When type 2 diabetes is diagnosed, the conversation almost always centers on one number: blood sugar. HbA1c. Fasting glucose. The target. The medication. The monitoring plan. What that conversation almost never includes is the organ that has been driving the process for the better part of a decade before the diagnosis was made.
The liver.
Non-alcoholic fatty liver disease does not develop after type 2 diabetes. In the overwhelming majority of cases, it develops before it — silently, measurably, and entirely addressably — while standard screening is looking elsewhere. The clinical evidence is unambiguous: hepatic insulin resistance and fatty liver accumulation precede the rise of fasting glucose and HbA1c by years, sometimes by a decade or more. NAFLD is not a complication of diabetes. It is one of its earliest upstream drivers. And because it is almost never diagnosed, discussed, or addressed in the standard diabetes conversation, the opportunity to intervene at the most reversible stage of the entire trajectory is consistently missed.
Importantly, fatty liver can also occur in people who appear thin or have a normal BMI, because lean fatty liver disease is often driven by insulin resistance, visceral fat, low muscle mass, and impaired hepatic fat export.
This post explains why the liver is the forgotten organ in the diabetes story, what the mechanistic pathway looks like from hepatic fat accumulation to full metabolic disease, and why identifying NAFLD early — before glucose becomes abnormal — is one of the highest-value interventions in modern metabolic medicine.
What you will learn:
Why NAFLD precedes type 2 diabetes by years in the biological sequence | The mechanistic pathway from hepatic fat accumulation to fasting glucose elevation | Why patients with controlled HbA1c can have a quietly deteriorating liver | Which markers actually reflect hepatic metabolic dysfunction before conventional thresholds are crossed | Why the liver is the missing piece in cases where diabetes is not responding as expected
Clinical Perspective: What I See in Practice
In over a decade of clinical practice, I cannot recall a single patient who arrived with a type 2 diabetes or prediabetes diagnosis and had been told anything meaningful about their liver. Not one. If their GP had been looking at the liver — if NAFLD had been identified, interpreted, and acted upon at the point when it first appeared — the diagnosis would almost certainly have been prediabetes rather than full type 2 diabetes. The liver was telling the story years earlier. No one was reading it.
This is not a minor oversight. The consequence of ignoring NAFLD in the diabetes conversation is that the upstream driver of the entire metabolic trajectory remains unaddressed while treatment focuses entirely on the downstream consequence. Patients receive medications that manage blood glucose. Their HbA1c improves, or stabilizes, and the clinical picture appears controlled. What is not measured — what is not even discussed — is the liver.
And in a significant proportion of these patients, the liver picture is quietly deteriorating behind the controlled glucose numbers. Hyperinsulinemia precedes high blood sugar. That is the biological sequence. Fasting insulin is elevated, the liver is accumulating fat, insulin resistance in hepatocytes is deepening — and none of this appears in the HbA1c until compensation finally fails. By then, the window for the most complete reversal has narrowed considerably.
What I explain to patients is the underlying mechanistic pathway in plain terms: what insulin actually does, why the liver becomes resistant to it, what happens when fat accumulates inside hepatocytes, and how that hepatic dysfunction feeds back into the entire metabolic picture. I explain it in the context of the dietary and lifestyle drivers — hyperinsulinemia from chronic carbohydrate load, fructose from HFCS being converted to hepatic fat faster than the liver can export it, eating multiple times a day without allowing the liver any metabolic rest, ultra-processed foods disrupting the gut-liver axis and flooding the liver with inflammatory signals.
The response is consistent. Patients are surprised — not by the complexity, but by the fact that after years of managing their condition, sometimes decades, no one has explained the underlying mechanism to them. That explanation changes everything. When people understand the biology, they make different choices — not from fear or obligation, but from a place of knowledge and genuine understanding of what their body is doing and why.
The pattern I see most persistently in clinical practice is not the dramatic case. It is the ordinary one: a patient with reasonably controlled blood sugar, medications working as intended, HbA1c in an acceptable range — and a liver picture that is telling a completely different story. ALT above the functional concern threshold. GGT trending upward. Triglycerides rising. TG/HDL ratio above 2.0. Fasting insulin, when it has been ordered, elevated well above the functional optimal. The blood sugar management is real. The metabolic dysfunction driving the liver deterioration is also real. They are not in conflict — they simply reflect different points in the same biological sequence. The medication is managing the downstream consequence. The upstream driver is operating unchecked.
