Why chasing cortisol while ignoring insulin is failing the patients who need help most — and what the evidence actually says.
Most patients who arrive with cortisol dysregulation have already been told some version of the same story. You are stressed. You need to relax more. Your cortisol test came back normal. Come back if things get worse. What they were rarely told — and what changes everything when they finally hear it — is this: the cortisol pattern they are experiencing is not primarily a stress management failure. It is the downstream consequence of a metabolic environment that has been forcing their stress axis to compensate for instability it was never designed to carry indefinitely.
Cortisol dysregulation is one of the most consistently misframed conditions in modern clinical practice. Conventional medicine recognizes cortisol pathology at its extremes — Cushing’s syndrome at the high end, Addison’s disease at the low end — and largely ignores the vast middle ground where the hypothalamic-pituitary-adrenal axis is functioning, but functioning badly. The rhythm is flattened or inverted. The signaling is dysregulated. The system is stuck in a chronic compensatory pattern that produces recognizable, reproducible symptoms — and normal lab values.
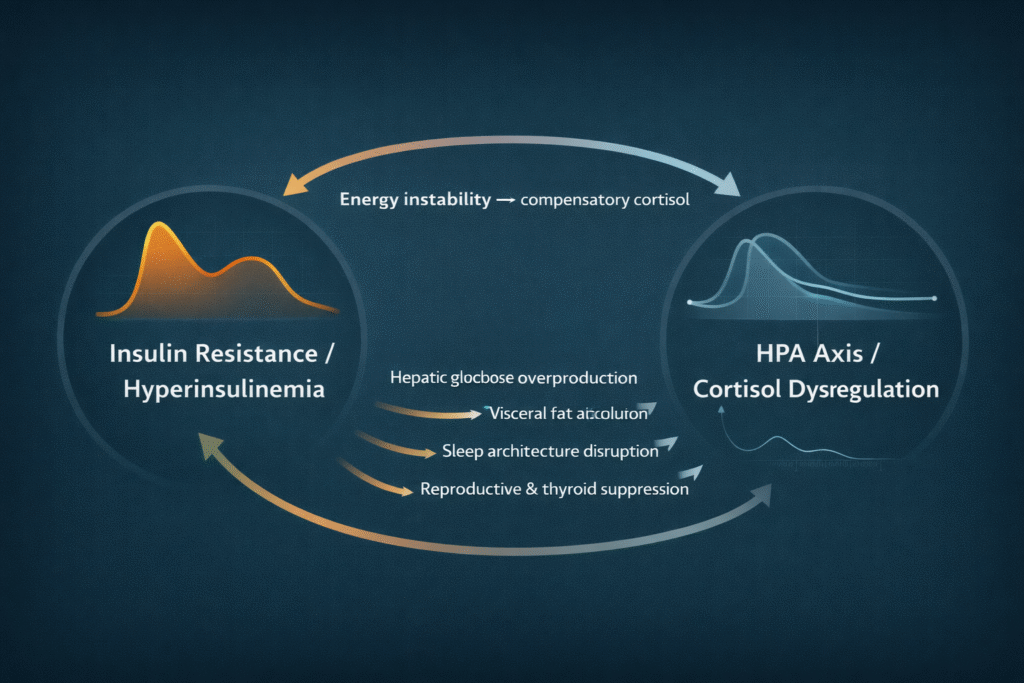
The metabolic science of cortisol tells a more precise and more actionable story. Chronic hyperinsulinemia and unstable blood glucose force the HPA axis into a compensatory role it was designed to play acutely, not chronically. Once cortisol is chronically elevated, it feeds back directly into the metabolic system — increasing hepatic glucose output, promoting visceral fat accumulation, worsening insulin resistance, disrupting sleep architecture, and suppressing the reproductive and thyroid axes. What begins as a metabolic problem becomes a hormonal amplification loop. Correct the metabolic environment, and the cortisol pattern follows.
