Hypothyroid symptoms but normal TSH — this is one of the most common and most consistently mishandled presentations in modern clinical practice. Fatigue that sleep does not fix. Cold intolerance that has become the new normal. Weight that accumulates despite effort. Brain fog that has quietly restructured the day around it. And a blood test that comes back normal, followed by a conversation that ends with: it is probably stress. Come back if things get worse.
Things get worse. And the test still comes back normal. Because the test was never measuring what is actually failing.
There is a specific kind of medical frustration that brings people to this practice. It is not the frustration of having received a difficult diagnosis. It is the frustration of having received no diagnosis at all — of having lived with a constellation of symptoms that have progressively worsened over years, of having had blood drawn and panels run and results reviewed, and of having been told, repeatedly, that everything looks normal. That the thyroid is fine. That it is probably stress. That it might be aging. That perhaps they should consider an antidepressant.
The patients who arrive with this history are not rare. They are, in functional metabolic practice, one of the most consistently encountered presentations. And almost without exception, when the full picture is finally assembled — not TSH alone, but the complete thyroid metabolism panel described in the thyroid metabolism post — the biology tells a coherent story that the standard assessment was never designed to read.
The story is this: the thyroid is not broken. It is adapting. The body is under metabolic stress — chronic hyperinsulinemia, low-grade inflammation, cortisol dysregulation, impaired liver function, nutritional gaps — and in response to that stress, it is deliberately slowing its metabolism. The thyroid system is working exactly as designed. The problem is the environment that has been instructing it to downregulate. And that environment has been ignored because the test being used to assess thyroid function measures only whether the instruction is being sent — not whether it is being executed, not whether the active hormone is being produced, not whether the cells receiving it are actually responding.
This post is for the patient who has been symptomatic for years and been told they are fine. It explains what is actually happening, why the standard panel misses it, what the full picture looks like, and what changes when the metabolic root is addressed rather than the downstream number.
What you will learn:
What the typical diagnostic journey looks like for patients with functional hypothyroidism — and why it takes so long | Which pattern of thyroid dysfunction is most common in clinical practice and what that patient looks like | Why the explanation that lands best is not “your thyroid is deficient” but “your body is adapting” | What the intervention timeline looks like when the metabolic environment is corrected | Why patients on levothyroxine can still feel completely hypothyroid — and what is actually happening in those cases
The Diagnostic Journey: What These Patients Have Been Through
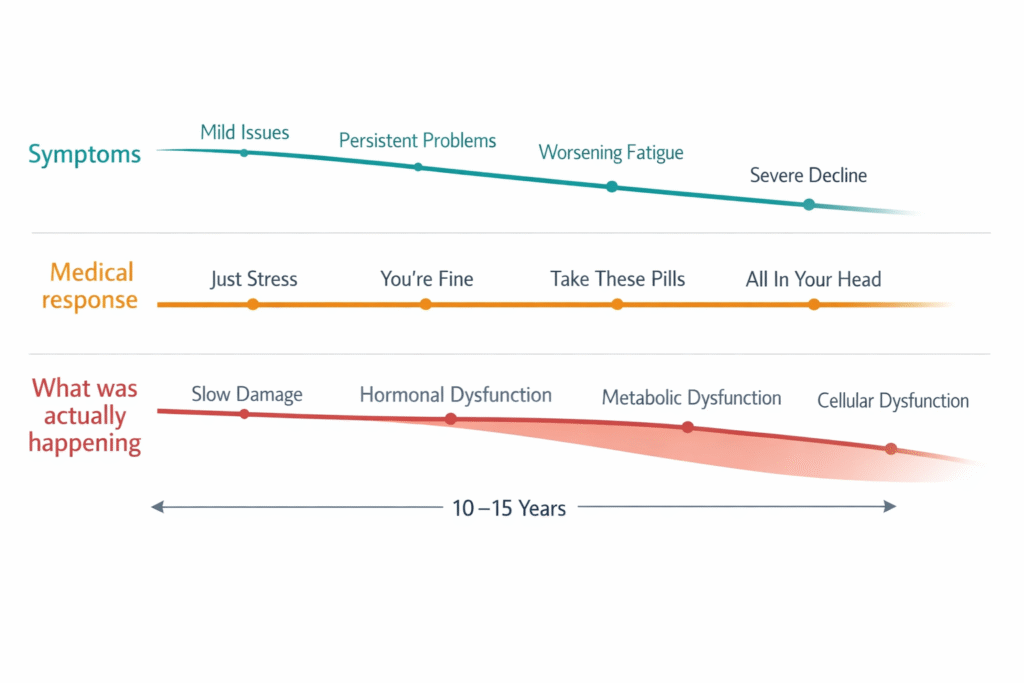
The history that patients with functional hypothyroidism bring to a first consultation is almost never new. It is typically a five to fifteen year slow decline — a gradual accumulation of symptoms that individually could be explained away and collectively paint an unmistakable picture that the standard diagnostic framework consistently fails to recognize.