The Biological Sequence: NAFLD Comes First
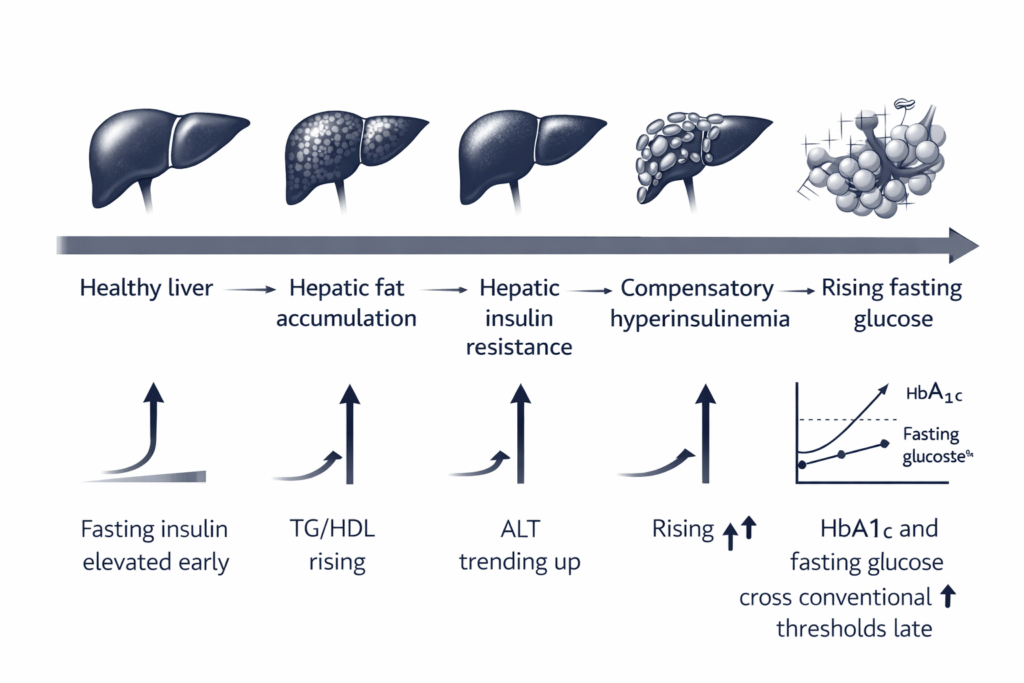
The relationship between NAFLD and type 2 diabetes is not one of complication and cause. It is one of sequence. And the sequence matters enormously for how both conditions are understood and addressed.
Insulin resistance begins in the liver before it becomes clinically apparent in glucose metabolism. The mechanism is specific and well-characterized. As hepatic fat accumulates — driven by chronic free fatty acid spillover from insulin-resistant adipose tissue, by hyperinsulinemia stimulating de novo lipogenesis, and by fructose metabolism generating hepatic triglycerides faster than the liver can export them — diacylglycerol accumulates inside hepatocytes. DAG activates PKCε, which phosphorylates the insulin receptor and directly impairs its kinase activity. The liver becomes resistant to insulin’s signal to suppress glucose production. Hepatic glucose output rises.
This is why the location of body fat matters: visceral fat is more metabolically dangerous than subcutaneous fat because it is inflammatory, insulin-resistant, and closely connected to hepatic fat accumulation.
At this stage, fasting glucose may still appear normal. The pancreas is compensating — secreting additional insulin to suppress the rising hepatic glucose output. Fasting insulin is elevated. HOMA-IR is climbing. The TG/HDL ratio is above 2.0. ALT may be trending above the functional concern threshold. But HbA1c and fasting glucose are still within the conventional normal range, and the standard screening panel produces no intervention signal.
This compensatory phase — established hepatic insulin resistance with maintained near-normal glucose through pancreatic overcompensation — is the window that lasts years, sometimes a decade, before conventional diagnosis. It is the window in which NAFLD is present, measurable, and clinically significant, while the patient is told their results are normal.
