Thyroid Metabolism is more than TSH: how thyroid hormones are produced, converted, and used — and why the standard panel misses most of what matters.
When a patient comes in with classic hypothyroid symptoms — persistent fatigue that sleep does not fix, unexplained weight gain, cold intolerance, brain fog, sluggish digestion, hair thinning — and their doctor tells them their thyroid is fine because their TSH is normal, something important has been missed. Not necessarily by carelessness. By a diagnostic framework that measures one control signal and concludes from it that the entire system is functioning. It is like checking whether the thermostat is set correctly and concluding from that alone that every room in the building is warm.
TSH — thyroid stimulating hormone — is a pituitary output. It tells you what the brain is signaling to the thyroid. It does not tell you how much active thyroid hormone is actually available at the cellular level, whether the conversion from inactive to active hormone is working correctly, whether binding proteins are sequestering hormone away from tissues, whether the body is producing a competing inactive form that blocks the active one at the receptor, or whether the cells receiving thyroid hormone are actually responding to it. A normal TSH rules out primary thyroid failure. It does not confirm that thyroid metabolism is functioning optimally. Those are different questions, and the standard panel only answers the first one.
Understanding thyroid metabolism — the full chain from hormone production through conversion, transport, and cellular action — is the prerequisite for understanding why so many people experience a hypothyroid picture with normal standard labs, and why the metabolic drivers of that picture respond to upstream intervention rather than thyroid hormone replacement alone.
What you will learn: What TSH actually measures — and what it does not | The T4 to T3 conversion pathway and the multiple points at which it can fail | What reverse T3 is and why it matters clinically | How thyroid-binding globulin determines free hormone availability | The nutrient cofactors that the thyroid system depends on | How insulin resistance, cortisol, and inflammation disrupt thyroid metabolism at multiple levels simultaneously
What the Standard Panel Actually Measures — and What It Misses
The standard thyroid panel in most clinical settings consists of a single measurement: TSH. In more thorough workups, free T4 is added. In functional medicine practice, this is the minimum starting point, not the complete picture.
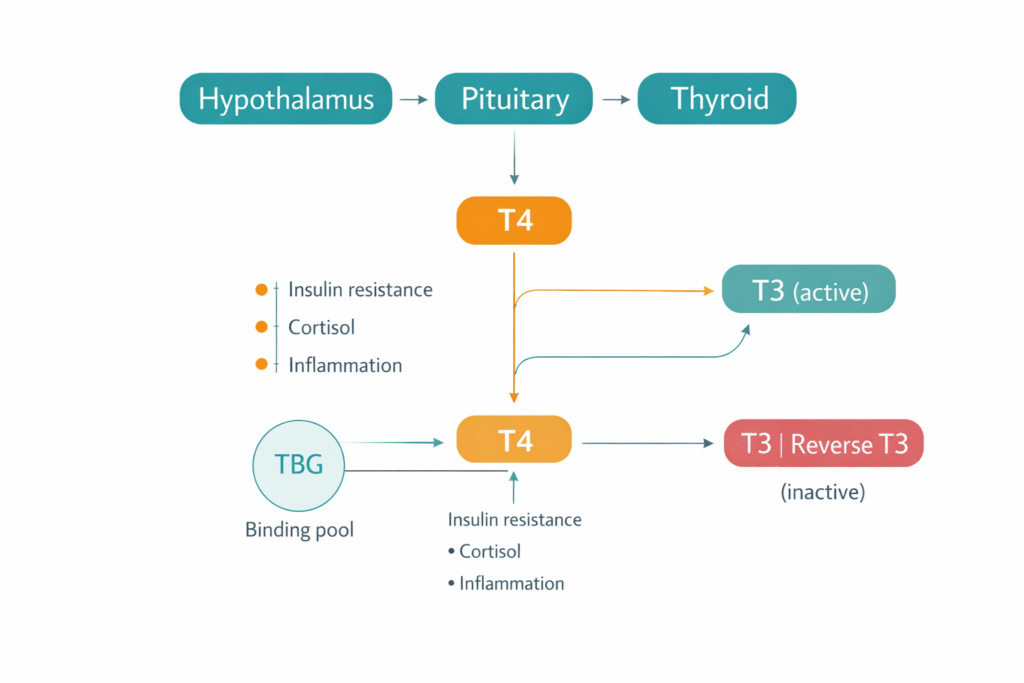
TSH is produced by the pituitary gland in response to signals from the hypothalamus — specifically thyrotropin-releasing hormone (TRH). When the hypothalamus detects that circulating thyroid hormone levels are low, it releases TRH, which prompts the pituitary to release TSH, which travels to the thyroid gland and stimulates it to produce thyroid hormones. This is the hypothalamic-pituitary-thyroid axis — a feedback loop designed to maintain thyroid hormone levels within a functional range.