Cortisol dysregulation describes a state in which the diurnal rhythm of cortisol is disrupted, inverted, or flattened. It is driven by dysfunction of the HPA axis — the Hypothalamic-Pituitary-Adrenal axis, your body’s central stress-response system. The hypothalamus senses stress and sends a signal, the pituitary amplifies it, and the adrenal glands respond by releasing cortisol. In a healthy system this cascade is precise, rhythmic, and self-limiting. When it becomes chronically dysregulated — through metabolic strain, poor sleep, or sustained psychological stress — it stops being a response to specific threats and becomes a permanent background state. And in that state, it reaches into every other hormonal system in the body: suppressing thyroid conversion, worsening insulin resistance, disrupting reproductive hormones, and impairing sleep architecture.
What you will learn:
What cortisol dysregulation actually means — and why a normal lab result does not rule it out | How insulin resistance drives the HPA axis into chronic compensatory activation | The four mechanisms through which chronic cortisol elevation worsens the metabolic condition producing it | The specific patient patterns that identify cortisol as a rate-limiting factor in metabolic recovery | What changes when the metabolic root is addressed, and in what order
The Conventional Story — and Where It Falls Short
Standard cortisol assessment measures a single morning serum value and compares it against a population reference range. What it captures is whether the adrenal glands are producing cortisol within expected bounds at one point in time. What it does not capture is how cortisol behaves across the full diurnal cycle, whether the morning cortisol awakening response is functioning correctly, whether evening cortisol is appropriately suppressed, whether there are pathological nighttime spikes, or whether the HPA axis is responding adaptively or in a state of chronic dysregulation.
This is the core clinical blind spot. A patient can have a perfectly normal morning cortisol value and a completely inverted diurnal rhythm — low in the morning when it should be high, elevated at night when it should be falling — and the standard test will report nothing abnormal. The symptoms that result from that inverted rhythm are real and consistent: difficulty waking, persistent morning fatigue, an energy pattern that runs backwards relative to the day, wired and alert when the brain should be winding down, unable to reach the deep sleep that restores metabolic and immune function.
Conventional medicine is designed to rule out dangerous extremes and structural pathology. It does that well. Cushing’s and Addison’s are serious conditions, and their exclusion matters. But the diagnostic system stops there. The vast territory between disease and optimal function — where the majority of patients with genuine HPA dysregulation actually live — is not addressed, and in many cases is not recognized as a clinical entity worth investigating.
The result is that patients are told their cortisol is normal, their symptoms are attributed to stress or aging or insufficient willpower, and the metabolic drivers producing their dysregulated stress axis continue operating, unexamined, for years.
Addressing insulin resistance without recognizing its role in HPA axis dysregulation means missing one of the most important feedback loops in metabolic medicine.
Clinical Perspective: What I See in Practice
In practice, what I see repeatedly is not isolated cortisol dysfunction. I see patterns — and they present with a consistency that makes the underlying mechanism unmistakable once you know what to look for.
Patients describe the same combination of symptoms. Tired during the day, unable to fully relax at night. Waking in the middle of the night, often around the same time — 2 to 3 in the morning — and lying there with a mind that will not quiet. Energy that rises and falls unpredictably, disconnected from how much they have eaten or slept. And almost always, some form of stress-driven or emotional eating in the background, particularly in the evening. The cravings are specific: sugar, refined carbohydrates, sometimes salt. Not hunger in the conventional sense. A biological pull toward fast energy at a time when the body is running on compensatory cortisol.
When you look deeper, another layer becomes clear. These patients are not just stressed. They are metabolically unstable. Their eating patterns revolve around refined carbohydrates and ultra-processed foods. Meals are late. Screens stay on into the night. The brain never fully switches off. And underneath all of this, there is almost always a degree of hyperinsulinemia — visible in fasting insulin, in a TG/HDL ratio that has been quietly climbing, in a waist circumference that does not match the rest of their body.
The connection is not obvious at first. These patients are usually told this is a stress problem. And stress is part of it. But it is not the full explanation. What becomes clear over time is that the more unstable their metabolic environment becomes, the more the body recruits cortisol to compensate. And the more cortisol compensates, the more it worsens the metabolic instability that required it in the first place. This is not two separate problems running in parallel. It is one system — metabolic instability driving cortisol compensation, which feeds back and deepens the metabolic instability.
