Cortisol is just the beginning. Here is what happens downstream — and why fixing stress means fixing metabolism first.
Most people understand that stress affects hormones. What they do not understand is the mechanism — or the scale of the damage.
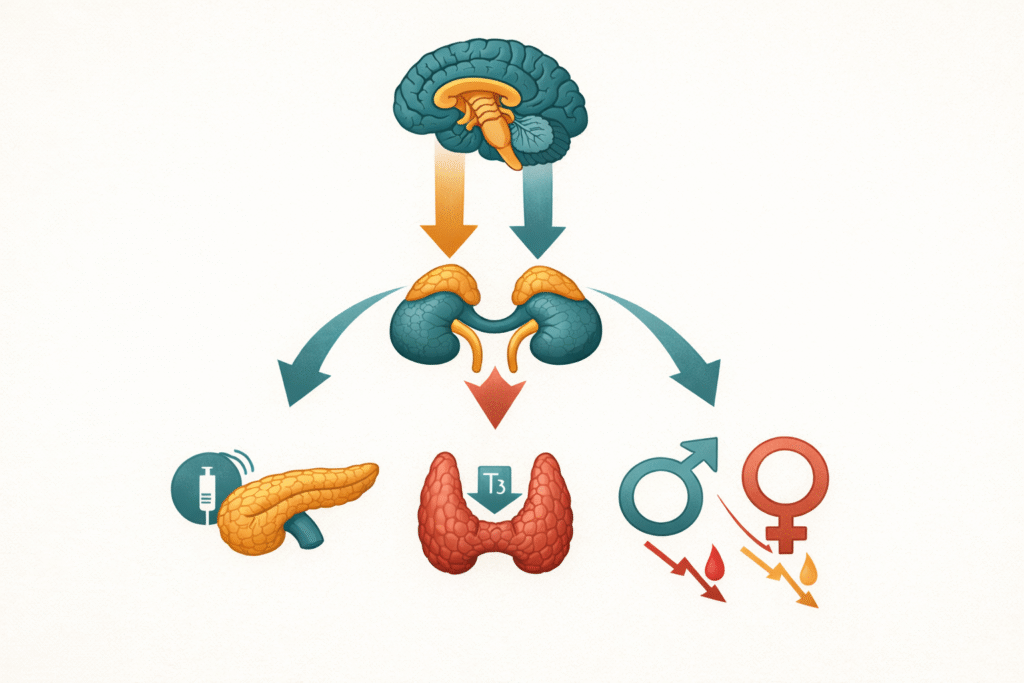
Stress and hormonal disruption are not loosely connected. They are causally linked through a cascade that begins in the HPA axis and ends in the thyroid, the reproductive system, the pancreas, and the liver. The downstream consequences are not vague or indirect. They are measurable, consistent, and in many patients, the primary driver of conditions that are being managed entirely at the wrong level.
This post explains what actually happens when the stress system chronically overactivates — how cortisol drives insulin, how insulin disrupts thyroid conversion, and how the entire hormonal hierarchy gets rerouted depending on whether you are a woman or a man. It explains the two phases of stress-driven dysfunction, which one causes more damage, and what the fastest clinical lever is for beginning to reverse it.
What you will learn:
The consistent lab signature behind stress-driven hormonal disruption | Why cortisol and insulin dysregulation is the core mechanism — not cortisol alone | The two clinical phases of HPA overactivation and which one does more damage | How chronic stress expresses differently in women versus men | The single intervention that starts to reset the system fastest | What the recovery timeline looks like when the root cause is addressed.
Why Stress Is a Metabolic Problem, Not Just a Psychological One
Stress and hormonal disruption are almost always framed as psychological — a matter of resilience, mindset, or workload management.
That framing misses the biology entirely.
Chronic stress is a metabolic event. When the HPA axis is chronically activated, cortisol rises — not in a single acute spike, but in a sustained, dysregulated pattern that reshapes how the liver handles glucose, how the pancreas responds with insulin, and how the thyroid converts its own hormones. Each of these downstream effects amplifies the others.
The result is not “feeling stressed.” The result is a metabolic environment that is actively driving fat accumulation, thyroid suppression, reproductive dysfunction, and inflammatory signaling — often years before a conventional panel flags anything as abnormal.
Stress is not a soft clinical variable. It is a primary upstream driver of insulin resistance, impaired thyroid conversion, and hormonal dysregulation. It belongs at the center of the metabolic picture — not as a footnote.
