How insulin resistance drives fatty liver comes down to three converging pathways — each independent, each driven by the same upstream hormonal disturbance, and each capable of producing hepatic fat accumulation in individuals whose weight, glucose, and liver enzymes all appear unremarkable on a standard panel. Understanding those pathways is what separates a diagnosis that arrives too late from one that could have been made years earlier.
Fatty liver is almost universally framed as a weight problem. It is not. It is an insulin problem. And understanding that distinction changes everything about how you detect it, interpret it, and address it.
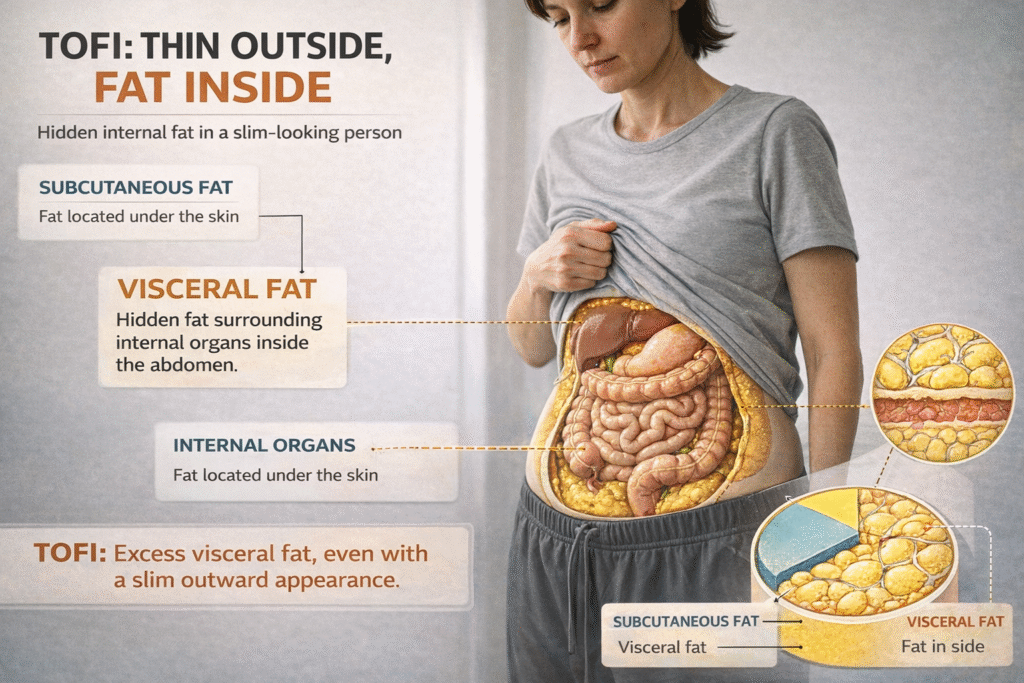
The clinical assumption — that fatty liver requires obesity to develop — is contradicted by the evidence. Studies consistently show that 10–20% of individuals with non-alcoholic fatty liver disease have a normal BMI. What they share with their overweight counterparts is not excess body weight. It is insulin resistance.
This article explains the precise mechanistic chain through which insulin resistance drives hepatic fat accumulation — in lean individuals and overweight individuals alike. Not as a surface summary, but as a full biological account of how a signaling problem in peripheral tissue becomes a structural problem inside the liver.
For the broader clinical framework, see my cornerstone article on fatty liver and metabolic dysfunction, where I explain how hepatic fat accumulation fits into the larger pattern of insulin resistance, visceral fat, inflammation, and metabolic disease.
What you will learn: Why insulin resistance — not caloric excess — is the central driver of fatty liver | The three-pathway model through which insulin resistance delivers fat to the liver | How the liver’s own insulin resistance then creates a self-reinforcing loop | Why standard liver enzyme tests miss this process entirely | What markers actually reflect the insulin-fatty liver connection
The Core Argument: Fatty Liver Is a Downstream Consequence of Insulin Resistance
Most clinical and popular explanations of fatty liver describe it as a storage problem — the liver accumulates fat because too much energy is coming in. This framing is incomplete. It describes the proximate mechanism without identifying the upstream driver.