As compensation strains and begins to fail — as beta cell functional reserve declines under the chronic demand of hyperinsulinemia, or as peripheral insulin resistance deepens to the point where even elevated insulin cannot maintain glucose control — fasting glucose rises into the impaired range, then HbA1c follows, and the conventional diabetes diagnosis is eventually made. At that point, the liver has typically been accumulating fat and driving hepatic insulin resistance for the better part of a decade. The diagnosis is not the beginning of the metabolic problem. It is the point at which the compensatory system finally runs out of capacity to hide it.
How the Liver Drives Fasting Glucose Elevation
Understanding why the liver is central to the diabetes picture requires understanding one specific and underappreciated feature of hepatic insulin resistance: its disproportionate effect on fasting glucose rather than postprandial glucose.
In a metabolically healthy individual, insulin suppresses hepatic glucose production overnight and between meals — keeping fasting glucose stable and low. This is one of insulin’s most critical functions. When the liver becomes insulin resistant, this suppression fails. Hepatic gluconeogenesis and glycogenolysis continue operating at an elevated rate even in the presence of elevated insulin. The liver produces and releases glucose into the bloodstream through the night and between meals, producing elevated fasting glucose that is disproportionately high relative to what dietary intake alone would predict.
This is why fasting glucose is often the first conventional marker to rise in the trajectory toward type 2 diabetes — and why it rises even in patients who have reduced their carbohydrate intake significantly. The glucose is not primarily coming from what they are eating. It is coming from a liver that has lost its ability to receive and respond to insulin’s suppression signal. Treating that elevated fasting glucose with medications that increase insulin secretion or reduce glucose absorption addresses the downstream number. It does not address the hepatic insulin resistance producing it.
The full mechanistic account of how insulin resistance drives this hepatic dysfunction is covered in detail in the post on how insulin resistance drives fatty liver. What matters at the clinical level is the directional understanding: elevated fasting glucose, in the context of rising triglycerides and borderline liver enzymes, is most commonly a hepatic signal — not a dietary one.
The Dietary Drivers That Fill the Liver With Fat
Three dietary patterns drive hepatic fat accumulation with particular efficiency — and all three operate largely independently of total caloric intake.
Chronic fructose consumption — primarily through high-fructose corn syrup in ultra-processed foods and soft drinks — is metabolized almost exclusively in the liver through a pathway that bypasses the regulatory checkpoints that govern glucose metabolism. Fructose enters the liver through GLUT5 transporters and is phosphorylated by fructokinase without the feedback inhibition that limits glucose phosphorylation by glucokinase. The result is unrestricted substrate delivery to hepatic lipogenic pathways. Fructose generates hepatic triglycerides faster than the liver can export them as VLDL, producing direct intrahepatic fat accumulation independent of insulin signaling.
This export process also depends on adequate choline status, because choline deficiency can impair VLDL export and contribute to hepatic fat accumulation. Regular consumption of HFCS-containing foods and beverages — even in quantities that would be considered moderate by conventional dietary standards — produces measurable increases in hepatic fat content.
Chronic carbohydrate loading without metabolic rest compounds this through a separate mechanism. When carbohydrate intake is continuous across multiple meals and snacks throughout the day, hepatic glycogen stores remain persistently full. A full glycogen store removes the primary regulatory brake on de novo lipogenesis — when there is nowhere to store incoming glucose as glycogen, the liver converts it to fat instead. Eating carbohydrate-dominant foods multiple times a day, as the standard Western dietary pattern recommends, ensures that hepatic lipogenesis runs at an elevated rate continuously.
Refined carbohydrates — stripped of fiber, micronutrients, and the structural complexity that slows their digestion — deliver glucose to the liver rapidly and at high concentration, producing acute insulin spikes that drive SREBP-1c activation and hepatic fat synthesis before the liver has any opportunity to clear what it has already accumulated. The combination of HFCS, refined carbohydrates, and continuous eating creates a hepatic fat accumulation environment that operates regardless of whether total caloric intake appears reasonable.
Why Controlled Blood Sugar Does Not Mean a Healthy Liver
One of the most clinically important patterns in the NAFLD-diabetes relationship is the patient whose glucose management appears successful by conventional standards while the liver picture is quietly progressing.
This pattern arises directly from the biological sequence described above. Medications that improve glycemic control — metformin, GLP-1 receptor agonists, SGLT2 inhibitors — address insulin resistance at specific points in the metabolic cascade. They reduce fasting glucose. They improve HbA1c. In some cases they produce modest weight loss. What they do not necessarily do is resolve the upstream hepatic fat accumulation and hepatic insulin resistance that initiated the sequence. The HbA1c improves because the downstream glucose consequence is being managed. The liver continues accumulating fat because the drivers — hyperinsulinemia, fructose metabolism, gut-liver axis disruption, adipose tissue FFA spillover — remain active.