One patient described this transition clearly. Before working together, she was a stressed and overweight manager — constantly tired, mentally drained, eating driven by pressure and long working hours. Sleep was inconsistent. Energy was unpredictable. Twelve weeks later, the description had changed entirely. Genuinely energized. Emotional eating gone. Food simpler but more nutrient-dense. Energy stable despite the same workload. Biomarkers improved in a way her doctor did not expect. Nothing in that process targeted cortisol directly. What changed was the metabolic environment. And cortisol followed.
There is also a specific pattern I see repeatedly in high-performing professionals — people who present as doing everything right. They exercise. They eat what they believe is a healthy diet. They are disciplined and consistent.
But underneath: they underfuel protein and over-rely on carbohydrates and snacking, often following the advice to eat every two to three hours — which keeps insulin elevated throughout the entire day. They push through fatigue with caffeine. They delay recovery — sleep, proper meals, genuine rest — because of work demands. They are simultaneously metabolically unstable and chronically neurologically stimulated. The result is the classic picture: wired at night, exhausted during the day, strong cravings, poor sleep quality — despite high levels of effort and apparent compliance.
This is not a failure of discipline. It is effort applied in the wrong physiological direction.
When patients ask me about their normal cortisol test result, I am careful about how I respond. I validate the result without dismissing the symptoms. A normal cortisol value means the adrenal glands are not failing — there is no Cushing’s, no Addison’s — and that is genuinely important information. But normal does not mean optimal.
It means the value falls within a broad statistical range derived from a population, measured at a single point in time. It does not tell you how cortisol behaves across the day. It does not tell you whether the diurnal rhythm is intact. It does not tell you how the brain is regulating the HPA axis, or whether the system is adapted and resilient or compensating and fragile.
The shift I offer patients is from asking whether cortisol is high or low to asking whether the stress system is functioning in a stable, adaptive rhythm. The adrenal glands are not failing. The signaling, the rhythm, and the adaptation are dysregulated. That is a different problem — and it has a different solution.
How Insulin Resistance Drives HPA Dysregulation: The Primary Mechanism
The relationship between insulin resistance and cortisol dysregulation is bidirectional — but in clinical practice, insulin dysregulation almost always sets the stage first. Understanding the sequence clarifies why targeting cortisol directly, without addressing the metabolic environment producing it, consistently falls short.
In the early stages of insulin resistance, fasting glucose often remains completely normal. The compensatory hyperinsulinemia that maintains that normal glucose is not captured by standard screening. What it produces, however, is chronic energy instability — the cellular inability to efficiently access and utilize fuel between meals and overnight creates predictable gaps in substrate availability. The brain, which depends on stable glucose delivery and cannot tolerate significant drops, responds by activating the HPA axis. Cortisol rises to stimulate hepatic glucose output and maintain cerebral fuel supply. This is physiologically appropriate as an acute response. When insulin resistance is chronic and the energy gaps are continuous, this cortisol activation becomes chronic as well.
The patient in this stage is not primarily stressed. They are metabolically unstable, and their stress axis is working correctly — compensating for an energy supply problem that their fasting insulin and HOMA-IR would reveal if anyone ordered them.
Once cortisol is chronically elevated, the feedback loop closes and the relationship becomes genuinely bidirectional. Cortisol stimulates hepatic glucose production through gluconeogenesis and glycogenolysis — raising blood glucose and requiring more insulin secretion in response. It promotes lipolysis, releasing free fatty acids that further impair hepatic and skeletal muscle insulin signaling. It directly increases visceral fat deposition through glucocorticoid receptor activation in visceral adipocytes — and visceral fat is itself metabolically active, producing the inflammatory cytokines that worsen insulin resistance further. Cortisol also suppresses the anabolic signaling — growth hormone, IGF-1, testosterone — that would otherwise support muscle mass and maintain peripheral glucose disposal capacity.