The upstream driver, in the overwhelming majority of cases, is insulin resistance.
Insulin resistance does not cause fatty liver through one pathway. It causes it through three converging pathways, each of which independently increases the fat load delivered to or produced inside the liver. When all three are operating simultaneously — which they typically are in established insulin resistance — hepatic fat accumulation is not a matter of if. It is a matter of when.
Understanding each pathway is what allows you to read metabolic markers intelligently and intervene at the root rather than at the symptom.
Clinical Perspective: What I See in Practice
The patient whose lab results are referenced below — shared here with her permission and fully anonymized — is not unusual in my practice. What is unusual is that she got answers.
Her ALT had been sitting at 78 U/L — nearly two and a half times the upper functional limit. Her fasting glucose was running between 174 and 186 mg/dL. Her triglycerides were elevated at 178 mg/dL, her HOMA-IR was 3.7, and her HbA1c was 7.6%. Her previous doctor had focused almost entirely on LDL — which, when interpreted in isolation from HDL and CRP, painted a misleadingly alarming picture.
The metabolic story hiding in her triglycerides, her insulin, and her liver enzyme was not being read at all. Within 16 weeks of addressing the upstream insulin drivers, her ALT had dropped to 32 U/L. A reduction of 46 points — not from a liver medication, but from correcting what was causing the liver to fail in the first place.
This pattern — elevated liver markers, elevated triglycerides, and a diagnosis that arrives late or not at all — is one I encounter consistently. What I see most often in patients presenting with fatty liver at a normal or near-normal weight is a specific dietary and lifestyle signature: frequent snacking throughout the day, significant intake of fructose from sodas and sweetened drinks, a high carbohydrate load built around what is culturally considered healthy food — in Germany, this frequently means large quantities of whole grain bread consumed from early morning — and a background of chronic work stress and emotional eating that keeps cortisol elevated and compounds insulin resistance through a separate pathway entirely.
These patients arrive having been told, if anything, that their liver enzyme elevation is mild and bears watching. Almost none of them have been told that the liver is an insulin-sensitive organ. None have heard of hepatic insulin resistance specifically. And none have been introduced to the gut-liver axis — the pathway through which intestinal permeability allows lipopolysaccharide to enter portal circulation, activate TLR4 on Kupffer cells, trigger NF-κB-mediated inflammation, and directly impair hepatic insulin signaling. When I explain this sequence in a consultation, the reaction is consistently the same: close attention, and then the question — why has no one ever explained this to me?
In the absence of fasting insulin — which, as I have described elsewhere, is rarely ordered in standard German clinical practice — the marker I rely on most to signal that hepatic insulin resistance is already underway is the triglyceride-to-HDL ratio. Elevated triglycerides in the context of low or moderate HDL, combined with any upward drift in ALT or GGT, tells me the liver is under metabolic stress before any imaging confirms it. When fasting insulin is subsequently added and comes back above 15 µIU/mL, the picture is complete. The fatty liver is not a weight problem. It never was. It is an insulin problem — and the data, interpreted correctly, makes that visible years before the conventional diagnostic system would flag it.
How Insulin Resistance Drives Fatty Liver
Pathway One: Adipose Tissue Insulin Resistance and Chronic Free Fatty Acid Spillover
In a metabolically healthy individual, adipose tissue performs a critical buffering function. After a meal, rising insulin suppresses lipolysis — the enzymatic breakdown of stored triglycerides into free fatty acids (FFAs) — by inhibiting hormone-sensitive lipase (HSL). Fat stays inside fat cells. The bloodstream is not flooded with fatty acids at the same moment it is absorbing dietary glucose.
This suppression of lipolysis by insulin is one of insulin’s most important metabolic functions. It coordinates the body’s fuel sources so that storage and oxidation happen in an organized, time-gated manner.
In insulin-resistant adipose tissue, this suppression fails.