The marker pattern that reveals this disconnect is consistent: controlled or improving HbA1c alongside rising or persistently elevated ALT, GGT above the functional optimal, triglycerides remaining elevated or rising, and TG/HDL ratio above 2.0. When fasting insulin is measured — which it almost never is in standard diabetes management — it remains elevated despite apparently controlled glucose, confirming that compensatory hyperinsulinemia is still operating and that the upstream insulin resistance has not been resolved.
This is not a failure of medication. It is a consequence of targeting the downstream consequence rather than the upstream driver. Managing blood sugar in the context of NAFLD without addressing the liver is analogous to managing blood pressure without addressing the vascular stiffness causing it. The number improves. The biological process driving it continues.
The Markers That Reveal What Glucose Cannot
Reading the NAFLD-diabetes connection in the lab requires going beyond the markers that standard diabetes management tracks.
ALT above the functional concern threshold of 35 U/L — even when it remains within the conventional normal range of below 56 U/L — is a hepatic metabolic stress signal in this context. In a patient with known diabetes or prediabetes, ALT trending from 25 toward 40 U/L over successive panels is a directional signal of hepatic fat accumulation that warrants investigation, not reassurance.
GGT above 20 U/L in the context of elevated fasting glucose reflects oxidative stress and hepatic metabolic burden that precedes structural damage. The conventional upper limit for GGT sits above 60 U/L — a threshold so permissive that by the time it is crossed, the hepatic metabolic dysfunction it reflects has been operating for years.
The TG/HDL ratio above 2.0 in mg/dL units is the lipid fingerprint of the hepatic insulin resistance and hyperinsulinemia driving both NAFLD and the downstream glucose dysregulation. It is one of the most accessible and most underutilized markers in the diabetes panel — present in almost every standard lipid test, almost never interpreted in the context of hepatic or insulin resistance.
Fasting insulin is the single most important missing marker in almost every diabetes and prediabetes panel. It directly quantifies the degree of compensatory hyperinsulinemia — the primary driver of both hepatic fat accumulation and the entire downstream trajectory toward full metabolic disease. A fasting insulin above 10 µIU/mL in the context of controlled HbA1c is not reassurance. It is confirmation that the upstream driver is still operating and that the biological sequence has not been interrupted, regardless of what the glucose number shows.
HOMA-IR — calculated from fasting insulin and fasting glucose — provides a composite picture of insulin resistance severity. Above 2.0 is significant. Above 2.9 reflects high metabolic and cardiovascular risk. In the context of NAFLD and diabetes, a HOMA-IR above 2.0 with a controlled HbA1c is one of the clearest signals that glucose management is succeeding downstream while metabolic dysfunction continues upstream.
A Note on Uncertainty
The mechanistic relationship between NAFLD and type 2 diabetes is well-established in the research literature. The directional sequence — hepatic fat accumulation and hepatic insulin resistance preceding glucose dysregulation — is supported by longitudinal cohort studies, tracer studies, and mechanistic research. What is less precisely quantified is the proportion of type 2 diabetes cases in which NAFLD is a primary upstream driver versus a concurrent consequence of shared metabolic drivers operating in parallel. The clinical implication is the same in either scenario: NAFLD and type 2 diabetes share upstream drivers that must be addressed together, and addressing glucose alone while leaving the hepatic picture unexamined will produce incomplete and often temporary metabolic improvement.
Next Steps
If you have a type 2 diabetes or prediabetes diagnosis and your liver has never been part of the conversation, the biology described in this post is almost certainly relevant to your case. The question is not whether the liver is involved — in the context of insulin resistance and metabolic syndrome, it almost always is. The question is how far the hepatic picture has progressed and which upstream drivers are most active.
A complete metabolic assessment — integrating fasting insulin, HOMA-IR, liver enzymes, TG/HDL ratio, and the full clinical picture — provides the information needed to identify where in the biological sequence the intervention should begin and how to measure progress at the organ level, not just the glucose level.
Your blood sugar is not the beginning of this story. Your liver probably is. And it has almost certainly been trying to tell you that for years.