The Diagnostic Journey: What These Patients Have Been Through
The history these patients bring is consistent. They have usually been symptomatic for years before anything actionable is identified.
The early phase is almost invisible clinically. Energy is lower than it should be. Sleep is lighter. The ability to recover from demanding periods — a project, a difficult month, a life transition — is reduced. These symptoms are real but individually dismissible. A busy period. Aging. Stress at work.
Then the pattern consolidates. Weight accumulates centrally despite no meaningful change in diet. Sleep breaks at 2 or 3 in the morning — consistently, without explanation. Concentration becomes effortful. The craving for caffeine and sugar moves from preference to dependency.
Labs are checked. Glucose is normal. TSH is normal. The panel comes back unremarkable. The conversation ends with the same phrase these patients have heard repeatedly: probably stress. Come back if things get worse.
Things get worse. The labs still look normal. Because the markers being checked are downstream — and the upstream dysregulation driving the symptoms has not been measured.
By the time these patients arrive in this practice, they have often been symptomatic for five to ten years. They are not fragile or anxious individuals. They are typically high-functioning, driven, managing significant professional and personal demands. The symptom they almost always lead with is not weight or labs. It is this: I’m exhausted — but I can’t switch off.
That sentence is a clinical signal. It points directly to a dysregulated cortisol rhythm — not burnout in the colloquial sense, but a measurable pattern of HPA axis overactivation that has begun reshaping the metabolic environment.
Clinical Perspective: What I See in Practice
When a patient arrives presenting stress and hormonal disruption, the first thing I am looking for is not a single elevated marker. I am looking for a pattern — because the individual numbers are often within conventional ranges while the functional picture is clearly abnormal.
The most consistent lab signature I see is this: cortisol dysregulation — not necessarily high in absolute terms, but with a flattened or inverted rhythm, often elevated in the evening when it should be declining. Alongside this, fasting insulin is elevated early — often well above my functional threshold of 5 µIU/mL — even when fasting glucose remains perfectly normal. And in the thyroid panel, when it is actually run in full: TSH normal, free T3 low or low-normal, reverse T3 elevated.
These three abnormalities are not independent findings. They are expressions of the same upstream driver.
Chronic cortisol signaling drives hepatic glucose output. The liver releases more glucose in response to cortisol. The pancreas responds with more insulin. That elevated insulin, combined with the cortisol itself, suppresses the deiodinase enzyme activity responsible for converting T4 to active T3 — and simultaneously increases the conversion toward reverse T3, the inactive competing form that acts as a metabolic brake.
The patient looks normal on a standard panel. They have a TSH of 2.1 and a fasting glucose of 89. What they actually have is a cortisol-insulin-thyroid loop that is suppressing their metabolic output, accumulating visceral fat, disrupting their sleep, and impairing their cognitive function. None of that is visible without the right questions and the right markers.
The typical patient this pattern belongs to is between 35 and 55 years old. More frequently a woman — though men present with the same physiology, typically later and with a different downstream expression. They are high-performing and chronically loaded: work, family, or both simultaneously. They often describe doing everything right on paper — eating reasonably, exercising, not drinking — while feeling consistently below capacity. Their recovery is inadequate. Sleep is light and non-restorative. Caffeine is doing the work that cortisol should be doing in the morning.
When I explain what is happening mechanistically, the response is almost always the same. Not resistance. Relief. Because what they have been told for years — that this is stress, that it is lifestyle, that they need to relax — has never explained the biology. The explanation that lands is the accurate one: your stress system has been chronically activated, and it has rerouted your hormonal hierarchy. This is not a psychological problem with physical symptoms. It is a metabolic problem with a neurological origin.
The sex-specific dimension of this pattern is clinically important and consistently underappreciated. In women, chronic HPA overactivation combined with insulin elevation converges on ovarian function. Insulin drives ovarian androgen production upward. The cortisol-insulin signaling disrupts ovulation and weakens the luteal phase, reducing progesterone output and producing a state of relative estrogen dominance — even without elevated estrogen in absolute terms. PCOS-like patterns emerge frequently in this population, sometimes without a formal PCOS diagnosis. The cycle becomes the first clinical signal that the system is under load.
In men, the downstream expression is different. Cortisol dominance suppresses the HPG axis — hypothalamic, pituitary, and gonadal signaling all decline together. Testosterone drops, often with normal LH and FSH early on, meaning the conventional male hormone panel misses it. Visceral fat accumulates. Drive, motivation, and recovery — the functional markers of androgen adequacy — decline before any lab confirms it. The mechanism is the same: HPA overactivation, insulin dysregulation, hormonal suppression. The organ system expressing the disruption is different.