HSL remains inappropriately active even in the presence of insulin. Fat cells continuously release FFAs into the portal and systemic circulation — not just after meals, but throughout the day and overnight. The fasted state, which should be characterized by controlled, regulated fat mobilization, instead becomes a state of chronic, unregulated FFA release.
The liver is the first organ downstream of this spillover. Via the portal vein, it receives a continuous and disproportionate FFA load that far exceeds its oxidative capacity. What the liver cannot oxidize for energy, it re-esterifies into triglycerides and stores — or packages into VLDL particles for export. When the rate of incoming FFAs exceeds both oxidative capacity and export capacity, triglycerides accumulate inside hepatocytes.
The landmark tracer study by Donnelly et al. (2005) quantified this precisely in humans with established fatty liver: approximately 60% of hepatic triglyceride derived from circulating FFAs, the majority of which were adipose-derived. Dietary fat accounted for only about 15%. The liver was not primarily a victim of what people ate. It was a victim of what their adipose tissue was releasing.
This is why fatty liver can develop — and frequently does — in individuals who eat a moderate diet and maintain a normal body weight. If adipose tissue insulin resistance is present, the liver is receiving a chronic excess FFA load regardless of what is on the plate.
Pathway Two: Hyperinsulinemia Drives De Novo Lipogenesis in the Liver
The second pathway operates through a different mechanism and compounds the first.
As peripheral insulin resistance develops in muscle and adipose tissue, the pancreas compensates by secreting more insulin — a state called hyperinsulinemia. This compensatory response maintains near-normal blood glucose for years or decades, which is why standard glucose testing misses early metabolic dysfunction. But the elevated insulin itself is not metabolically neutral.
The liver retains insulin sensitivity — particularly for the lipogenic (fat-producing) actions of insulin — significantly longer than peripheral tissues do. This selective hepatic insulin sensitivity is one of the most clinically important and least discussed aspects of the insulin resistance story.
What it means in practice: while muscle and fat cells are increasingly resistant to insulin’s glucose-disposal signal, the liver continues to respond robustly to insulin’s signal to produce fat through de novo lipogenesis (DNL) — the conversion of excess carbohydrate substrate into triglycerides.
Insulin activates the transcription factor SREBP-1c (sterol regulatory element-binding protein 1c) in hepatocytes, which upregulates the full enzymatic machinery of DNL — including fatty acid synthase (FAS), acetyl-CoA carboxylase (ACC), and stearoyl-CoA desaturase (SCD-1). The result is increased endogenous fat production inside the liver, driven directly by the elevated insulin levels that are themselves the consequence of peripheral insulin resistance.
This is the metabolic paradox of selective hepatic insulin resistance: the liver is resistant to insulin’s glucose-suppressing effects but remains sensitive to its fat-producing effects. Chronically elevated insulin therefore simultaneously fails to suppress hepatic glucose output and actively drives hepatic fat production. Both consequences worsen the metabolic picture.
Fructose deserves specific mention here, not as an independent villain but as a substrate that amplifies this pathway in vulnerable individuals. Fructose is metabolized almost exclusively in the liver, bypasses the main regulatory checkpoint of glycolysis (phosphofructokinase), and is rapidly converted to acetyl-CoA — the primary building block for DNL. In the context of hyperinsulinemia, excess carbohydrate intake, and low muscle glucose disposal capacity, high fructose intake can substantially accelerate hepatic fat production. The context determines the magnitude of the effect.
Pathway Three: Impaired Hepatic Fat Oxidation and Export
The third pathway is less discussed but mechanistically important. Insulin resistance does not only increase the fat delivered to the liver — it simultaneously impairs the liver’s ability to clear it.
Beta-oxidation — the process by which the liver breaks down fatty acids for energy — requires entry of fatty acids into the mitochondria via the carnitine shuttle (carnitine palmitoyltransferase 1, CPT-1). Malonyl-CoA, an intermediate produced during active DNL, inhibits CPT-1 and effectively blocks mitochondrial fatty acid entry. When DNL is upregulated by hyperinsulinemia, malonyl-CoA accumulates, CPT-1 is inhibited, and the liver’s capacity to oxidize the incoming fat load is simultaneously reduced.