It begins subtly. Fatigue that is slightly beyond what rest explains. Reduced resilience to stress. A few kilograms of weight gain that does not respond to the usual adjustments. The kind of changes that are easy to attribute to a busy period, a stressful year, the natural progression of aging. Labs get checked — TSH is measured, comes back within the reference range, and the conversation ends. Normal. Come back if things get worse.
Things get worse. Cold intolerance appears — hands and feet that stay cold regardless of the season, a core temperature that never quite feels right. Brain fog settles in: the word that is just out of reach, the concentration that requires effort where it once came naturally, the cognitive flatness that colleagues and family notice before the patient fully registers it themselves. Mood changes follow — a low-grade flatness, reduced motivation, a heaviness that does not have an obvious emotional cause. The metabolism slows visibly: the same dietary pattern that once maintained weight now produces gradual gain, and the efforts to reverse it produce diminishing returns.
At this point, several things typically happen — and almost none of them address what is actually occurring. The patient is labeled rather than understood. Stress. Burnout. Perimenopause. Depression. Aging. Each label comes with its own management approach: an antidepressant for the mood component, generic advice to eat less and move more for the weight, stress management recommendations for the fatigue. Sometimes levothyroxine is prescribed — not because the conversion problem has been identified, but because TSH has nudged toward the upper end of the reference range and a clinical decision is made to treat the number. The TSH normalizes. The patient still feels terrible. The case is considered closed.
Meanwhile, the real physiology continues unaddressed. Impaired T4 to T3 conversion driven by insulin resistance and chronic inflammation. Rising reverse T3 — the metabolic brake signal the body is producing in response to a stressed system. Mitochondrial downregulation reducing the cellular energy capacity that both thyroid hormone production and conversion require. The same metabolic environment feeding back on itself, worsening with each year it remains uncorrected.
By the time these patients reach this practice, they are not borderline. They are deep in functional hypothyroidism — with normal labs and a five to fifteen year history of being told so.
Clinical Perspective: What I See in Practice
The most common pattern I encounter is not primary thyroid failure. It is impaired T4 to T3 conversion — not a problem with the thyroid gland itself, but a problem with the metabolic signaling environment in which thyroid hormone is supposed to be activated and used. The thyroid is producing T4. The conversion machinery is not efficiently converting it to active T3. And the body is simultaneously diverting conversion toward reverse T3 — the inactive competing form — as a deliberate energy-conservation response to a system under chronic metabolic stress.
The patient this pattern belongs to has a recognizable profile. They are typically between 35 and 60 years old. There is slight to moderate weight gain, predominantly central, that has resisted effort. Chronic fatigue — not the acute tiredness of a bad night’s sleep, but the persistent, unrefreshing exhaustion that has become the new baseline. Brain fog. Low mood that does not have an obvious emotional origin.
Cold intolerance that their GP has noted but not connected to anything actionable. And a history of chronic lifestyle strain — years of high stress, poor sleep, irregular eating, or the high-performing professional pattern of overwork and inadequate recovery described in the cortisol dysregulation post. The full hormonal cascade that chronic stress produces — from cortisol through insulin to thyroid suppression — is mapped in detail in how chronic stress rewires the entire hormonal hierarchy.