What TSH reflects, therefore, is the pituitary’s assessment of circulating thyroid hormone levels. It is a control signal, not a tissue-level measurement. A normal TSH tells you that the pituitary is not detecting a significant deficit in circulating thyroid hormone. It does not tell you whether the hormone it is detecting is the active form capable of driving cellular metabolism, whether that hormone is bound to proteins that prevent it from reaching tissues, whether peripheral conversion from inactive to active hormone is working efficiently, or whether cells are responding normally to the hormone signal they are receiving.
The clinically meaningful thyroid panel — the minimum required to actually assess thyroid metabolism rather than pituitary signaling alone — includes TSH, free T4, free T3, reverse T3, and thyroid antibodies (TPO and thyroglobulin). Without free T3 and reverse T3, you are blind to the actual metabolic output of the thyroid system. You are assessing the instruction without examining whether it was executed. This gap explains a significant proportion of the patients who present with genuine hypothyroid symptoms and have been told, sometimes for years, that their thyroid is normal.
Clinical Perspective: What I See in Practice
When a patient arrives with a folder of thyroid results and a history of being told everything looks fine, the first thing I do is look at what was actually measured — because in the majority of cases, it is TSH alone, or TSH with total T4. Free T3 is almost never included. Reverse T3 is almost never ordered. Antibodies are checked inconsistently, and TBG is rarely on the panel at all.
What I see when the full picture is finally assembled is consistent across a wide range of patient presentations. TSH is often normal — sometimes even on the lower end of normal, which in a pattern of pituitary suppression by elevated cortisol can actually indicate underactivation of the thyroid axis rather than normal function. Free T4 may be adequate. But free T3 is low or low-normal. And reverse T3 is elevated — sometimes significantly. The patient has sufficient inactive hormone circulating. What they do not have is efficient conversion of that hormone into its active form, or efficient delivery of the active form to the cells that need it.
These are not thyroid failure cases. They are thyroid metabolism cases — and that distinction changes everything about the intervention. A patient whose hypothyroid symptoms reflect impaired T4 to T3 conversion driven by insulin resistance, chronic inflammation, and cortisol dysregulation does not have a thyroid problem in the primary sense. They have a metabolic environment that is suppressing the conversion and signaling steps that make thyroid hormone functional. Replace the hormone without addressing the conversion impairment, and the replacement T4 faces the same metabolic obstacles as the endogenous T4 did.
I also see a specific pattern in women with elevated estrogen — whether from oral contraceptives, HRT, or the estrogen dominance picture described in other posts — where TBG is elevated, sequestering thyroid hormone away from tissues and producing a hypothyroid symptom picture despite normal free T4 and TSH. And the inverse in women with PCOS and elevated testosterone, where low TBG creates excess free hormone that paradoxically leads to receptor resistance and the same hypothyroid symptoms through a completely different mechanism. The symptoms can be identical. The lab patterns, the drivers, and the interventions are completely different. You cannot distinguish them without the full panel.
The nutrient picture is equally consistent. Low selenium — from diets relying on plant proteins with minimal seafood or organ meat — impairs the deiodinase enzyme activity that drives T4 to T3 conversion. Low zinc — from low red meat intake and high phytate diets that block mineral absorption — reduces hormone production and receptor sensitivity. Functional iron deficiency — often present in women with heavy periods and normalized by doctors because ferritin is within the conventional range — impairs thyroid peroxidase activity, the enzyme responsible for incorporating iodine into thyroid hormones. These are not exotic deficiencies. They are the predictable consequences of modern dietary patterns in a population that has been told to reduce animal foods.
The T4 to T3 Conversion Pathway: Where the System Most Often Fails
The thyroid gland produces thyroid hormones in two primary forms: thyroxine (T4) and triiodothyronine (T3). Despite the fact that T3 is the biologically active form — the hormone that enters cells, binds thyroid hormone receptors, and drives the metabolic processes that determine energy, temperature regulation, cognitive function, and tissue repair — over 90% of what the thyroid secretes is T4. T4 is essentially a prohormone: biologically inactive until it is converted to T3 by the removal of one iodine atom. This conversion is not a passive process. It is an active, energy-dependent enzymatic step that occurs primarily in the liver, kidneys, and peripheral tissues, catalyzed by a family of enzymes called deiodinases.