Additionally, VLDL export — the liver’s mechanism for packaging and shipping excess triglycerides into circulation — has a finite capacity. When the rate of fat delivery and production exceeds export capacity, hepatic triglyceride accumulation becomes inevitable regardless of dietary behavior. This export process also depends on adequate choline status, because choline deficiency can impair VLDL export and make hepatic triglyceride accumulation more likely.
The liver is receiving more fat via pathway one, producing more fat via pathway two, and oxidizing and exporting less fat via pathway three. All three vectors are driven by the same upstream problem: insulin resistance and its compensatory hyperinsulinemia.
How Hepatic Fat Then Creates Its Own Insulin Resistance: The Feedback Loop
Once triglycerides begin accumulating inside hepatocytes, a secondary and self-reinforcing process begins.
Triglyceride itself is relatively inert. But the metabolic intermediates generated during active lipid flux — particularly diacylglycerol (DAG) — are not. DAG accumulates in hepatocytes as triglyceride synthesis and turnover increase, and it activates a specific isoform of protein kinase C: PKCε.
PKCε translocates to the cell membrane and directly phosphorylates the insulin receptor at a threonine residue (Thr1160), impairing its kinase activity. This reduces the downstream phosphorylation of insulin receptor substrate 1 (IRS-1), which in turn impairs the PI3K–Akt signaling cascade that normally suppresses hepatic glucose production.
The result is hepatic insulin resistance — the liver loses its ability to respond to insulin’s signal to stop producing glucose. It continues gluconeogenesis and glycogenolysis even in the fed, insulin-elevated state. Fasting glucose may remain technically normal for years, but fasting insulin rises steadily as the pancreas compensates for the liver’s unresponsiveness.
This creates the central feedback loop:
Peripheral insulin resistance → hyperinsulinemia → hepatic fat accumulation → DAG → PKCε → hepatic insulin resistance → greater hyperinsulinemia → more hepatic fat accumulation.
Each step amplifies the next. The process is self-sustaining once established, which is why early identification — before structural liver changes occur — matters so much clinically.
Why Standard Liver Tests Miss This Entirely
ALT and AST — the liver enzymes included in most standard blood panels — reflect hepatocyte damage and cell death. They are markers of inflammation and injury, not of fat accumulation or insulin signaling impairment.
Multiple studies have confirmed that ALT and AST can remain entirely within the laboratory reference range in the presence of significant hepatic steatosis. A 2019 analysis published in the Journal of Hepatology found that a substantial proportion of individuals with confirmed fatty liver on imaging had normal ALT values. GGT is somewhat more sensitive to oxidative stress and metabolic dysfunction but is not definitive.
Normal liver enzymes do not exclude fatty liver. They do not exclude hepatic insulin resistance. They do not reflect the DAG–PKCε mechanism operating inside hepatocytes. They are the wrong tool for the question.
The markers that actually reflect the insulin–fatty liver connection are metabolic, not hepatic:
Fasting insulin: The most direct measure of compensatory hyperinsulinemia — the hormonal driver of both pathway two and the feedback loop. A fasting insulin above 10 µIU/mL warrants investigation regardless of liver enzyme values.
HOMA-IR: Combines fasting insulin and fasting glucose to quantify the degree of insulin resistance. A HOMA-IR above 2.0 indicates significant insulin resistance; above 2.9 is associated with substantially elevated metabolic and cardiovascular risk.
Triglyceride-to-HDL ratio: Reflects the dyslipidemic pattern driven by hyperinsulinemia — elevated VLDL triglyceride synthesis and suppressed HDL. A triglyceride-to-HDL ratio above 2.0 (using mg/dL) or 0.9 (using mmol/L) is a robust clinical proxy for insulin resistance and correlates strongly with hepatic fat accumulation.
Waist-to-height ratio: Visceral adiposity — even in normal-weight individuals — is both a marker and a driver of adipose tissue insulin resistance. A waist-to-height ratio above 0.5 indicates metabolically significant central fat regardless of BMI.
