Most patients who develop fatty liver believe it happened because of what they ate. Too much fat. Too many calories. A diet that needed correcting. This is the explanation they have been given, and it is not entirely wrong — but it is so incomplete that it leaves the actual mechanism entirely unaddressed.
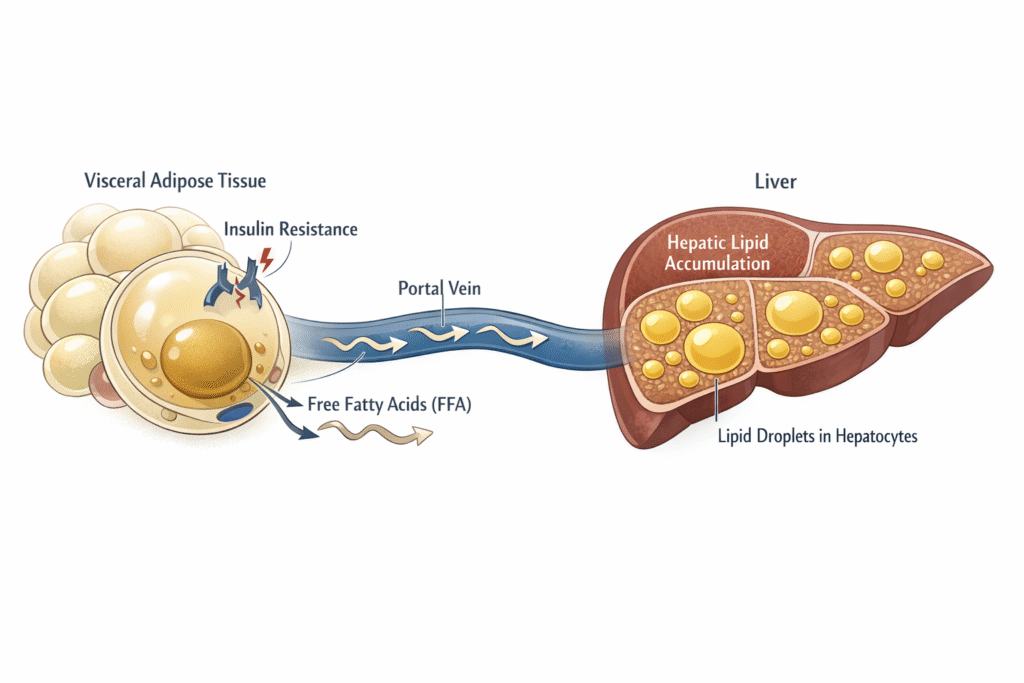
The dominant driver of hepatic fat accumulation in the majority of patients with metabolic dysfunction is not what enters the liver from the diet. It is what enters the liver from the fat tissue — continuously, around the clock, regardless of what is on the plate. When adipose tissue becomes insulin resistant, it loses the ability to regulate the release of free fatty acids into circulation. The result is a relentless, unregulated supply of fat flooding into portal circulation and arriving at the liver faster than it can be processed. The liver accumulates fat not because it is being overfed. It accumulates fat because the fat storage system upstream has broken down.
This mechanism — adipose tissue insulin resistance and the free fatty acid spillover it produces — is the most important and most consistently overlooked driver of fatty liver in metabolic medicine. Understanding it changes what you measure, what you address, and why interventions that focus only on dietary fat reduction consistently produce incomplete results.
What you will learn:
What adipose tissue insulin resistance is and how it develops | Why insulin-resistant fat cells release free fatty acids around the clock regardless of meals | How this FFA spillover floods the liver and drives hepatic fat accumulation | Why this pattern appears in both overweight and normal-weight individuals | Which standard lab markers allow you to infer this process without a free fatty acid test | How quickly the liver picture responds when the upstream insulin load is corrected
Clinical Perspective: What I See in Practice
In over a decade of clinical practice, the pattern that repeats most consistently across patients with fatty liver is this: the liver is not the primary problem, and the diet is not the complete explanation. The fat tissue is the problem. And until that is addressed, neither dietary change nor medication produces the outcome the patient is hoping for.