Same root. Different downstream target. Sex-specific physiology determines where the system breaks first.
The HPA Axis: Where Stress Enters the Metabolic System
The hypothalamic-pituitary-adrenal axis is the body’s primary stress response system. When a stressor — physical, psychological, or metabolic — is perceived, the hypothalamus releases corticotropin-releasing hormone (CRH). The pituitary responds with ACTH. The adrenal cortex responds with cortisol.
In an acute stress event, this is adaptive. Cortisol mobilizes glucose, sharpens focus, suppresses non-essential functions, and prepares the body for action. The event passes. Cortisol declines. The system resets.
In chronic stress, the event does not pass. The HPA axis remains activated. Cortisol remains elevated — or, as the system progresses, becomes dysregulated, losing its diurnal rhythm. The morning peak flattens. The evening decline fails to occur. The system that was designed for short-term mobilization is now operating in continuous output mode.
This sustained activation initiates three overlapping metabolic disruptions. First, cortisol drives hepatic glucose production continuously — not because energy is needed, but because the stress signal has not cleared. Fasting glucose rises modestly. Fasting insulin rises more significantly, because the pancreas is compensating for the cortisol-driven glucose load. This is the early insulin resistance signature — elevated fasting insulin with normal glucose — that the standard panel does not detect because fasting glucose is checked and fasting insulin is not.
Second, cortisol directly suppresses the immune tolerance mechanisms that regulate autoimmune activity and gut barrier integrity. Chronic cortisol impairs tight junction proteins, increases intestinal permeability, and creates the gut-immune axis disruption described in the Hashimoto’s post — a relevant pathway for patients in whom stress is a trigger for autoimmune flare.
Third, cortisol directly alters thyroid metabolism. The deiodinase enzymes responsible for converting T4 to active T3 are cortisol-sensitive. Chronic cortisol elevation suppresses type 1 and type 2 deiodinase activity, reducing T3 output. Simultaneously, it upregulates type 3 deiodinase, which converts T4 to reverse T3 rather than active T3. The result is a predictable thyroid pattern: normal TSH, low free T3, elevated reverse T3 — and a patient who is metabolically hypothyroid with a normal standard panel.
The full conversion model — and the five functional patterns through which thyroid dysfunction presents when this suppression is chronic — is detailed in the thyroid metabolism framework.
The Two Phases: Wired and Flat — and Which One Does More Damage
Stress-driven hormonal disruption does not present as a single clinical picture. It presents in two distinct phases that are stages of the same progression, not separate conditions.
The first phase is the wired state. Cortisol is dysregulated but often elevated — particularly in the evening, when it should be declining. Sleep is disrupted: the patient wakes between 2 and 4 AM, cannot return to sleep easily, and rises unrefreshed. They are anxious, hyperalert, and cognitively active in ways that feel uncontrolled. They are still functioning — this is when most patients seek help, because the system is loud but not yet collapsed.
This is also the phase that causes the bulk of the downstream metabolic damage.
In the wired phase, cortisol is actively driving hepatic glucose output and compensatory insulin elevation. Insulin resistance is building. Visceral fat is accumulating. Thyroid conversion is being actively suppressed. The inflammatory environment is being maintained. Sleep disruption — driven by the elevated evening cortisol — worsens insulin resistance independently, creating a second pathway of damage that compounds the first.
The second phase is the flat or burned-out state. After sustained HPA overactivation, the system begins to downregulate. Cortisol output flattens. The morning peak disappears or becomes inadequate. The patient is no longer anxious and wired — they are simply exhausted, with no stress response available. Drive, motivation, and emotional resilience are all reduced.
The flat phase is less metabolically active but harder to reverse. The system has adaptively shut down. Thyroid output is suppressed not by active cortisol interference but by a downregulated metabolic state. Energy production is globally reduced. The restoration path is longer because the system is not overactivated — it is depleted.
Most patients presenting with stress and hormonal disruption are in the wired phase, or transitioning between the two. Identifying which phase they are in shapes the intervention sequence — because the wired patient needs cortisol rhythm restoration and metabolic stabilization, while the flat patient needs rebuilding rather than calming.
Insulin Resistance as the Bridge Between Stress and Hormonal Disruption
The connection between stress, cortisol, and insulin is the mechanism that most clinical frameworks underemphasize.
