational position held loosely — it is a deeply conditioned belief reinforced by decades of public health messaging, social pressure, and media amplification. Patients who have been telling themselves for years that they are protecting their heart by avoiding egg yolks do not release that belief in one consultation. What changes the belief durably is not argument. It is mechanism, explained clearly, connected to their specific markers, and then confirmed by the lab results that follow the dietary change.
When the dietary change is made and sustained, the markers that reflect VLDL export capacity and one-carbon metabolism begin to move. Homocysteine falls as B12 and choline availability restore the methylation cycle. Triglycerides fall as VLDL export becomes more efficient and hepatic fat accumulation decreases. Liver enzymes move toward the functional optimal range. The timeline depends on how long the deficiency has been present and how completely the dietary change is implemented — but meaningful marker movement is typically visible within 8 to 12 weeks of consistent high-choline animal food intake.
For patients in whom dietary change is insufficient or for whom the transition is too slow, phosphatidylcholine supplementation can support VLDL assembly more directly. Lecithin — a commercial form of phosphatidylcholine derived from sunflower or soy — provides the preformed phospholipid without requiring the CDP-choline synthesis steps. CDP-choline and alpha-GPC are alternative choline forms with good bioavailability. These supplements do not replace the full nutritional package that animal foods provide — the B12, the retinol, the heme iron, the bioavailable protein — but they address the specific VLDL export deficit while dietary habits are being restructured.
A Note on Uncertainty
The relationship between choline deficiency and fatty liver is established in both animal models and controlled human depletion studies — the latter demonstrating that healthy adults on choline-deficient diets develop hepatic steatosis within weeks, which reverses upon choline repletion. The mechanism through which choline supports VLDL assembly — via phosphatidylcholine synthesis through the CDP-choline pathway — is well-characterized biochemically.
What is less precisely quantified in routine clinical practice is the contribution of choline deficiency relative to insulin resistance, de novo lipogenesis , and FFA spillover in any given patient’s hepatic fat accumulation. This is particularly relevant when considering conditions like lean fatty liver disease. In most patients with established metabolic syndrome and fatty liver, multiple mechanisms are operating simultaneously. Choline insufficiency compounds the others — it does not typically act in isolation in the clinical presentations seen in this practice. The intervention addresses all mechanisms in parallel, but recognizing choline’s specific contribution changes the dietary recommendations fundamentally — particularly the instruction to reintroduce, rather than avoid, the animal foods that standard care has been restricting.
Practical Implications
If you have been avoiding eggs, restricting meat, and using plant-based alternatives on the advice of a clinician concerned about your cholesterol — and if your liver enzymes are above the functional concern threshold, your homocysteine is elevated, your B12 is borderline or low, and your triglycerides remain above the functional optimal despite dietary effort — choline insufficiency is almost certainly part of your picture.
The practical starting point is reintroducing egg yolks. Not egg whites. The yolk. Three to five per day is not an extreme intervention — it is the dietary pattern that provides adequate choline, adequate B12, adequate fat-soluble vitamins, and the phospholipid substrate that your liver needs to export the fat it has accumulated. If your cholesterol rises modestly as a result — as it may — read the full marker context before reacting to the number. A rising HDL, falling triglycerides, improving TG/HDL ratio, falling homocysteine, and normalizing liver enzymes in the context of a slightly higher LDL is not a deteriorating metabolic picture. It is an improving one.
Your liver cannot export fat without phosphatidylcholine. Phosphatidylcholine requires choline. Choline comes from eggs. The advice to avoid eggs to protect your liver is, mechanistically, one of the most precisely wrong pieces of dietary guidance in modern clinical practice.
People Also Ask
What does choline do for the liver?
Choline is required for the synthesis of phosphatidylcholine, the primary phospholipid in VLDL particle membranes. VLDL is the lipoprotein that the liver assembles to package and export triglycerides into the circulation. Without adequate choline, VLDL assembly is impaired, triglyceride export is reduced, and fat accumulates inside hepatocytes —
What foods are highest in choline?
Egg yolk is the most practical and accessible concentrated source, providing approximately 147 mg per large yolk. Beef liver is the most concentrated source per gram, providing approximately 356 mg per 100 grams. Chicken liver, beef, shrimp, salmon, cod, and dairy contribute meaningfully. Plant foods contain significantly lower concentrations and cannot reliably meet choline requirements without animal food inclusion.
Can a vegan or plant-based diet cause fatty liver through choline deficiency?
Yes — through two converging mechanisms. The CDP-choline pathway is directly limited by insufficient dietary choline from animal foods. The PEMT pathway — which synthesizes phosphatidylcholine endogenously using methyl groups from one-carbon metabolism — is limited by declining B12 and folate status, which are most bioavailable from animal foods. Both pathways are compromised simultaneously by low animal food intake, reducing hepatic VLDL export capacity and promoting hepatic fat accumulation independent of carbohydrate intake or insulin resistance.