Post-meal glucose response: Exaggerated postprandial glucose excursions reflect the impaired peripheral glucose disposal that drives compensatory hyperinsulinemia. Where continuous glucose monitoring is available, it provides direct insight into the glycemic environment that feeds pathway two.
The Lean Individual: Why Normal Weight Provides No Protection
The mechanisms described above operate independently of total body fat mass. What determines whether fatty liver develops is not how much fat a person carries overall — it is whether their adipose tissue is insulin resistant, whether their pancreas is secreting excess insulin, and whether their liver is receiving a fat load it cannot clear.
In lean individuals, two specific vulnerabilities amplify this risk. First, subcutaneous fat — the fat stored under the skin — has a limited storage capacity that varies between individuals. When that capacity is exceeded, fat is preferentially directed toward visceral depots and ectopic sites, including the liver and skeletal muscle. A lean person with limited subcutaneous fat expandability may reach hepatic fat accumulation at a lower total body fat percentage than an obese individual with high subcutaneous storage capacity.
Second, lean individuals with low muscle mass have reduced peripheral glucose disposal capacity. Skeletal muscle accounts for approximately 80% of insulin-mediated glucose uptake. Low muscle mass means a higher fraction of ingested carbohydrate must be handled by the liver, increasing the substrate pressure for DNL and raising the insulin secretion required to maintain glycemic control.
This is the metabolic profile of lean fatty liver: normal BMI, visceral fat dominance, low muscle mass, chronically elevated fasting insulin, and a liver quietly accumulating fat through all three pathways described above — while every standard lab test returns unremarkable results.
For a full clinical discussion of this phenotype, see: Lean Fatty Liver Disease: Why Slim People Develop Fatty Liver Too.
Connecting This to the Broader Insulin Resistance Trajectory
Hepatic insulin resistance — established through the DAG–PKCε mechanism — is not an isolated liver problem. It is a central node in the broader insulin resistance trajectory that, if unaddressed, progresses toward type 2 diabetes, cardiovascular disease, and non-alcoholic steatohepatitis (NASH) with potential fibrosis.
The sequence is clinically predictable: peripheral insulin resistance develops first, primarily in skeletal muscle. Compensatory hyperinsulinemia follows. Hepatic fat accumulates through the three pathways described. Hepatic insulin resistance develops via the DAG–PKCε loop. The liver begins producing glucose autonomously, raising fasting glucose. Eventually beta cell compensation fails and frank type 2 diabetes emerges.
Every stage of this sequence except the final one is fully reversible. The liver has substantial regenerative capacity, and hepatic steatosis — in the absence of established fibrosis — responds rapidly to interventions that reduce hyperinsulinemia and restore insulin sensitivity. Reductions in liver fat of 30–40% within 8–12 weeks have been documented with low-carbohydrate dietary interventions in controlled trials, driven primarily by the reduction in insulin levels rather than caloric restriction alone.
This is why the fasting insulin level is not merely a diabetes screening tool. It is a window into the full cascade — from adipose tissue dysfunction through hepatic fat accumulation to the insulin resistance feedback loop — that standard metabolic screening is structurally blind to.
For the foundational explanation of hyperinsulinemia and what it does across organ systems, the hyperinsulinemia post provides the broader clinical context for everything described here.
A Note on Uncertainty
The mechanisms described in this article — adipose FFA spillover, SREBP-1c-mediated DNL, and DAG–PKCε-mediated hepatic insulin resistance — are among the best-characterized pathways in metabolic research and have been validated across human tracer studies, hepatic biopsy data, and controlled dietary interventions. They are not speculative.
What remains more variable is the relative contribution of each pathway in any given individual. In some, adipose FFA spillover dominates. In others, elevated DNL driven by high carbohydrate intake and hyperinsulinemia is the primary driver. In practice, both are typically present to varying degrees, and the clinical approach — reducing insulin load, improving adipose tissue sensitivity, rebuilding muscle mass — addresses all three pathways simultaneously.