The deiodinase enzymes — specifically type 1 (D1) and type 2 (D2) deiodinase — are selenium-dependent. Without adequate selenium, their activity is impaired, and conversion from T4 to T3 falls. This is one of the most direct and well-characterized nutrient-thyroid interactions in metabolic medicine, and it explains why selenium deficiency produces a functional hypothyroid picture even when the thyroid gland itself is completely intact and producing normal amounts of T4.
Beyond selenium, the conversion pathway is vulnerable to disruption by several metabolic conditions that are increasingly prevalent in the modern clinical population. Chronic inflammation — driven by insulin resistance, gut dysbiosis, ultra-processed food consumption, and visceral adiposity — produces inflammatory cytokines including TNF-alpha and IL-6 that directly suppress deiodinase activity.
Elevated cortisol, as described in the cortisol dysregulation post, inhibits both D1 and D2 activity and simultaneously increases the activity of D3 — the deiodinase that converts T4 not into active T3 but into reverse T3, the inactive competing form. Liver dysfunction — from fatty liver driven by de novo lipogenesis and insulin resistance-driven hepatic damage — impairs the primary peripheral conversion site. Caloric restriction and significant metabolic stress shift the conversion balance toward reverse T3 as an energy-conservation adaptation.
Insulin resistance deserves particular attention as a driver of conversion failure because its effects converge on the pathway through multiple simultaneous mechanisms. Chronic hyperinsulinemia drives the low-grade inflammation that impairs deiodinase enzyme activity. It disrupts mitochondrial function and lowers the cellular energy availability that the conversion process requires. And it increases oxidative stress, which shifts the enzymatic balance toward reverse T3 production. Insulin resistance does not merely coexist with impaired thyroid conversion — it actively drives it through three independent biological routes.
The clinical consequence is a patient who has adequate TSH signaling, adequate T4 production, but insufficient T3 delivery to tissues — presenting with the full hypothyroid symptom picture while their standard thyroid panel reports nothing abnormal.
Reverse T3: The Metabolic Brake
When the body converts T4, it has two options. It can remove iodine from the outer ring of the T4 molecule to produce T3 — the active form. Or it can remove iodine from the inner ring to produce reverse T3 (rT3) — a structurally similar but biologically inactive molecule that competes with T3 for binding at thyroid hormone receptors without activating them.
Reverse T3 is not simply a waste product. It is a physiologically regulated signal — the body’s deliberate downregulation of cellular metabolism in response to stress, inflammation, energy deficiency, or perceived threat. In evolutionary terms, when resources are scarce, injury is present, or survival is uncertain, reducing metabolic rate makes biological sense. The organism conserves fuel, reduces the energy cost of normal cellular function, and shifts resources toward immune activation and tissue repair. Reverse T3 is one of the mechanisms through which this conservation is enacted.
The problem arises when the stressors driving reverse T3 production are chronic rather than acute — when insulin resistance, chronic inflammation, cortisol dysregulation, and metabolic instability are persistent features of the patient’s biology rather than temporary responses to a specific threat. In this environment, reverse T3 production becomes chronically elevated, T3 receptor sites are chronically occupied by an inactive competitor, and the patient experiences the metabolic consequences of functional hypothyroidism regardless of what their TSH or T4 values show.
In clinical practice, the pattern is recognizable. TSH is normal. Free T4 is normal. Free T3 is low-normal. Reverse T3 is elevated. Classic hypothyroid symptoms are present: fatigue, cold extremities, cognitive slowing, difficulty with weight management, slow bowel transit, flat mood. The standard interpretation — thyroid is fine — misses the conversion and receptor-level problem entirely, because it does not measure the variables where the dysfunction is located.
Reverse T3 is not a thyroid hormone problem. It is a metabolic signal telling you that the system has shifted into conservation mode — and that the upstream drivers of that shift need to be identified and addressed.
Thyroid-Binding Globulin and the Free Hormone Question
Thyroid hormones in the circulation do not travel freely. The majority are bound to carrier proteins — primarily thyroid-binding globulin (TBG), with smaller amounts bound to transthyretin and albumin. Only the unbound, free fraction of thyroid hormone is biologically active and available for cellular uptake. This is why measuring total T4 or total T3 without assessing free fractions or TBG itself provides an incomplete and potentially misleading picture.