The patients who arrive with this mechanism active look different from one another on the surface. Most are overweight to some degree — central adiposity is the most common presentation, and visceral fat is almost always part of the picture. But I also see this in patients closer to a normal weight: the TOFI profile — thin outside, fat inside — is becoming increasingly common in my practice, and these patients consistently surprise their GPs. They do not look metabolically unwell.
Their BMI is unremarkable. Their liver enzymes may only be slightly above the functional concern threshold. But fasting insulin tells a different story, and when you calculate the TG/HDL ratio from a standard lipid panel that is almost never interpreted in this context, the picture becomes coherent.
What I rely on in practice to infer adipose tissue insulin resistance and FFA spillover is a combination of three markers: the TG/HDL ratio, fasting insulin, and liver enzymes — specifically ALT and GGT read against functional thresholds, not conventional ones. Free fatty acids are almost never ordered in standard panels, and GPs rarely request them even when the clinical picture strongly suggests this mechanism. So the inference has to be made from the markers that are already present in the panel — and when you read them together, the story is usually clear.
Fasting insulin, critically, is almost never ordered by GPs in Germany and receives almost no attention when it is. This is the single most significant diagnostic gap in this patient population. A fasting insulin of 18 µIU/mL sitting in a panel with no comment, no follow-up, and no clinical action taken — while the TG/HDL ratio sits above 2.5 and GGT is at 34 U/L — is a coherent and urgent metabolic picture that has been systematically overlooked.
The moment that stands out most clearly in patient conversations is when I explain that their fat tissue has become insulin resistant — that it is not just the liver that has a problem, but the fat cells themselves, which are releasing fat into the bloodstream in a way that the body can no longer regulate. The response is almost universally the same: complete surprise. Not a vague awareness that something was wrong. Genuine surprise that this is even possible.
Most patients have never heard that fat cells respond to insulin. They have never been told that their fat tissue has a signaling function, or that when that function breaks down, the liver pays the price first. That explanation — delivered in plain terms, connected directly to their lab values and their symptoms — changes everything about how they engage with the protocol.
Once the upstream insulin load is corrected through dietary quality, carbohydrate reduction, and structured fasting, the liver picture responds. In most patients, meaningful improvement in triglycerides and liver enzymes is visible within 8 to 12 weeks. In patients who apply the protocol with particular consistency and commitment, the response can appear earlier — sometimes within six weeks. The rate of improvement is not linear and is not the same in every patient. What is consistent is the direction: when the FFA spillover is reduced by restoring adipose tissue insulin sensitivity, the liver begins to clear the fat it has accumulated, and the markers move accordingly.
What Adipose Tissue Insulin Resistance Actually Means
To understand why free fatty acid spillover drives fatty liver, the normal function of adipose tissue under insulin signaling needs to be understood first — because it is the failure of that function that produces the problem.
In a metabolically healthy individual, adipose tissue is not passive storage. It is an active endocrine organ that responds to insulin with precise, time-gated regulation of fat release. After a meal, as insulin rises in response to glucose and other macronutrients, it suppresses lipolysis in adipose tissue through a specific mechanism: insulin activates phosphodiesterase 3B, which degrades cyclic AMP, which deactivates protein kinase A, which in turn deactivates hormone-sensitive lipase — the enzyme responsible for cleaving triglycerides stored in fat cells into free fatty acids for release into circulation.
In practical terms, when insulin is elevated, fat cells hold onto their contents. The fat release signal is switched off. This is by design. In the postprandial state, the body is in an energy-receiving mode, and the fat storage system is appropriately suppressed.
Between meals, as insulin falls, the brake is released. Hormone-sensitive lipase becomes active, adipose tissue releases free fatty acids into circulation, and those fatty acids travel to the liver and muscle to be oxidized for energy. This is the normal fasting-state energy supply. It is regulated, proportionate, and time-limited — tied to the actual energy needs of the body and the real nutritional state of the person.