The combined clinical picture is distinctive and recognizable. A patient who is tired but wired. Unstable energy with strong cravings, particularly in the afternoon and evening. Central fat accumulation that resists dietary effort. Disrupted sleep, especially nocturnal waking between 2 and 4 in the morning when cortisol should be at its nadir. Difficulty losing weight despite what appears to be a reasonable protocol. Labs that show elevated fasting insulin, rising triglycerides, trending inflammatory markers — with glucose still normal and HbA1c unchanged. The metabolic dysfunction is progressing. The standard panel is not detecting it.
How this same cortisol-immune mechanism contributes to autoimmune thyroid disease specifically is covered in why Hashimoto’s is an immune problem with a metabolic root.
The Four Mechanisms Through Which Chronic Cortisol Worsens Metabolic Health
Mechanism 1: Hepatic Glucose Overproduction and Secondary Hyperinsulinemia
Cortisol stimulates hepatic glucose output through two converging pathways: it activates gluconeogenesis — the synthesis of new glucose from amino acid and glycerol substrates — and promotes glycogenolysis, the breakdown of hepatic glycogen stores. The result is persistent hepatic glucose delivery into the circulation regardless of dietary intake. In a metabolically healthy individual with intact insulin sensitivity, this glucose is efficiently cleared by peripheral tissues.
In a patient with existing insulin resistance, it compounds the already-elevated glucose load and requires additional insulin secretion to manage. Cortisol-driven hepatic glucose production and insulin resistance create a self-reinforcing cycle: more hepatic glucose requires more insulin, more insulin worsens insulin resistance, and worsening insulin resistance requires more cortisol to maintain energy stability.
Mechanism 2: Visceral Fat Accumulation and the Inflammatory Amplification Loop
Glucocorticoid receptors are expressed at high density in visceral adipose tissue. Chronically elevated cortisol activates these receptors, promoting preferential fat deposition in the visceral compartment — around the abdominal organs, in the omentum, and in the liver. This is not simply cosmetic. Visceral fat is metabolically active tissue that releases free fatty acids directly into the portal circulation, driving hepatic lipid accumulation and de novo lipogenesis. It also produces the inflammatory cytokines — TNF-alpha, IL-6, resistin — that directly interfere with insulin receptor signaling in liver, muscle, and adipose tissue. Cortisol-driven visceral fat accumulation therefore compounds insulin resistance through both a lipid substrate mechanism and an inflammatory signaling mechanism simultaneously.
Mechanism 3: Disruption of Sleep Architecture and Circadian Metabolic Regulation
Cortisol follows a precise diurnal rhythm in a healthy HPA axis: rising sharply in the 30–45 minutes after waking — the cortisol awakening response — then declining gradually through the day to reach its nadir in the late evening, allowing the transition to sleep and the nocturnal recovery processes that restore insulin sensitivity, clear inflammatory debris, and support mitochondrial repair. When cortisol is chronically dysregulated — flattened in the morning, elevated in the evening, spiking nocturnally — this entire architecture is disrupted.
Evening cortisol elevation delays sleep onset and suppresses slow-wave sleep, which is the primary phase during which growth hormone is secreted and skeletal muscle insulin sensitivity is restored. Nocturnal spikes produce the characteristic 2–3 AM waking that patients describe — a cortisol-driven arousal at the point in the night when the axis should be in its deepest suppression. As described in the sleep and metabolic health post, even short-term sleep disruption produces measurable insulin resistance independent of diet — meaning that cortisol-driven sleep disruption creates a third independent pathway through which HPA dysregulation worsens the metabolic condition driving it.
The bidirectional relationship between sleep disruption and insulin resistance — and how each worsens the cortisol loop — is detailed in how sleep deprivation accelerates metabolic dysfunction.
Mechanism 4: Suppression of the Reproductive and Thyroid Axes
Chronically elevated cortisol suppresses GnRH pulsatility at the hypothalamic level, reducing LH and FSH release from the pituitary and impairing ovarian and testicular function downstream. In women, this manifests as irregular cycles, weakened luteal phases, and reduced progesterone — contributing directly to the relative estrogen dominance picture described in the estrogen dominance post. In men, it reduces testosterone production, compounding the metabolic syndrome-driven testosterone suppression discussed in the low testosterone post.