TBG levels are regulated by several hormonal inputs. Estrogen increases TBG production — meaning that women on oral contraceptives or estrogen-containing hormone replacement therapy, or women with the estrogen dominance picture driven by insulin resistance and low SHBG, may have elevated TBG that binds a higher proportion of their circulating thyroid hormone, reducing free hormone availability and producing hypothyroid symptoms despite normal total thyroid hormone levels and normal TSH. This is clinical pattern 3 — hypothyroidism due to elevated TBG — and it is almost never identified in standard practice because TBG is almost never measured.
The inverse situation — pattern 4 — occurs when TBG is low, as is seen in women with PCOS, elevated testosterone, and significant insulin resistance. Low TBG allows more free thyroid hormone to circulate, which in turn can lead to receptor downregulation and resistance. Despite having adequate or even elevated free hormone levels, cells become less responsive, and the same hypothyroid symptom picture emerges through a completely different mechanism. Addressing insulin resistance and the androgen excess it produces is the intervention — not thyroid hormone supplementation, which would worsen the free hormone excess without resolving the receptor problem.
This is the clinical complexity that makes thyroid assessment genuinely demanding: identical symptom presentations can reflect mechanistically opposite hormonal configurations, and treating them identically not only fails to help but can actively harm. The full panel — including TBG, free T3, free T4, and reverse T3 — is not excessive investigation. It is the minimum required to distinguish between these patterns.
The Five Functional Patterns of Thyroid Dysfunction
Beyond primary thyroid failure — where the gland itself is damaged or destroyed, as in advanced Hashimoto’s disease or post-surgical hypothyroidism — thyroid dysfunction in clinical practice presents in five distinct functional patterns, each with a different mechanism, a different lab fingerprint, and a different intervention requirement.
Pattern 1: Hypothyroidism due to pituitary dysfunction. The thyroid gland is structurally intact and capable of producing hormone, but the pituitary is not providing adequate stimulation. Elevated cortisol from chronic stress, blood sugar instability, insulin resistance, or HPA axis dysregulation suppresses hypothalamic TRH release and pituitary TSH output. The result is a TSH that falls within the broad conventional reference range but sits below the functional optimal of 1.8–3.0 µIU/mL — insufficient stimulation of a competent gland. T4 is low as a consequence. The intervention addresses the upstream HPA and metabolic drivers, not the thyroid.
Pattern 2: Impaired T4 to T3 conversion. TSH and T4 are normal. T3 is low. Reverse T3 may be elevated. The gland is producing hormone; the peripheral conversion machinery is impaired by inflammation, cortisol, liver dysfunction, selenium deficiency, or insulin resistance. This is the most common functional pattern in metabolically dysregulated patients and the one most consistently missed by standard panels that do not include free T3 or reverse T3.
Pattern 3: Hypothyroidism due to elevated TBG. TSH and T4 appear normal, but elevated TBG — driven by high estrogen from oral contraceptives, HRT, or metabolic estrogen dominance — is sequestering thyroid hormone and reducing free availability. T3 uptake is low. Free T3 is reduced despite adequate total hormone. The intervention addresses the estrogen excess and its metabolic drivers.
Pattern 4: Hypothyroidism due to decreased TBG. The inverse pattern — low TBG from high testosterone in PCOS or severe insulin resistance increases free hormone but drives receptor resistance. TSH and T4 appear normal, T3 is elevated, T3 uptake is high, but the patient is symptomatically hypothyroid. Addressing the insulin resistance and androgen excess is the primary intervention.
Pattern 5: Thyroid hormone resistance. The most diagnostically challenging pattern. All standard markers appear entirely normal because the dysfunction is at the receptor level — not in hormone production, not in conversion, not in binding, but in cellular responsiveness to the hormone signal. Thyroid resistance is associated with chronic cortisol elevation, elevated homocysteine, and genetic polymorphisms affecting thyroid receptor sensitivity. Current standard laboratory testing does not assess receptor function. The clinical picture — persistent hypothyroid symptoms with a completely normal full thyroid panel — is the primary diagnostic signal.