Adipose tissue insulin resistance breaks this regulation at the signaling level. When fat cells become insulin resistant — through the same mechanism that drives insulin resistance in liver and muscle, involving diacylglycerol accumulation, PKC activation, and IRS-1 serine phosphorylation — they lose their sensitivity to insulin’s antilipolytic signal. Hormone-sensitive lipase remains partially or fully active regardless of the insulin level. For the broader liver-centered mechanism, see how insulin resistance drives fatty liver.
Free fatty acid release continues around the clock: in the fed state when it should be suppressed, overnight when the liver should be receiving only a modest fasting-state energy supply, and in the early morning hours when hepatic fat accumulation is most likely to be compounded by the cortisol-driven awakening response.
The result is a qualitatively different fat release pattern from normal physiology. Instead of regulated, meal-gated lipolysis tied to energy need, the liver receives a continuous, unregulated supply of free fatty acids that far exceeds both its oxidative capacity and its export capacity at any given moment.
How FFA Spillover Floods the Liver
The liver receives the products of the portal circulation first — before any other organ does. Everything released from visceral adipose tissue into the portal vein arrives at the liver at high concentration before it is diluted into the systemic circulation. This anatomical position makes the liver uniquely vulnerable to visceral fat dysfunction. When visceral adipose tissue — the fat depot surrounding the abdominal organs — becomes insulin resistant and begins releasing excess free fatty acids, the liver is the first organ downstream.
What happens inside the hepatocyte when the FFA load exceeds normal physiological levels follows a specific and well-characterized sequence. The liver attempts to handle the incoming fatty acids through two primary routes: beta-oxidation, which burns fatty acids for energy, and VLDL synthesis and export, which packages them into triglyceride-rich lipoproteins and ships them into systemic circulation. In a state of modest FFA elevation, these two mechanisms are sufficient. The liver clears what it receives without net fat accumulation.
When the incoming FFA load is chronically elevated — as it is in the context of adipose tissue insulin resistance — the system is overwhelmed on both sides. Beta-oxidation capacity is finite, determined by mitochondrial number and function, and when it is saturated, excess fatty acids accumulate in the cytoplasm. VLDL export capacity is also finite — the liver can only synthesize and secrete VLDL at a limited rate, governed in part by the availability of apolipoprotein B-100 and the packaging machinery inside hepatocytes.
What cannot be burned and cannot be exported is re-esterified into triglycerides and stored inside hepatocytes as lipid droplets. This export pathway also depends on adequate choline status, because choline deficiency can impair VLDL export and make hepatic triglyceride accumulation more likely.
This is the cellular origin of fatty liver in the context of adipose tissue insulin resistance. The hepatocyte is not accumulating fat because it is generating fat from dietary substrate. It is accumulating fat because it is receiving more fat from the circulation than it can clear through its normal exit routes. The source is not the kitchen. The source is the fat tissue upstream.
The further consequence of this intrahepatic fat accumulation is the development of hepatic insulin resistance through the diacylglycerol-PKCε axis. As triglycerides and their metabolic byproducts accumulate inside hepatocytes, diacylglycerol — a potent activator of protein kinase C epsilon — builds up in the hepatic cytoplasm. PKCε phosphorylates the insulin receptor at threonine 1160, directly impairing its kinase activity and blunting the downstream insulin signaling cascade.
The liver becomes insulin resistant as a direct consequence of the fat it has accumulated from the FFA spillover upstream. The cycle is now self-reinforcing: adipose tissue insulin resistance drives FFA spillover, FFA spillover drives hepatic fat accumulation, hepatic fat accumulation drives hepatic insulin resistance, and hepatic insulin resistance drives compensatory hyperinsulinemia, which drives further visceral fat deposition and worsens adipose tissue insulin resistance.
This is also why NAFLD and type 2 diabetes are biologically connected long before glucose becomes diagnostic.
Why This Is Not Just an Overweight Problem
The conventional assumption about fatty liver and free fatty acid spillover is that it is a consequence of obesity — that excess body fat is the source of the excess FFA, and that weight loss is therefore the primary solution. This assumption is partially correct and largely misleading.