People Also Ask
Does NAFLD cause type 2 diabetes?
The relationship is bidirectional, but the biological sequence in most cases places NAFLD upstream of the conventional diabetes diagnosis. Hepatic insulin resistance — driven by intrahepatic fat accumulation — impairs the liver’s ability to suppress glucose production in response to insulin, driving fasting glucose elevation years before HbA1c crosses the conventional diagnostic threshold. NAFLD is both a consequence of the same upstream insulin resistance that drives diabetes and an independent contributor to its progression through hepatic glucose dysregulation.
Can you have fatty liver with normal blood sugar?
Yes — and this is one of the most important clinical realities in metabolic medicine. Fatty liver develops during the compensatory phase of insulin resistance, when the pancreas is secreting excess insulin to maintain near-normal glucose levels. During this phase, which can last a decade or more, NAFLD is present and progressing while fasting glucose and HbA1c remain within conventional normal ranges. This is why fasting insulin and liver enzymes are more sensitive early markers than glucose-based tests for identifying this stage of metabolic dysfunction.
What liver markers should be checked in diabetes?
Beyond the standard ALT and AST, the most clinically informative markers in the NAFLD-diabetes context are GGT — which reflects oxidative stress and hepatic metabolic burden before ALT rises — and the TG/HDL ratio, which reflects the lipid fingerprint of hepatic insulin resistance and hyperinsulinemia. Fasting insulin and HOMA-IR are not liver markers per se, but they are essential for understanding the degree of insulin resistance driving both the hepatic fat accumulation and the glucose dysregulation simultaneously.
Why is my HbA1c controlled but my liver enzymes are still high?
Controlled HbA1c means that the downstream glucose consequence of insulin resistance is being managed — by medication, dietary change, or both. It does not mean that the upstream drivers of hepatic fat accumulation have been resolved. If fasting insulin remains elevated, the TG/HDL ratio remains above 2.0, and dietary patterns continue to include HFCS, refined carbohydrates, and frequent carbohydrate loading, the liver will continue accumulating fat regardless of what the HbA1c shows.
How does fructose cause fatty liver?
Fructose — primarily consumed through high-fructose corn syrup in ultra-processed foods and soft drinks — is metabolized almost exclusively in the liver through a pathway that lacks the regulatory feedback mechanisms that govern glucose metabolism. Fructokinase phosphorylates fructose without inhibition, delivering unrestricted substrate to hepatic lipogenic pathways and generating triglycerides faster than the liver can export them. Regular fructose consumption from HFCS produces direct intrahepatic fat accumulation independent of total caloric intake or insulin signaling status.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
- Targher G, Bertolini L, Rodella S, et al. Nonalcoholic fatty liver disease is independently associated with an increased incidence of cardiovascular events in type 2 diabetic patients. Diabetes Care. 2007;30(8):2119–2121.
- Petersen KF, Dufour S, Befroy D, et al. Reversal of nonalcoholic hepatic steatosis, hepatic insulin resistance, and hyperglycemia by moderate weight reduction in patients with type 2 diabetes. Diabetes. 2005;54(3):603–608.
- Samuel VT, Shulman GI. The pathogenesis of insulin resistance: integrating signaling pathways and substrate flux. Journal of Clinical Investigation. 2016;126(1):12–22.
- Donnelly KL, Smith CI, Schwarzenberg SJ, et al. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. Journal of Clinical Investigation. 2005;115(5):1343–1351.
- Loomba R, Sanyal AJ. The global NAFLD epidemic. Nature Reviews Gastroenterology and Hepatology. 2013;10(11):686–690.
- Fabbrini E, Sullivan S, Klein S. Obesity and nonalcoholic fatty liver disease: biochemical, metabolic, and clinical implications. Hepatology. 2010;51(2):679–689.
- Stanhope KL, Schwarz JM, Keim NL, et al. Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids and decreases insulin sensitivity in overweight/obese humans. Journal of Clinical Investigation. 2009;119(5):1322–1334.
- Jornayvaz FR, Shulman GI. Diacylglycerol activation of protein kinase Cε and hepatic insulin resistance. Cell Metabolism. 2012;15(5):574–584.
- Cani PD, et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes. 2007;56(7):1761–1772.
- Reaven GM. Role of insulin resistance in human disease. Diabetes. 1988;37(12):1595–1607.