The Nutrient Foundation: What the Thyroid System Requires
Thyroid hormone synthesis, conversion, and receptor function all depend on specific micronutrients whose adequacy is determined primarily by dietary pattern. In clinical practice, the patients with impaired thyroid metabolism are almost never deficient in a single nutrient. They present with a low-density, high-stress metabolic environment that disables the whole system simultaneously.
Selenium is the most critical conversion cofactor. The deiodinase enzymes are selenoproteins — their function is directly proportional to selenium availability. Selenium deficiency impairs T4 to T3 conversion, increases oxidative stress in thyroid tissue, and worsens the impact of iodine imbalance. The dietary patterns producing selenium deficiency in the clinical population are consistent: low seafood and eggs intake, minimal or no organ meat consumption, and reliance on plant proteins from soils with variable selenium content.
Zinc is required for thyroid hormone synthesis and for the thyroid hormone receptor binding that allows cellular response. Zinc deficiency has been associated with reduced T3 production and impaired receptor sensitivity. The dietary drivers are plant-heavy diets high in phytates — compounds in grains, legumes, and seeds that bind zinc and other minerals and significantly reduce their bioavailability — combined with low red meat intake, which is the most bioavailable dietary zinc source.
Iron — specifically functional iron adequacy — is required for thyroid peroxidase (TPO) activity, the enzyme that incorporates iodine into thyroid hormones during synthesis. Iron deficiency impairs TPO function and reduces thyroid hormone production independently of iodine status. Functional iron deficiency is particularly common in women with heavy menstrual bleeding — a frequent consequence of the estrogen dominance picture — and is often missed when ferritin sits within the conventional reference range but falls below the functional optimal of 70–100 µg/L.
Iodine provides the structural backbone of both T4 and T3 — the numbers in their names reflect the number of iodine atoms they contain. Globally, iodine deficiency remains the leading cause of hypothyroidism. In Western populations with iodized salt and dietary variety, frank deficiency is less common, but erratic intake — combined with inadequate selenium to buffer the effects of iodine variability on thyroid tissue — can destabilize thyroid function. Excessive iodine intake without adequate selenium appears to increase the risk of autoimmune thyroid flare, particularly in Hashimoto’s patients — a nuance that makes supplementation without the full nutritional context potentially counterproductive.
The dietary pattern producing these deficiencies simultaneously is not difficult to identify: low animal protein intake, chronic caloric restriction, high reliance on refined plant foods with elevated phytate content, and the near-universal avoidance of organ meats and seafood that once provided the concentrated micronutrient density the thyroid system depends on.
How Insulin Resistance, Cortisol, and Inflammation Converge on Thyroid Metabolism
The thyroid system does not fail in isolation. In the majority of patients presenting with functional thyroid dysfunction — as opposed to primary autoimmune destruction — the impairment is the downstream consequence of a metabolic environment shaped by insulin resistance, cortisol dysregulation, and the chronic low-grade inflammation they both generate and sustain.
Insulin resistance drives thyroid dysfunction through the conversion impairment described above — via inflammation, mitochondrial dysfunction, and oxidative stress shifting conversion toward reverse T3. It also produces the hepatic dysfunction that impairs the liver’s role as the primary peripheral conversion site, and it drives the visceral adiposity that amplifies the inflammatory cytokine load suppressing deiodinase activity. In women, it generates the estrogen dominance and androgen excess patterns that dysregulate TBG and alter free hormone availability through patterns 3 and 4 respectively.
Cortisol dysregulation — almost always present alongside insulin resistance in the patients this practice sees, and almost always a downstream consequence of the same metabolic instability — independently suppresses hypothalamic TRH and pituitary TSH output, directly inhibits deiodinase enzymes, and actively promotes reverse T3 production. As described in the cortisol dysregulation post, cortisol is frequently the first adaptive layer — the metabolic instability comes first, the cortisol compensation follows, and the thyroid slowdown comes as the third downstream consequence of a system conserving energy in response to perceived chronic threat – a cascade explored in full in how chronic stress rewires the entire hormonal system.
The clinical timeline in most patients with functional thyroid dysfunction therefore runs: dietary and lifestyle-driven insulin resistance → compensatory hyperinsulinemia and metabolic instability → HPA dysregulation and chronic cortisol elevation → impaired T4 to T3 conversion, increased reverse T3, suppressed pituitary TSH output, and progressive thyroid hormone resistance. By the time the thyroid panel is ordered, the problem has been developing for years — and the panel, measuring only TSH, misses the mechanism entirely.