Cortisol is insulin-antagonizing. Every time cortisol rises — in response to a psychological stressor, a poor night of sleep, a blood sugar crash, or a demanding workday — it drives hepatic glucose production, raises circulating glucose, and forces a compensatory insulin response. Repeat this cycle daily, for months or years, and the result is chronic hyperinsulinemia — elevated fasting insulin with a rising HOMA-IR and a worsening TG/HDL ratio — even in patients who eat reasonably and exercise regularly.
This is clinically critical: insulin resistance in a chronically stressed patient is not always dietary in origin. It can be predominantly cortisol-driven. A patient following a clean diet and maintaining exercise discipline can develop significant insulin resistance through the cortisol-glucose-insulin loop alone — particularly if sleep is impaired, recovery is inadequate, and the mental load is sustained.
The elevated insulin then feeds back into every other hormonal disruption. It suppresses sex hormone-binding globulin, altering the free hormone environment. It drives ovarian androgen production in women, contributing to the PCOS-like pattern described in the PCOS and insulin resistance post. It suppresses testosterone in men through direct effects on gonadotropin signaling. It worsens thyroid conversion by maintaining the inflammatory environment that cortisol initiated.
Insulin resistance is not a downstream consequence of stress-driven hormonal disruption. It is the bridge through which stress dysregulates every other hormonal system.
How Stress Expresses Differently in Women and Men
The same upstream driver — HPA overactivation and insulin dysregulation — produces different downstream expressions depending on sex-specific physiology.
In women, the hormonal disruption concentrates in reproductive function first. Chronic cortisol and insulin signaling converge on ovarian physiology. Elevated insulin drives ovarian androgen production upward. The cortisol-driven disruption of LH pulsatility impairs ovulation and weakens the luteal phase. Progesterone output declines. The result is relative estrogen dominance — not because estrogen is necessarily elevated in absolute terms, but because progesterone is insufficient to balance it. The pattern resembles PCOS, often without a formal diagnosis. Cycle irregularity, luteal phase spotting, worsening PMS, and central weight gain are the clinical signals.
The estrogen dominance picture that develops in this context — described in detail in the estrogen dominance post — feeds back further on thyroid function. Elevated estrogen increases thyroid-binding globulin production in the liver. More TBG means more sequestration of thyroid hormone, reducing free T3 availability at the tissue level. A stressed woman with insulin elevation and relative estrogen dominance is running a multilayered thyroid suppression that has nothing to do with primary thyroid gland failure.
In men, the expression is different but the mechanism is identical. Chronic cortisol dominance suppresses the HPG axis at the hypothalamic level. GnRH pulsatility is reduced. LH and FSH decline, or remain normal early while testosterone production is already falling. The testosterone decline presents clinically as reduced drive, motivation, and recovery capacity — the performance markers that decline before any lab flags it. Visceral fat accumulates, and the aromatase activity in that fat tissue converts testosterone to estrogen, compounding the testosterone deficit.
Men in this state are not deficient in testosterone because their testes are failing. They are deficient because their stress system is suppressing the axis that instructs testosterone production. Address the cortisol-insulin loop, and testosterone often recovers — without exogenous supplementation.
What Changes When the Upstream Drivers Are Corrected
The recovery timeline when stress-driven hormonal disruption is genuinely addressed follows a consistent and predictable sequence.
The fastest lever — and the one that begins to shift the cortisol-insulin loop most reliably — is stabilizing blood sugar early in the day. The first meal is the critical intervention point. A protein-and-fat-dominant breakfast eliminates the cortisol-driven glucose spike that sets the metabolic tone for the entire day. It reduces the compensatory insulin demand. It sends a safety signal to the HPA axis — a physiological cue that fuel is available and abundant — and begins to downshift the stress-state activation.
This is not a minor lifestyle adjustment. It is a direct interruption of the cortisol-glucose-insulin cycle. In patients who make this change consistently, the first signal they notice is almost always the same: I feel more stable. Not weight loss. Not immediate energy improvement. Stability — fewer crashes, reduced cravings for sugar and caffeine, sleep beginning to deepen within one to two weeks.
Between weeks four and eight, the deeper metabolic shift becomes apparent. Fasting insulin begins to fall. The TG/HDL ratio improves. hsCRP declines as the inflammatory load reduces. Brain fog lifts. The 2–4 AM waking pattern — one of the most reliable signals of cortisol dysregulation — begins to resolve as the evening cortisol spike that was disrupting sleep reduces. Mood stabilizes, not through psychological intervention but through the normalization of the metabolic signaling that was driving it.