Cortisol also inhibits the conversion of T4 to active T3 at the peripheral level and can suppress TSH centrally — producing a functional hypothyroid picture with normal thyroid panel results that reflects impaired thyroid hormone activation rather than primary thyroid pathology. These reproductive and thyroid effects mean that unaddressed cortisol dysregulation can simultaneously drive or worsen PCOS, low testosterone, menstrual irregularity, and hypothyroid symptoms — all through a single upstream mechanism.
The Lifestyle and Dietary Drivers: What Is Actually Producing the Pattern
The drivers of cortisol dysregulation in the patients who present to this practice are rarely dramatic. They are chronic, low-level mismatches between biology and a lifestyle that the modern environment has made entirely normal.
The most consistent pattern involves late eating — particularly refined carbohydrates and ultra-processed foods consumed in the evening — which produces unstable overnight glucose and requires cortisol to manage the resulting substrate gaps through the night. Chronic hyperinsulinemia from continuous carbohydrate loading across the day creates the energy instability that forces cortisol into a compensatory role even before evening. Blue light exposure and cognitive overstimulation at night delay the circadian cortisol decline that should begin in the late afternoon — keeping the system in a state of alertness it cannot easily exit.
Mental load carried into the evening with no genuine neural downregulation prevents the parasympathetic transition that normal cortisol suppression requires. Irregular eating patterns — particularly the combination of under-eating during the day and overeating at night — produce a chronic mismatch between biological timing and nutrient delivery that disrupts both insulin signaling and the cortisol rhythm simultaneously.
In high-performing professionals, this pattern takes a specific form that is worth identifying precisely, because it presents as discipline rather than dysfunction. These patients eat frequently — often following advice to eat every two to three hours to “keep metabolism high” — which maintains elevated insulin throughout the entire day and prevents the between-meal insulin valleys that would allow energy stability and cortisol normalization.
They exercise consistently but often train at high intensity while chronically sleep-deprived, adding a significant physiological stressor to a system already running in compensation mode. They use caffeine to override fatigue signals that are the body’s attempt to communicate a recovery requirement. And they consistently prioritize work over sleep and recovery, treating rest as a variable rather than a non-negotiable biological input.
The result is a person who is, by conventional health metrics, doing many things correctly — and who is simultaneously metabolically unstable, chronically cortisol-elevated, and progressively accumulating the visceral fat and insulin resistance that their lifestyle appears to argue against. The problem is not the quality of the effort. It is that the effort is organized around productivity rather than around the physiological recovery that metabolic function requires.
When Cortisol Becomes the Rate-Limiting Factor
In most patients, cortisol dysregulation is a downstream consequence of insulin resistance and metabolic instability. Address the metabolic root, and the cortisol pattern normalizes as a consequence. But there is a specific patient profile in which cortisol becomes the primary rate-limiting factor in metabolic recovery — and failing to recognize it explains a significant proportion of cases where a technically correct protocol produces inadequate results.
These are patients who are eating correctly — animal-based protein at adequate intake, carbohydrates appropriately restricted, ultra-processed foods eliminated. They are fasting correctly — irregular intermittent fasting, not rigid daily patterns that cause metabolic adaptation. They are training consistently. Their dietary compliance is genuine and documented by improving fasting insulin and TG/HDL numbers. And yet: fat loss has stalled, energy remains unstable, sleep is still disrupted, and the overall sense of metabolic momentum is absent.
In these cases, the limiting factor is almost always the recovery side of the system. Sleep timing is inconsistent. Cognitive stress load remains chronically high. The nervous system is maintained in a state of sustained activation with insufficient parasympathetic recovery between bouts. And in this environment, the body does not perceive a safe enough condition to downregulate cortisol and shift into the recovery and fat mobilization mode that metabolic progress requires.
This is not metaphor. Cortisol suppresses lipolysis — fat release from adipose tissue — and maintains a hormonal environment oriented toward fuel conservation and glucose availability rather than fat oxidation. A patient whose cortisol remains chronically elevated, regardless of the quality of their dietary protocol, is running a hormonal signal that directly counteracts the fat loss and metabolic restoration they are attempting to achieve. Once sleep timing, meal timing, nervous system load, and recovery practices are corrected in these patients, progress typically resumes — often quite quickly, reflecting pent-up metabolic readiness that the cortisol environment had been suppressing.