What is homocysteine and why does it matter for liver fat?
Homocysteine is an intermediate in one-carbon metabolism — the biochemical cycle through which methyl groups are transferred for multiple cellular functions, including phosphatidylcholine synthesis via the PEMT pathway. When B12 and folate are insufficient, homocysteine accumulates in the blood because the remethylation step back to methionine is impaired. Elevated homocysteine signals both the cardiovascular risk associated with impaired one-carbon metabolism and the specific impairment of PEMT-mediated phosphatidylcholine synthesis — making it a clinically useful indirect marker of choline insufficiency and reduced hepatic VLDL export capacity.
Is it safe to eat eggs every day?
In the context of a metabolically healthy dietary pattern — adequate animal protein, elimination of ultra-processed foods, absence of chronic hyperinsulinemia — daily egg consumption is not only safe but metabolically beneficial. The dietary cholesterol in eggs is regulated by hepatic feedback mechanisms in the majority of individuals. The choline, fat-soluble vitamins, omega-3 fatty acids, and bioavailable protein in egg yolk support liver function, one-carbon metabolism, and metabolic health in ways that no plant-based alternative replicates. Controlled trials and large observational studies do not support an association between egg consumption and cardiovascular disease in metabolically healthy individuals.
Why do my liver enzymes stay elevated even though I eat very little fat?
Liver enzyme elevation in the context of a low-fat, low-animal-food diet may reflect precisely the mechanism described in this post. Choline insufficiency from avoiding egg yolk, organ meat, and animal fat impairs VLDL assembly, reducing the liver’s ability to export accumulated fat. Simultaneously, the carbohydrate load that replaces animal fat in plant-based dietary patterns drives de novo lipogenesis and maintains the insulin elevation that activates SREBP-1c. The liver is accumulating fat from two directions — reduced export capacity from choline deficiency, increased production from carbohydrate-driven lipogenesis — while the dietary advice directing the patient toward this pattern continues to be reiterated by mainstream clinical guidance.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Zeisel SH, da Costa KA. Choline: an essential nutrient for public health. Nutrition Reviews. 2009;67(11):615–623. 🔗 https://pubmed.ncbi.nlm.nih.gov/19906248/
2. Yao ZM, Vance DE. The active synthesis of phosphatidylcholine is required for very low density lipoprotein secretion from rat hepatocytes. Journal of Biological Chemistry. 1988;263(6):2998–3004. 🔗 https://pubmed.ncbi.nlm.nih.gov/3277970/
3. da Costa KA, Niculescu MD, Craciunescu CN, Fischer LM, Zeisel SH. Choline deficiency increases lymphocyte apoptosis and DNA strand breaks. Nutrition Research. 2006;26(11):545–552. 🔗 https://pubmed.ncbi.nlm.nih.gov/17097613/
4. Corbin KD, Zeisel SH. Choline metabolism provides novel insights into nonalcoholic fatty liver disease and its progression. Current Opinion in Gastroenterology. 2012;28(2):159–165. 🔗 https://pubmed.ncbi.nlm.nih.gov/22134222/
5. Buchman AL, Dubin MD, Moukarzel AA, et al. Choline deficiency: a cause of hepatic steatosis during parenteral nutrition that can be reversed with intravenous choline supplementation. Hepatology. 1995;22(5):1399–1403. 🔗 https://pubmed.ncbi.nlm.nih.gov/7591621/
6. Fischer LM, Scearce JA, Mar MH, et al. Ad libitum choline intake in healthy individuals meets or exceeds the proposed adequate intake level. Journal of Nutrition. 2005;135(4):826–829. 🔗 https://pubmed.ncbi.nlm.nih.gov/15795440/
7. Vance JE, Vance DE. Phospholipid biosynthesis in mammalian cells. Biochemistry and Cell Biology. 2004;82(1):113–128. 🔗 https://pubmed.ncbi.nlm.nih.gov/15052333/
8. Resseguie ME, Song J, Niculescu MD, da Costa KA, Randall TA, Zeisel SH. Phosphatidylethanolamine N-methyltransferase (PEMT) gene expression is induced by estrogen in human and mouse primary hepatocytes. FASEB Journal. 2007;21(10):2622–2632. 🔗 https://pubmed.ncbi.nlm.nih.gov/17395833/
9. Hollenbeck CB. The importance of being choline. Journal of the American Dietetic Association. 2010;110(8):1162–1165. 🔗 https://pubmed.ncbi.nlm.nih.gov/20656091/
10. Bidulescu A, Chambless LE, Siega-Riz AM, Zeisel SH, Heiss G. Usual choline and betaine dietary intake and incident coronary heart disease: the Atherosclerosis Risk in Communities (ARIC) study. BMC Cardiovascular Disorders. 2007;7:20. 🔗 https://pubmed.ncbi.nlm.nih.gov/17659082/