This is why the therapeutic approach to functional thyroid dysfunction begins upstream. Lower the insulin load. Reduce the inflammatory environment driving conversion impairment. Address the cortisol dysregulation suppressing hypothalamic-pituitary-thyroid signaling. Restore the nutrient cofactors the deiodinase enzymes require. In the majority of functional thyroid cases, the thyroid system does not need to be replaced. It needs the metabolic environment that has been impairing its function to be corrected.
A Note on Uncertainty
The five functional patterns described in this post represent clinically useful frameworks for organizing thyroid dysfunction beyond primary failure. They are not universally accepted diagnostic categories in conventional endocrinology, and the evidence base for some — particularly thyroid hormone resistance in the absence of genetic receptor mutations — relies more on clinical observation and mechanistic reasoning than on large randomized trial data.
Thyroid assessment and any decisions regarding thyroid hormone replacement or supplementation should involve a qualified physician. The functional thresholds described here — TSH optimal range 1.8–3.0, free T3 and reverse T3 interpretation — represent functional medicine perspectives that differ from conventional reference ranges and should be interpreted in clinical context, not applied in isolation. Environmental toxin exposure as a driver of thyroid dysfunction — covered in the Hashimoto’s post — adds a further layer of clinical complexity that requires individual assessment.
Practical Implications
If you have been told your thyroid is normal but you are experiencing persistent fatigue, cold intolerance, unexplained weight gain, brain fog, or hair thinning — and if your assessment consisted of TSH alone, or TSH with total T4 — the actual metabolic output of your thyroid system has not been evaluated.
The practical starting point is requesting a full thyroid panel: TSH, free T4, free T3, reverse T3, and TPO and thyroglobulin antibodies. Alongside this, fasting insulin and the TG/HDL ratio establish whether the insulin resistance driving conversion impairment is present. Ferritin, selenium, zinc, and vitamin D complete the picture of the nutrient environment in which thyroid metabolism is operating.
For a detailed account of what this presentation looks like clinically and why it takes years to identify, see what the full clinical picture of this dysfunction looks like in practice.
Your TSH is a pituitary signal. It tells you one thing about one part of the system. The thyroid metabolism that determines how you actually feel operates downstream of that signal — in the conversion, the binding, the delivery, and the cellular response — and that is where the clinical picture lives.
Thyroid biology operates within a larger hormonal-metabolic framework — for the unified clinical picture of how metabolic dysfunction distorts thyroid, estrogen, testosterone, cortisol, and reproductive hormone signaling, see the cornerstone on why many hormonal problems are actually metabolic problems.
People Also Ask
What is the difference between T4 and T3?
T4 (thyroxine) is the primary hormone produced by the thyroid gland, but it is biologically inactive — it is a prohormone that must be converted to T3 (triiodothyronine) to drive cellular metabolism. Over 90% of thyroid output is T4. Conversion to active T3 occurs primarily in the liver, kidneys, and peripheral tissues via selenium-dependent deiodinase enzymes. This conversion step is the most common point of failure in functional thyroid dysfunction and is not assessed by standard TSH-only panels.
Why is TSH alone not enough to assess thyroid function?
TSH is a pituitary output — it reflects the brain’s signaling to the thyroid, not the availability or activity of thyroid hormone at the cellular level. A normal TSH does not confirm adequate free T3 production, efficient T4 to T3 conversion, appropriate reverse T3 levels, or normal cellular responsiveness to thyroid hormone. Patients can have normal TSH and genuine functional hypothyroidism due to conversion impairment, TBG dysregulation, reverse T3 excess, or receptor resistance — none of which are visible on a TSH-only panel.
What is reverse T3 and why does it matter?
Reverse T3 is an inactive form of thyroid hormone produced when T4 is converted via the inner-ring deiodination pathway rather than the outer-ring pathway that produces active T3. It competes with T3 for binding at thyroid hormone receptors without activating them, effectively blocking thyroid hormone action at the cellular level. Reverse T3 production is upregulated by chronic stress, elevated cortisol, inflammation, insulin resistance, caloric restriction, and liver dysfunction — the same conditions that impair active T3 production. Elevated reverse T3 with normal TSH is a recognized pattern of functional hypothyroidism.
How does insulin resistance affect thyroid function?