Cortisol is rarely the root cause of metabolic dysfunction. It is very frequently the rate-limiting factor when the recovery dimension of the protocol is underweighted.
The Intervention: What Addresses Cortisol Dysregulation at Its Root
The intervention that corrects cortisol dysregulation is not designed to modulate cortisol directly. It is designed to remove the metabolic instability and lifestyle mismatches that are forcing the HPA axis into chronic compensatory activation.
Stabilize the insulin load first. Eliminating ultra-processed foods, refined carbohydrates, and continuous snacking removes the primary substrate driving the energy instability that recruits cortisol as a compensatory mechanism. Moving toward adequate animal-based protein at 1.6g per kg of ideal body weight and approximately 50 grams of carbohydrates from whole food sources addresses both the insulin load and the substrate quality that determines metabolic stability. Irregular intermittent fasting — creating genuine between-meal intervals that allow insulin to fall — reduces the chronic hyperinsulinemia that forces cortisol to compensate for metabolic instability, without the rigid daily pattern that triggers cortisol elevation through meal anticipation stress.
Restore meal and sleep timing to biological alignment. The circadian rhythm of cortisol is synchronized by light exposure and meal timing. Eating the majority of caloric intake earlier in the day, completing eating well before sleep, and ensuring consistent sleep and wake times aligns the cortisol diurnal rhythm with its biological design. Evening eating — particularly carbohydrate-dense food — disrupts overnight glucose stability and drives the nocturnal cortisol spikes that produce the characteristic 2–3 AM waking. This is not primarily a sleep hygiene issue. It is a metabolic timing issue.
Eliminate evening neurological stimulation. Blue light exposure suppresses melatonin and delays the cortisol decline that should begin in the late afternoon. Cognitive work, screen exposure, and any content that activates the threat-detection circuitry of the brain maintains sympathetic tone into the period when the nervous system requires parasympathetic dominance to allow sleep onset and cortisol suppression. The practical threshold is consistent: screens off at least 90 minutes before sleep, cognitive demands concluded well before that, and a genuine transition period that allows the nervous system to begin the downregulation it requires.
Address the protein and micronutrient gaps that impair stress resilience. Magnesium is required for the enzymatic conversion of cortisol precursors and for the GABA signaling that mediates parasympathetic nervous system tone. Its deficiency is endemic in populations eating ultra-processed diets and is compounded by the increased urinary magnesium loss that chronic stress produces. B vitamins — particularly B5, B6, and B12 — support adrenal function and the methylation pathways required for cortisol metabolism and elimination. Vitamin D receptor signaling modulates HPA axis sensitivity, and the functional target of 50–70 ng/mL is rarely achieved without deliberate D3 supplementation combined with K2.
Protect recovery as a non-negotiable metabolic input. In the high-performing patients for whom cortisol is the rate-limiting factor, this requires a genuine reframing of recovery — not as a reward for completed work, but as the primary biological input without which metabolic restoration cannot proceed. Reducing training intensity during the period of HPA recovery, protecting sleep duration and consistency, and building genuine parasympathetic recovery windows into the daily schedule are not peripheral lifestyle adjustments. They are metabolic interventions addressing the hormonal environment that determines whether fat mobilization, insulin sensitivity restoration, and hormonal balance are physiologically possible.
GP collaboration throughout. Any medication changes — including those affecting cortisol, blood pressure, or sleep — must be made under the supervision of the prescribing physician. The functional medicine protocol addresses the upstream drivers. It does not replace medical management of any condition for which medication has been prescribed.
A Note on Uncertainty
The mechanisms described in this post — cortisol-driven hepatic glucose production, visceral fat accumulation through glucocorticoid receptor activation, disruption of sleep architecture and circadian regulation, and suppression of reproductive and thyroid axes — are individually well-established in the research literature. What is less clearly quantified is the relative contribution of cortisol dysregulation versus primary insulin resistance in any given patient, and the proportion of patients in whom HPA dysregulation represents a primary driver versus a secondary amplifier.