Between weeks eight and sixteen, hormonal recovery becomes measurable. In women, cycle regularity begins to improve as luteal phase progesterone output recovers. The estrogen dominance pattern softens as insulin declines and SHBG rises. In men, testosterone begins to recover as HPG axis suppression lifts. Free T3 rises in the thyroid panel as conversion efficiency improves in the now less-inflamed, lower-cortisol environment. Reverse T3 falls.
Beyond three to six months, what patients describe is a fundamental shift in how they relate to load. Not the absence of stress — but the restoration of resilience. The capacity to absorb demand and recover from it. The system is no longer in continuous activation mode. It is responsive rather than reactive.
A Note on Uncertainty
The framework described here — HPA axis overactivation as a primary driver of insulin resistance, thyroid conversion impairment, and sex-specific reproductive hormone disruption — is supported by a consistent and growing body of mechanistic research. What remains less clearly established in large randomized trial data is the precise relative contribution of cortisol dysregulation versus dietary factors in any individual patient, and the proportion of patients in whom cortisol-driven insulin resistance fully normalizes through lifestyle intervention alone.
Cortisol rhythm assessment using salivary cortisol at multiple time points is not standard clinical practice and is not universally available. The functional patterns described in this post — particularly the wired-to-flat progression — are clinically well-recognized but are not formal diagnostic categories in conventional endocrinology.
All decisions regarding hormone therapy — thyroid medication, sex hormone supplementation, or adrenal support protocols — must be made in collaboration with the prescribing physician. The approach described here addresses upstream metabolic and lifestyle drivers. It does not replace medical management of confirmed endocrine conditions.
Practical Implications
If you are chronically stressed — carrying a sustained mental load, sleeping poorly, waking in the early hours, feeling tired but unable to switch off — the hormonal consequences described in this post are not theoretical. They are measurable, and they are likely already operating.
The practical starting point is a full metabolic assessment: fasting insulin, HOMA-IR, hsCRP, and the TG/HDL ratio. Alongside this: a complete thyroid panel including free T3 and reverse T3, and — in women — a full reproductive hormone panel timed to the luteal phase. In men: total and free testosterone, SHBG, and LH.
If fasting insulin is elevated and free T3 is low with normal TSH, the cortisol-insulin-thyroid loop is already active. The intervention begins at the metabolic level: stabilize blood sugar, prioritize protein at the first meal, protect sleep, and reduce the cortisol-driven glucose output that is feeding the whole cycle.
The stress is real. The hormonal disruption it causes is real. And both are addressable — not through stress management advice, but through correcting the metabolic environment that chronic stress has created.
Chronic stress doesn’t just raise cortisol. It reroutes your entire hormonal hierarchy. Change the metabolic environment — and the system begins to find its way back.
The relationship between stress and hormonal disruption is one expression of a broader principle — that hormones reflect the metabolic environment they operate within, and restoring the environment is often what restores the hormonal signaling.
People Also Ask
How does chronic stress cause hormonal disruption?
Chronic stress activates the HPA axis, producing sustained cortisol elevation or rhythm dysregulation. Cortisol drives hepatic glucose output, which forces compensatory insulin release. Elevated insulin and cortisol together suppress thyroid conversion — reducing active T3 and raising reverse T3. In women, the cortisol-insulin signal disrupts ovarian function, reduces progesterone, and creates relative estrogen dominance. In men, it suppresses the HPG axis and reduces testosterone. The mechanism is a cascade — not cortisol alone, but the entire metabolic and hormonal environment cortisol dysregulation creates.
What are the signs of stress-driven hormonal disruption?
The most consistent clinical signs are fatigue that sleep does not resolve, waking between 2 and 4 AM, central fat accumulation despite dietary effort, brain fog, and low mood without clear emotional cause. In women, cycle irregularity, worsening PMS, and luteal phase weakness are common. In men, reduced drive, motivation, and recovery capacity are the early signals. Lab findings include elevated fasting insulin with normal fasting glucose, low free T3 with normal TSH, and a rising TG/HDL ratio.
Can stress cause insulin resistance?
Yes — directly. Cortisol is insulin-antagonizing. Every sustained cortisol elevation drives hepatic glucose production and forces a compensatory insulin response. In chronically stressed patients, this cycle runs continuously, producing elevated fasting insulin and progressing insulin resistance independent of dietary factors. Patients with clean diets and regular exercise can develop significant insulin resistance through the cortisol-glucose-insulin loop alone — particularly when sleep is impaired and mental load is sustained.
How does stress affect thyroid function?