Insulin resistance impairs thyroid metabolism through three simultaneous mechanisms: it drives chronic low-grade inflammation that suppresses deiodinase enzyme activity, disrupts mitochondrial function and reduces the cellular energy required for T4 to T3 conversion, and increases oxidative stress that shifts conversion toward reverse T3. It also produces the hepatic dysfunction that impairs the liver’s role as the primary conversion site, and in women generates hormonal patterns that dysregulate thyroid-binding globulin and free hormone availability.
What nutrients are essential for thyroid function?
The primary thyroid-supporting nutrients are selenium (required for deiodinase enzyme function and T4 to T3 conversion), zinc (required for hormone synthesis and receptor sensitivity), iron (required for thyroid peroxidase activity during hormone synthesis), and iodine (the structural component of both T4 and T3). These deficiencies rarely occur in isolation — they reflect the low micronutrient density, high phytate, low animal protein dietary pattern that is increasingly common in populations advised to reduce meat and seafood consumption.
What is thyroid-binding globulin and why does it matter?
Thyroid-binding globulin (TBG) is the primary carrier protein for thyroid hormones in the bloodstream. Only the unbound, free fraction of thyroid hormone is biologically active. When TBG is elevated — as occurs with high estrogen from oral contraceptives or metabolic estrogen dominance — more hormone is bound and unavailable to tissues, producing hypothyroid symptoms despite normal total thyroid hormone levels. When TBG is low — as occurs with high testosterone in PCOS and insulin resistance — excess free hormone can drive receptor resistance and paradoxically produce the same hypothyroid symptom picture through the opposite mechanism.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Bianco AC, Salvatore D, Gereben B, Berry MJ, Larsen PR. Biochemistry, cellular and molecular biology, and physiological roles of the iodothyronine selenodeiodinases. Endocrine Reviews. 2002;23(1):38–89. 🔗 https://pubmed.ncbi.nlm.nih.gov/11844744/
2. Köhrle J. Selenium and the thyroid. Current Opinion in Endocrinology, Diabetes and Obesity. 2015;22(5):392–401. 🔗 https://pubmed.ncbi.nlm.nih.gov/26186940/
3. Larsen PR, Davies TF. Hypothyroidism and thyroiditis. In: Kronenberg HM, et al., eds. Williams Textbook of Endocrinology. 11th ed. Saunders; 2008.
4. Ortiga-Carvalho TM, Chiamolera MI, Pazos-Moura CC, Wondisford FE. Hypothalamus-pituitary-thyroid axis. Comprehensive Physiology. 2016;6(3):1387–1428. 🔗 https://pubmed.ncbi.nlm.nih.gov/27347897/
5. Mendis-Handagama SM, Ariyaratne HB. Differentiation of the adult Leydig cell population in the postnatal testis. Biology of Reproduction. 2001;65(3):660–671.
6. Hoermann R, Midgley JE, Larisch R, Dietrich JW. Homeostatic control of the thyroid-pituitary axis: perspectives for diagnosis and treatment. Frontiers in Endocrinology. 2015;6:177. 🔗 https://pubmed.ncbi.nlm.nih.gov/26635726/
7. Duntas LH, Wartofsky L. Cardiovascular risk and subclinical hypothyroidism: focus on lipids and new emerging risk factors. What is the evidence? Thyroid. 2007;17(11):1075–1084. 🔗 https://pubmed.ncbi.nlm.nih.gov/18047435/
8. Mancini A, Di Segni C, Raimondo S, et al. Thyroid hormones, oxidative stress, and inflammation. Mediators of Inflammation. 2016;2016:6757154. 🔗 https://pubmed.ncbi.nlm.nih.gov/27051079/
9. Zimmermann MB, Köhrle J. The impact of iron and selenium deficiencies on iodine and thyroid metabolism: biochemistry and relevance to public health. Thyroid. 2002;12(10):867–878. 🔗 https://pubmed.ncbi.nlm.nih.gov/12487769/
10. Wiersinga WM. Paradigm shifts in thyroid hormone replacement therapies for hypothyroidism. Nature Reviews Endocrinology. 2014;10(3):164–174. 🔗 https://pubmed.ncbi.nlm.nih.gov/24419358/
11. Brent GA. Mechanisms of thyroid hormone action. Journal of Clinical Investigation. 2012;122(9):3035–3043. 🔗 https://pubmed.ncbi.nlm.nih.gov/22945636/
12. Chaker L, Bianco AC, Jonklaas J, Peeters RP. Hypothyroidism. The Lancet. 2017;390(10101):1550–1562. 🔗 https://pubmed.ncbi.nlm.nih.gov/28336049/