Underneath this clinical picture, the metabolic environment is consistent: insulin resistance with elevated fasting insulin and a rising TG/HDL ratio. Chronic low-grade inflammation visible in hsCRP. Elevated cortisol driving reverse T3 production and suppressing deiodinase enzyme activity. A liver-gut axis underperforming as a conversion site. The lab pattern, when the full panel is finally run, is equally consistent: TSH normal, free T4 normal, free T3 low or low-normal, reverse T3 elevated if measured. The thyroid gland is responding appropriately to a stressed system. It is the system that is the problem.
When I explain this to patients, the response is almost never resistance. It is relief. Sometimes profound relief — the kind that comes after years of being told that nothing is wrong while feeling clearly unwell. The explanation that lands best is also the most accurate one: your thyroid is not broken. It is adapting. Your body is under metabolic stress — insulin, inflammation, stress hormones — and it is intentionally slowing your metabolism to protect you. The thyroid system is working as designed. The solution is not giving more hormone. It is removing the signals that are telling your body to slow down in the first place.
That reframe — from defect to adaptation, from drug dependency to restoring physiology — changes everything about how patients engage with what comes next. They are not managing a chronic disease. They are correcting an environment. And for most of them, after years of being told their symptoms were not real or not treatable, the idea that the biology makes sense and that the upstream drivers are addressable is genuinely transformative.
There is also a specific and extremely common patient in this space that deserves its own description: the person who has been on levothyroxine for years and still feels completely hypothyroid. Their TSH has been normalized by the medication. Their case has been considered successfully treated. And they are exhausted, cold, gaining weight, cognitively foggy, and increasingly frustrated that the treatment they were told would fix the problem has not fixed it.
What is happening in these cases is almost always the same. Levothyroxine is T4 — the inactive prohormone. It has been prescribed to replace the hormone the thyroid is producing, and the TSH has normalized in response to the additional T4 in circulation. But the conversion problem — the impaired T4 to T3 pathway driven by insulin resistance, inflammation, elevated cortisol, and liver dysfunction — was never addressed. The additional T4 being supplied by the medication is facing exactly the same conversion obstacles as the endogenous T4 was. Worse, the excess T4 in some cases is being actively diverted toward reverse T3, compounding the metabolic brake signal. The patient is biochemically treated. They are still functionally hypothyroid.
In some of these cases, increasing the levothyroxine dose makes things worse rather than better — because more T4 in a system with impaired and inflammation-suppressed deiodinase activity means more substrate for reverse T3 production rather than more active T3. The system is saying: I have enough hormone. But I am not going to activate it. Giving it more raw material does not change that decision. Removing the metabolic signals driving the decision does.
Hypothyroid Symptoms but Normal TSH: Why the Standard Panel Cannot See This
The gap between what patients experience and what standard thyroid testing detects is not a failure of the patients to describe their symptoms accurately. It is a structural limitation of a diagnostic framework that was designed to identify primary thyroid disease — gland failure, autoimmune destruction, surgical absence — and is being applied to a completely different category of dysfunction.
TSH reflects pituitary signaling. It is a sensitive marker for primary hypothyroidism — when the thyroid gland itself is failing to produce adequate hormone, TSH rises in response to the deficit. In that clinical context, TSH is a useful and appropriate screening tool. In the context of functional hypothyroidism — where the gland is producing hormone but the conversion, transport, or receptor response is impaired — TSH provides almost no information about the actual metabolic output of the thyroid system.
A patient with impaired T4 to T3 conversion has adequate circulating T4. The pituitary detects adequate T4. TSH remains normal. The fact that the T4 is not being efficiently converted to active T3 — that free T3 is low, that reverse T3 is competing for receptor binding, that cells are receiving a downregulated thyroid signal — is entirely invisible to the TSH measurement. The test has done its job correctly. It has confirmed that the pituitary is not detecting a T4 deficit. It has said nothing about whether the T4 is becoming functional.
The minimum panel that allows genuine assessment of thyroid metabolism — TSH, free T4, free T3, reverse T3, and TPO and thyroglobulin antibodies — is not standard practice in most clinical settings because the conventional framework does not recognize the conversion and signaling patterns it would reveal as actionable diagnostic categories. This is the core clinical gap. And it is the gap that leaves patients symptomatic with normal labs for years, receiving treatments aimed at the downstream number rather than the upstream mechanism.