Salivary cortisol testing across multiple time points — morning, midday, evening, and bedtime — provides a more complete picture of diurnal rhythm than single serum measurements, and is appropriate where clinical presentation suggests significant HPA dysregulation. The interpretation of these results, and any decisions about adaptogenic or pharmaceutical support for HPA function, should involve a qualified clinician. The functional medicine framework described here addresses the metabolic and lifestyle drivers of cortisol dysregulation. It does not constitute a diagnosis of any cortisol-related condition, and it does not replace the evaluation and management of primary adrenal or pituitary pathology.
Practical Implications
If you are experiencing persistent fatigue that does not resolve with sleep, energy that crashes in the afternoon and returns in the evening, difficulty falling or staying asleep despite genuine tiredness, strong cravings for carbohydrates or sugar in the evening, and a sense of stress that feels disproportionate to circumstances — and if your cortisol test has come back normal and your symptoms have been attributed to stress, aging, or lifestyle — the upstream metabolic and rhythmic drivers of your HPA dysregulation have not been assessed.
The practical starting point is measuring fasting insulin and calculating your TG/HDL ratio from your existing lipid panel. If fasting insulin is above 10 µIU/mL and TG/HDL is above 2.0, the metabolic instability driving compensatory cortisol activation is identifiable and addressable. From there, the dietary, timing, and recovery interventions described above address the system producing the pattern — not the symptom the pattern is generating.
Your cortisol result is not the problem. It is the consequence. The system driving it is upstream — and that is where the work begins.
For the full picture of how chronic cortisol elevation reroutes every downstream hormone — thyroid, reproductive, and metabolic — see how chronic stress rewires the entire hormonal system.
Cortisol is one node in a larger network — for the integrated clinical framework on the metabolic drivers behind hormonal dysfunction across thyroid, sex hormones, and the stress axis, see the cluster cornerstone.
People Also Ask
What is cortisol dysregulation?
Cortisol dysregulation describes a state in which the diurnal rhythm of cortisol — high in the morning, declining through the day, low at night — is disrupted, inverted, or flattened, producing symptoms without necessarily generating abnormal values on a standard morning cortisol test. It is distinct from Cushing’s syndrome and Addison’s disease, which represent extremes of cortisol excess and deficiency respectively. In the majority of clinical cases, cortisol dysregulation reflects a dysregulated HPA axis rhythm driven by metabolic instability, circadian misalignment, and chronic physiological and psychological stress load.
Can insulin resistance cause cortisol dysregulation?
Yes. Chronic hyperinsulinemia and the resulting metabolic energy instability force the HPA axis into a compensatory role — using cortisol to maintain blood glucose stability during the energy gaps that insulin resistance creates. This compensatory cortisol elevation then feeds back to worsen insulin resistance through hepatic glucose overproduction, visceral fat accumulation, and impaired sleep architecture. In clinical practice, insulin resistance typically precedes and sets the stage for cortisol dysregulation, after which the relationship becomes bidirectional.
Why does my cortisol test come back normal if I have these symptoms?
Because a standard single-point morning cortisol measurement captures only whether cortisol is within a broad population reference range at one moment in time. It does not assess whether the diurnal rhythm is intact, whether evening cortisol is appropriately suppressed, or whether there are pathological nocturnal spikes. A normal test result rules out structural adrenal pathology. It does not confirm that the HPA axis is functioning in a stable, adaptive rhythm.
What are the symptoms of cortisol dysregulation?
The consistent symptom cluster includes persistent daytime fatigue that does not resolve with sleep, energy that is lowest in the morning and rises in the evening, difficulty falling asleep despite tiredness, nocturnal waking (characteristically around 2–3 AM), strong afternoon and evening cravings for carbohydrates or sugar, poor stress tolerance, emotional or stress-driven eating, difficulty losing visceral fat despite dietary effort, brain fog, and reduced resilience to psychological demands.
What lifestyle factors drive cortisol dysregulation?