Chronic cortisol suppresses the deiodinase enzymes that convert T4 to active T3, and upregulates type 3 deiodinase which converts T4 to reverse T3 instead. The result is a predictable pattern: normal TSH, low or low-normal free T3, elevated reverse T3. The thyroid gland is not failing. The conversion environment is impaired by cortisol and insulin signaling. Standard TSH testing does not detect this pattern — which is why patients remain symptomatic with normal labs.
Does stress affect testosterone in men?
Yes. Chronic HPA overactivation suppresses the HPG axis — the hypothalamic-pituitary-gonadal system responsible for testosterone production. GnRH pulsatility decreases, LH and FSH decline or remain falsely normal, and testosterone production falls. Visceral fat accumulation — driven by the same cortisol-insulin loop — adds aromatase-driven conversion of testosterone to estrogen. Men in this state are not failing to produce testosterone. Their stress system is suppressing the signal that instructs production.
How long does it take for hormones to normalize after reducing chronic stress?
The timeline follows a consistent pattern in clinical practice. Blood sugar and energy stabilization within one to two weeks of correcting the first meal and dietary quality. Measurable improvements in fasting insulin, TG/HDL ratio, and inflammatory markers between weeks four and eight. Thyroid panel improvements — rising free T3, falling reverse T3 — between weeks eight and sixteen. Reproductive hormone recovery in women and testosterone normalization in men typically between three and six months, depending on the depth of prior dysregulation and the completeness of the metabolic correction.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Tsigos C, Chrousos GP. Hypothalamic-pituitary-adrenal axis, neuroendocrine factors and stress. Journal of Psychosomatic Research. 2002;53(4):865–871. 🔗 https://pubmed.ncbi.nlm.nih.gov/12377295/
2. Björntorp P, Rosmond R. Obesity and cortisol. Nutrition. 2000;16(10):924–936. 🔗 https://pubmed.ncbi.nlm.nih.gov/11054598/
3. Kyrou I, Tsigos C. Stress hormones: physiological stress and regulation of metabolism. Current Opinion in Pharmacology. 2009;9(6):787–793. 🔗 https://pubmed.ncbi.nlm.nih.gov/19758844/
4. Ranabir S, Reetu K. Stress and hormones. Indian Journal of Endocrinology and Metabolism. 2011;15(1):18–22. 🔗 https://pubmed.ncbi.nlm.nih.gov/21584161/
5. Bianco AC, Kim BW. Deiodinases: implications of the local control of thyroid hormone action. Journal of Clinical Investigation. 2006;116(10):2571–2579. 🔗 https://pubmed.ncbi.nlm.nih.gov/17016550/
6. Gleason CE, Carlsson CM, Johnson SC, et al. Insulin resistance and hippocampal volume in women at risk for Alzheimer’s disease. Neurobiology of Aging. 2011;32(3):506–514. 🔗 https://pubmed.ncbi.nlm.nih.gov/19446936/
7. Moyer AE, Rodin J, Grilo CM, et al. Stress-induced cortisol response and fat distribution in women. Obesity Research. 1994;2(3):255–262. 🔗 https://pubmed.ncbi.nlm.nih.gov/16353426/
8. Pasquali R, Vicennati V, Cacciari M, Pagotto U. The hypothalamic-pituitary-adrenal axis activity in obesity and the metabolic syndrome. Annals of the New York Academy of Sciences. 2006;1083:111–128. 🔗 https://pubmed.ncbi.nlm.nih.gov/17148736/
9. Whirledge S, Cidlowski JA. Glucocorticoids, stress, and fertility. Minerva Endocrinologica. 2010;35(2):109–125. 🔗 https://pubmed.ncbi.nlm.nih.gov/20595939/
10. Cumming DC, Quigley ME, Yen SS. Acute suppression of circulating testosterone levels by cortisol in men. Journal of Clinical Endocrinology & Metabolism. 1983;57(3):671–673. 🔗 https://pubmed.ncbi.nlm.nih.gov/6348068/
11. Epel ES, McEwen B, Seeman T, et al. Stress and body shape: stress-induced cortisol secretion is consistently greater among women with central fat. Psychosomatic Medicine. 2000;62(5):623–632. 🔗 https://pubmed.ncbi.nlm.nih.gov/11020089/
12. Raber J. Detrimental effects of chronic hypothalamic-pituitary-adrenal axis activation. Molecular Neurobiology. 1998;18(1):1–22. 🔗 https://pubmed.ncbi.nlm.nih.gov/9824848/