The Five Patterns — and How to Distinguish Them Clinically
As detailed in the thyroid metabolism post, functional hypothyroidism presents in five distinct patterns, each with a different mechanism and a different intervention requirement. In patient-facing practice, the ability to distinguish between them determines whether the intervention helps or misses entirely.
The most common pattern — impaired T4 to T3 conversion — presents with normal TSH, normal free T4, low or low-normal free T3, and elevated reverse T3. The driver is metabolic: insulin resistance, chronic inflammation, elevated cortisol, and impaired liver function. The intervention addresses those drivers. Thyroid hormone replacement without addressing the conversion environment gives the body more T4 to divert toward reverse T3.
Pituitary suppression — pattern 1 from Post 1 — presents with TSH that is normal on the conventional scale but below the functional optimal of 1.8–3.0 µIU/mL, combined with low T4. The pituitary is not adequately stimulating a competent thyroid gland because HPA axis dysregulation, chronic stress, or blood sugar instability has suppressed hypothalamic TRH and pituitary TSH output. The thyroid gland is structurally normal and capable. The problem is upstream. Addressing the cortisol and insulin drivers restores adequate pituitary stimulation.
Elevated TBG — pattern 3 — presents most commonly in women on oral contraceptives or with the estrogen dominance picture described in the estrogen dominance post. High estrogen increases hepatic TBG production, sequestering thyroid hormone and reducing free availability. TSH and total T4 look normal. Free T3 is reduced. Addressing the estrogen load and its metabolic drivers — particularly the insulin resistance driving SHBG suppression and free estrogen excess — is the intervention.
Low TBG — pattern 4 — is the inverse, seen in women with PCOS and elevated androgens. Low TBG increases free hormone but drives receptor downregulation. The patient is symptomatically hypothyroid despite elevated free T3. Reducing the insulin resistance that is driving the androgen excess corrects TBG and restores receptor sensitivity.
Thyroid hormone resistance — pattern 5 — is the most diagnostically challenging because all standard markers appear entirely normal. The dysfunction is at the receptor level. It is associated with chronic cortisol elevation, elevated homocysteine, and genetic factors affecting receptor sensitivity. The clinical signal is the only available diagnostic indicator: persistent hypothyroid symptoms with a completely normal comprehensive thyroid panel.
What Changes When the Metabolic Environment Is Corrected
When the intervention addresses the metabolic drivers rather than the downstream hormone level, the timeline of recovery follows a consistent and predictable sequence.
In the first one to three weeks, energy begins to stabilize. This is the earliest and most reliable signal — the sharp afternoon energy crashes soften, brain fog begins to lift, hunger and cravings normalize. These changes reflect the initial stabilization of blood glucose and the early reduction in the compensatory cortisol signaling that has been suppressing thyroid conversion. Patients often describe this phase as the first sign in years that something is actually different, rather than just managed.
Between weeks four and eight, the deeper metabolic shift becomes apparent. Temperature tolerance improves — the chronic cold intolerance that patients have adapted to over years begins to resolve as cellular metabolic rate starts to recover. Sleep deepens. Mood stabilizes. The first measurable lab shifts appear in the metabolic panel: fasting insulin falling, triglycerides improving, the TG/HDL ratio moving in the right direction. The thyroid panel itself has not yet moved significantly at this stage — what has moved is the metabolic environment that was suppressing thyroid function.
Between weeks eight and sixteen, thyroid expression begins to improve measurably. Free T3 rises as conversion efficiency increases in the now less-inflamed, better-nourished, lower-cortisol environment. Reverse T3 falls as the metabolic brake signal is withdrawn. Resting metabolic rate increases. Body composition begins to change — not from caloric restriction, but from restored metabolic signaling that allows the body to stop conserving fuel and begin oxidizing it normally.
Beyond three to six months, what patients describe is not the management of a condition but the restoration of a state they had forgotten was possible. Sustainable energy through the full day. Fat loss that occurs without forcing caloric deficit. Thyroid symptoms largely resolved. And in patients who were on levothyroxine when the metabolic work began — this is where the clinical picture becomes particularly significant. As insulin resistance, inflammation, and cortisol signaling normalize, conversion efficiency improves and the body no longer requires the same pharmacological T4 load to maintain adequate free T3. Dose reductions become appropriate, managed in collaboration with the prescribing physician. In some cases — particularly where there is no underlying autoimmune destruction — partial or full discontinuation becomes possible.