The most consistent drivers in clinical practice are chronic hyperinsulinemia from continuous carbohydrate loading and snacking, late evening eating of refined carbohydrates producing unstable overnight glucose, blue light and cognitive overstimulation at night delaying the cortisol decline, mental load carried into the evening without genuine nervous system downregulation, and irregular or insufficient sleep. In high-performing professionals, the specific pattern of frequent eating, caffeine overuse, high-intensity training on inadequate recovery, and chronic sleep deprivation driven by work demands produces a metabolically unstable and chronically cortisol-elevated state despite apparent dietary discipline.
Can cortisol dysregulation prevent fat loss even with a correct diet?
Yes — and this is one of the most clinically significant and least recognized mechanisms of stalled metabolic progress. Chronically elevated cortisol suppresses lipolysis and maintains a hormonal environment oriented toward glucose availability and fuel conservation rather than fat oxidation. In patients whose dietary protocol is technically correct but whose sleep, stress load, and recovery remain inadequate, the cortisol environment prevents the metabolic shift that dietary intervention alone cannot achieve. Correcting the recovery dimension — sleep timing, meal timing, and nervous system load — typically restores metabolic progress in these cases.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Björntorp P, Rosmond R. Obesity and cortisol. Nutrition. 2000;16(10):924–936. 🔗 https://pubmed.ncbi.nlm.nih.gov/11054598/
2. Reaven GM. Role of insulin resistance in human disease. Diabetes. 1988;37(12):1595–1607. 🔗 https://pubmed.ncbi.nlm.nih.gov/3056758/
3. Dallman MF, Pecoraro N, Akana SF, et al. Chronic stress and obesity: a new view of “comfort food.” Proceedings of the National Academy of Sciences. 2003;100(20):11696–11701. 🔗 https://pubmed.ncbi.nlm.nih.gov/12975524/
4. Rosmond R, Dallman MF, Björntorp P. Stress-related cortisol secretion in men: relationships with abdominal obesity and endocrine, metabolic and hemodynamic abnormalities. Journal of Clinical Endocrinology & Metabolism. 1998;83(6):1853–1859. 🔗 https://pubmed.ncbi.nlm.nih.gov/9626108/
5. Spiegel K, Leproult R, Van Cauter E. Impact of sleep debt on metabolic and endocrine function. The Lancet. 1999;354(9188):1435–1439. 🔗 https://pubmed.ncbi.nlm.nih.gov/10543671/
6. Adam TC, Epel ES. Stress, eating and the reward system. Physiology & Behavior. 2007;91(4):449–458. 🔗 https://pubmed.ncbi.nlm.nih.gov/17543357/
7. Tsigos C, Chrousos GP. Hypothalamic-pituitary-adrenal axis, neuroendocrine factors and stress. Journal of Psychosomatic Research. 2002;53(4):865–871. 🔗 https://pubmed.ncbi.nlm.nih.gov/12377295/
8. Epel ES, McEwen B, Seeman T, et al. Stress and body shape: stress-induced cortisol secretion is consistently greater among women with central fat. Psychosomatic Medicine. 2000;62(5):623–632. 🔗 https://pubmed.ncbi.nlm.nih.gov/11020091/
9. Leproult R, Copinschi G, Buxton O, Van Cauter E. Sleep loss results in an elevation of cortisol levels the next evening. Sleep. 1997;20(10):865–870. 🔗 https://pubmed.ncbi.nlm.nih.gov/9415946/
10. Chrousos GP. Stress and disorders of the stress system. Nature Reviews Endocrinology. 2009;5(7):374–381. 🔗 https://pubmed.ncbi.nlm.nih.gov/19488073/
11. Stalder T, Kirschbaum C, Kudielka BM, et al. Assessment of the cortisol awakening response: expert consensus guidelines. Psychoneuroendocrinology. 2016;63:414–432. 🔗 https://pubmed.ncbi.nlm.nih.gov/26563344/
12. Reutrakul S, Van Cauter E. Sleep influences on obesity, insulin resistance, and risk of type 2 diabetes. Metabolism. 2018;84:56–66. 🔗 https://pubmed.ncbi.nlm.nih.gov/29510179/