Next Steps
If you have elevated fasting insulin, a rising triglyceride-to-HDL ratio, increasing waist circumference, or any of the early insulin resistance signals described across this cluster — the appropriate next step is a structured metabolic assessment that integrates these markers into a full mechanistic picture.
Standard screening will not find this. It is not designed to. The tools that detect the insulin-fatty liver connection early are metabolic markers, interpreted against physiologically meaningful thresholds — not laboratory reference ranges built from populations where insulin resistance is already the norm.
The liver does not accumulate fat randomly. It accumulates fat because the hormonal and metabolic signals driving that accumulation have not been identified and corrected. Identifying those signals is where intervention begins.
People Also Ask
Does insulin resistance cause fatty liver?
Yes — through three converging pathways. Adipose tissue insulin resistance drives chronic free fatty acid spillover to the liver. Compensatory hyperinsulinemia activates hepatic de novo lipogenesis via SREBP-1c. And elevated insulin simultaneously impairs hepatic fat oxidation. All three mechanisms are driven by insulin resistance and operate independently of total body weight.
Can you have fatty liver with normal liver enzymes?
Yes. ALT and AST reflect hepatocyte damage and inflammation — not fat accumulation or insulin signaling impairment. Multiple studies confirm that a significant proportion of individuals with confirmed hepatic steatosis on imaging have normal ALT values. Fasting insulin, HOMA-IR, and the triglyceride-to-HDL ratio are more sensitive early markers.
Why does the liver accumulate fat in insulin resistance?
The liver receives excess free fatty acids from insulin-resistant adipose tissue, produces more fat internally through hyperinsulinemia-driven de novo lipogenesis, and has reduced capacity to oxidize or export that fat. The combination of increased delivery, increased production, and decreased clearance creates inevitable triglyceride accumulation inside hepatocytes.
What is the DAG–PKCε mechanism?
Diacylglycerol (DAG) is a lipid intermediate that accumulates in hepatocytes during active fat flux. It activates protein kinase C epsilon (PKCε), which directly impairs insulin receptor signaling in the liver — creating hepatic insulin resistance. This is the molecular mechanism by which fatty liver causes its own insulin resistance, creating a self-reinforcing feedback loop.
How does fatty liver connect to type 2 diabetes?
Hepatic insulin resistance — driven by the DAG–PKCε mechanism — causes the liver to continue producing glucose autonomously even in the presence of insulin. This forces further compensatory insulin secretion, progressively strains beta cell function, and eventually contributes to the fasting glucose elevation that marks the transition from insulin resistance to prediabetes and type 2 diabetes.
Is fatty liver reversible?
In the absence of established fibrosis, yes — and often rapidly. Low-carbohydrate dietary interventions that directly reduce insulin levels have demonstrated 30–40% reductions in liver fat within 8–12 weeks in controlled trials. The liver’s regenerative capacity is significant when the upstream insulin and metabolic signals driving fat accumulation are corrected.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
- Donnelly KL, et al. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. Journal of Clinical Investigation. 2005;115(5):1343–1351.
- Jornayvaz FR, Shulman GI. Diacylglycerol activation of protein kinase Cε and hepatic insulin resistance. Cell Metabolism. 2012;15(5):574–584.
- Brown MS, Goldstein JL. Selective versus total insulin resistance: a pathogenic paradox. Cell Metabolism. 2008;7(2):95–96.
- Samuel VT, et al. Mechanism of hepatic insulin resistance in non-alcoholic fatty liver disease. Journal of Biological Chemistry. 2004;279(31):32345–32353.
- Fabbrini E, et al. Intrahepatic fat, not visceral fat, is linked with metabolic complications of obesity. PNAS. 2009;106(36):15430–15435.
- Reaven GM. Role of insulin resistance in human disease. Diabetes. 1988;37(12):1595–1607.
- Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiological Reviews. 2018;98(4):2133–2223.
- Luukkonen PK, et al. Saturated fat is more metabolically harmful for the human liver than unsaturated fat or simple sugars. Diabetes Care. 2018;41(8):1732–1739.