This is not an argument against levothyroxine. It is an argument for understanding that levothyroxine given to a metabolically dysregulated patient is working against a conversion environment that remains impaired. Correct the environment, and the medication requirement changes accordingly.
Hashimoto’s Thyroiditis: Why the Immune System Targets the Thyroid
Patients with Hashimoto’s thyroiditis represent a distinct and important category within the hypothyroid picture. Hashimoto’s is the most common cause of hypothyroidism in Western populations — and it is frequently misunderstood as a single-cause condition when it is, mechanistically, a multi-hit process in which immune tolerance to thyroid tissue progressively breaks down.
What actually happens is this: the immune system stops recognizing thyroid proteins — specifically TPO and thyroglobulin — as self. Thyroid antigens are presented to T cells under inflammatory conditions, a Th1 and Th17 dominant immune response develops, cytotoxic T cells infiltrate thyroid tissue, and B cells produce the antibodies that appear on the lab report. Those antibodies — TPO-Ab and Tg-Ab — are markers of immune activity. They are not the root cause. They are evidence that the immune process is already underway.
What drives the loss of tolerance in the first place is not a single factor but a convergence of conditions. Increased intestinal permeability (leaky gut) allows bacterial fragments, food antigens, and LPS to enter circulation, producing chronic immune activation and the molecular mimicry through which the immune system begins cross-reacting with thyroid tissue. Insulin resistance, chronic low-grade inflammation, and HPA axis dysregulation shift the immune environment toward autoimmune reactivity.
Selenium deficiency impairs the glutathione peroxidase system that would otherwise buffer the oxidative stress generated during thyroid hormone synthesis — and in the presence of high iodine intake without adequate selenium, the resulting hydrogen peroxide accumulation damages thyroid tissue, exposes antigens, and can trigger or accelerate the autoimmune cascade. Iodine excess in a selenium-deficient system is therefore a possible amplifier of Hashimoto’s — not the universal root cause, but a meaningful contributor in the right metabolic context.
The critical clinical distinction is this: levothyroxine replaces the hormone that a damaged thyroid can no longer produce in sufficient quantity. It does not address the immune process producing the damage. A patient with Hashimoto’s who is medicated but whose gut permeability, insulin resistance, inflammatory load, and micronutrient status remain uncorrected is receiving hormone replacement while the autoimmune destruction continues. The metabolic and lifestyle interventions described in this post remain relevant and often significantly reduce the autoimmune flare cycle — but the Hashimoto’s picture requires specific additional consideration that goes beyond functional conversion impairment alone. The full mechanistic picture — gut-immune axis, molecular mimicry, the iodine-selenium interaction, and what actually changes the autoimmune trajectory — is covered in the dedicated Hashimoto’s post.
A Note on Uncertainty
The clinical patterns described in this post represent a functional medicine framework for understanding thyroid dysfunction beyond primary gland failure. They are not universally accepted diagnostic categories in conventional endocrinology, and the evidence base for some patterns — particularly thyroid hormone resistance and the clinical significance of elevated reverse T3 in the absence of primary thyroid disease — relies more heavily on mechanistic reasoning and clinical observation than on large randomized trial data.
Any assessment of thyroid function, and any decisions regarding thyroid hormone therapy, dose adjustment, or discontinuation, must be made in collaboration with the prescribing physician. Levothyroxine dose changes should never be made without medical supervision. The functional medicine approach described here addresses the metabolic environment in which thyroid hormone is produced, converted, and used — it does not constitute a diagnosis of any thyroid condition, and it does not replace the medical management of confirmed hypothyroidism or autoimmune thyroid disease.
Patients with Hashimoto’s thyroiditis — autoimmune thyroid destruction — represent a distinct clinical category in which the gland itself is being progressively damaged. The metabolic and lifestyle interventions described here remain relevant and often dramatically reduce the autoimmune flare cycle, but the autoimmune dimension requires specific additional consideration covered in the Hashimoto’s post.
Practical Implications
If you have been symptomatic for years — fatigue, cold intolerance, weight gain that resists effort, brain fog, low mood — and your thyroid has been assessed by TSH alone and declared normal, the metabolic mechanisms producing your symptoms have not been evaluated. A normal TSH rules out primary thyroid gland failure. It does not assess whether your T4 is converting to active T3, whether your reverse T3 is elevated, whether your TBG is sequestering hormone away from tissues, or whether your cells are responding to the thyroid signal they are receiving.
The practical starting point is requesting the full thyroid panel — TSH, free T4, free T3, reverse T3, and thyroid antibodies — alongside fasting insulin, HOMA-IR, hsCRP, and the TG/HDL ratio. These metabolic markers are not separate from the thyroid question. They are the upstream drivers of the thyroid dysfunction — and they are where the intervention begins.
If you are on levothyroxine and still symptomatic, the question worth asking is not whether the dose is high enough. It is whether the metabolic environment converting your T4 to active T3 has ever been assessed. If it has not, the dose question is secondary to the conversion question — and increasing T4 in an unconverted system may worsen rather than improve the clinical picture.
Your thyroid is not broken. It is adapting to an environment that has been driving it to slow down. Change the environment — and the thyroid follows.
The pattern of hypothyroid symptoms with normal TSH is one example of a much broader phenomenon — the full clinical framework for hormonal symptoms with metabolic roots explains why this pattern is increasingly common in modern practice.
People Also Ask
Why do I feel hypothyroid if my TSH is normal?
Because TSH measures pituitary signaling — not the actual metabolic output of your thyroid system. A normal TSH confirms that your pituitary is not detecting a significant T4 deficit. It does not confirm that your T4 is efficiently converting to active T3, that reverse T3 is not blocking T3 at the receptor level, that thyroid-binding globulin is not sequestering free hormone away from tissues, or that your cells are responding normally to thyroid hormone. Functional hypothyroidism — with normal TSH and genuine hypothyroid symptoms — is driven by conversion impairment, TBG dysregulation, reverse T3 excess, or receptor resistance, none of which are visible on a TSH-only panel.
What is the most common cause of feeling hypothyroid with normal labs?
In clinical practice, the most common pattern is impaired T4 to T3 conversion driven by insulin resistance, chronic low-grade inflammation, elevated cortisol, and liver dysfunction. The thyroid is producing adequate T4. The peripheral conversion machinery — primarily selenium-dependent deiodinase enzymes — is operating in an inflammatory, insulin-resistant, cortisol-elevated environment that suppresses its activity and diverts conversion toward reverse T3. The result is adequate circulating T4, insufficient active T3, and the full hypothyroid symptom picture with a normal standard panel.
Why am I still hypothyroid on levothyroxine?
Levothyroxine is T4 — the inactive prohormone. If the metabolic environment impairing T4 to T3 conversion has not been addressed, the additional T4 from medication faces the same conversion obstacles as endogenous T4. In some cases, excess T4 in an inflamed, cortisol-elevated system is actively diverted toward reverse T3, compounding the metabolic brake signal. TSH normalizes because the pituitary detects adequate T4 in circulation — but free T3 remains low and the patient remains functionally hypothyroid. The medication has treated the number without correcting the mechanism.
How long does it take to recover from functional hypothyroidism?
When the metabolic drivers are genuinely addressed — insulin load reduced, inflammation resolved, cortisol dysregulation corrected, nutrient cofactors restored — recovery follows a consistent timeline. Early energy stabilization and brain fog improvement within one to three weeks. Improved temperature tolerance, sleep quality, and mood between weeks four and eight. Measurable improvements in free T3 and reverse T3 between weeks eight and sixteen. Full metabolic restoration and sustained symptom resolution typically between three and six months. Patients on levothyroxine often require dose review and reduction during this period, under physician supervision.
What tests should I ask for if I think I have functional hypothyroidism?
The minimum meaningful thyroid panel includes TSH, free T4, free T3, reverse T3, and TPO and thyroglobulin antibodies. Alongside this, fasting insulin, HOMA-IR, hsCRP, and the TG/HDL ratio establish the metabolic environment driving conversion impairment. Ferritin, selenium, zinc, and vitamin D complete the nutritional picture. This combination allows the full thyroid metabolism picture to be assessed — not just whether the pituitary is signaling correctly, but whether the active hormone is being produced, delivered, and used.
Can hypothyroid symptoms improve without thyroid medication?
In cases of functional hypothyroidism — where the primary driver is metabolic rather than autoimmune destruction of the gland — yes. When insulin resistance, chronic inflammation, cortisol dysregulation, and nutritional deficiencies are corrected, the conversion environment improves, reverse T3 falls, and the thyroid system begins to function more effectively within its own capacity. Patients already on levothyroxine frequently require dose reduction as the metabolic picture improves. Whether medication can be fully discontinued depends on the individual case — particularly on whether autoimmune damage has reduced the gland’s intrinsic production capacity — and must be managed in collaboration with the prescribing physician.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Chaker L, Bianco AC, Jonklaas J, Peeters RP. Hypothyroidism. The Lancet. 2017;390(10101):1550–1562. 🔗 https://pubmed.ncbi.nlm.nih.gov/28336049/
2. Jonklaas J, Bianco AC, Bauer AJ, et al. Guidelines for the treatment of hypothyroidism. Thyroid. 2014;24(12):1670–1751. 🔗 https://pubmed.ncbi.nlm.nih.gov/25266247/
3. Hoermann R, Midgley JE, Larisch R, Dietrich JW. Homeostatic control of the thyroid-pituitary axis: perspectives for diagnosis and treatment. Frontiers in Endocrinology. 2015;6:177. 🔗 https://pubmed.ncbi.nlm.nih.gov/26635726/
4. Bianco AC, Salvatore D, Gereben B, Berry MJ, Larsen PR. Biochemistry, cellular and molecular biology, and physiological roles of the iodothyronine selenodeiodinases. Endocrine Reviews. 2002;23(1):38–89. 🔗 https://pubmed.ncbi.nlm.nih.gov/11844744/
5. Wiersinga WM. Paradigm shifts in thyroid hormone replacement therapies for hypothyroidism. Nature Reviews Endocrinology. 2014;10(3):164–174. 🔗 https://pubmed.ncbi.nlm.nih.gov/24419358/
6. Visser WE, Friesema EC, Visser TJ. Minireview: thyroid hormone transporters: the knowns and the unknowns. Molecular Endocrinology. 2011;25(1):1–14. 🔗 https://pubmed.ncbi.nlm.nih.gov/20660303/
7. Ortiga-Carvalho TM, Chiamolera MI, Pazos-Moura CC, Wondisford FE. Hypothalamus-pituitary-thyroid axis. Comprehensive Physiology. 2016;6(3):1387–1428. 🔗 https://pubmed.ncbi.nlm.nih.gov/27347897/
8. Duntas LH, Maillis A. Hypothyroidism and depression: salient aspects of pathogenesis and management. Minerva Endocrinologica. 2013;38(4):365–377. 🔗 https://pubmed.ncbi.nlm.nih.gov/24126551/
9. Idrees T, Palmer S, Krouss W, Gurchiek T, Palmer L. T4 only replacement and residual symptoms: a new perspective. Endocrine Connections. 2022;11(6). 🔗 https://pubmed.ncbi.nlm.nih.gov/35416801/
10. Larisch R, Midgley JEM, Dietrich JW, Hoermann R. Symptomatic relief is related to serum free triiodothyronine concentrations during follow-up in levothyroxine-treated patients with differentiated thyroid cancer. Experimental and Clinical Endocrinology & Diabetes. 2018;126(9):546–552. 🔗 https://pubmed.ncbi.nlm.nih.gov/29186683/
11. Mancini A, Di Segni C, Raimondo S, et al. Thyroid hormones, oxidative stress, and inflammation. Mediators of Inflammation. 2016;2016:6757154. 🔗 https://pubmed.ncbi.nlm.nih.gov/27051079/
12. Tsigos C, Chrousos GP. Hypothalamic-pituitary-adrenal axis, neuroendocrine factors and stress. Journal of Psychosomatic Research. 2002;53(4):865–871. 🔗 https://pubmed.ncbi.nlm.nih.gov/12377295/