It is partially correct because visceral adiposity is the most common clinical context in which adipose tissue insulin resistance develops and FFA spillover becomes clinically significant. Visceral fat — metabolically active, poorly regulated, and in direct anatomical communication with the portal vein — is more prone to developing insulin resistance than subcutaneous fat, and it is the primary source of the portal FFA load that drives hepatic fat accumulation. This is why the distinction between visceral fat and subcutaneous fat matters clinically: the problem is not simply how much fat a person carries, but where that fat is stored and how metabolically active it has become. Patients with central adiposity are at significantly elevated risk for exactly this reason.
But it is misleading because adipose tissue insulin resistance can develop and produce clinically significant FFA spillover in individuals who are not obese, and even in individuals who are lean by conventional BMI standards. The TOFI phenotype — thin outside, fat inside — is the clearest expression of this. In TOFI individuals, the subcutaneous fat compartment is relatively small, giving a normal or near-normal BMI, but visceral fat deposition is disproportionately elevated.
The metabolic consequence of the visceral component is identical to that seen in overtly obese individuals: insulin-resistant visceral adipocytes releasing excess free fatty acids into portal circulation, driving hepatic fat accumulation and the downstream metabolic cascade, while standard screening — focused on BMI and weight — produces no intervention signal.
Athletes and active individuals represent a related and increasingly common variant. Insulin resistance in athletes, driven by ultra-processed sports nutrition products disrupting the gut-liver axis and by dietary patterns that maintain chronically elevated insulin despite high training volumes, can produce adipose tissue insulin resistance in the context of a body composition that appears metabolically healthy from the outside.
The post on insulin resistance in athletes covers this pattern in full. What is relevant here is that FFA spillover is a mechanism tied to adipose tissue insulin signaling failure — not to body weight — and body weight alone is an unreliable proxy for whether this mechanism is active.
The clinical implication is that screening for adipose tissue insulin resistance and FFA spillover cannot rely on BMI, weight, or physical appearance. It requires lab-based assessment of the markers that reflect this process — and specifically the combination of markers that reveal the full picture when read together.
The Parallel Driver: De Novo Lipogenesis
FFA spillover from adipose tissue is the dominant source of hepatic fat in most patients with metabolic fatty liver — accounting for approximately 60% of the intrahepatic triglyceride pool in established NAFLD, according to tracer studies. But a second mechanism operates simultaneously and compounds the hepatic fat burden: de novo lipogenesis, the conversion of carbohydrate substrate into fat inside the liver itself.
De novo lipogenesis is directly driven by insulin through the transcription factor SREBP-1c. In a state of chronic hyperinsulinemia — which is itself a consequence of the compensatory insulin secretion that insulin resistance provokes — SREBP-1c is constitutively activated, and the liver continuously converts incoming carbohydrate substrate into triglycerides. This process operates on top of the FFA spillover arriving from adipose tissue, adding a second fat source that is entirely internal to the liver.
The metabolic paradox at the center of this mechanism — which the post on fatty liver and metabolic dysfunction addresses in the broader organ network context — is that the liver develops selective insulin resistance. It becomes resistant to insulin’s ability to suppress glucose production while remaining fully sensitive to insulin’s fat-producing effects through SREBP-1c. The same hyperinsulinemia that fails to control fasting glucose continues to drive hepatic fat synthesis. This explains why rising fasting glucose and accumulating triglycerides appear simultaneously in the same patient — they are not separate problems. They are two outputs of the same selective signaling failure.
The practical implication for the patient is important: reducing dietary fat intake without addressing the insulin load does not switch off de novo lipogenesis. The driver is not dietary fat. The driver is insulin. And the driver of excess insulin is the chronic carbohydrate load, the continuous eating pattern without metabolic rest, and the ultra-processed food environment that keeps the insulin signal elevated around the clock. Until that upstream signal is corrected, both de novo lipogenesis and adipose tissue FFA spillover continue to load the liver with fat regardless of what the dietary fat intake shows.
The Lab Markers That Reveal What a Free Fatty Acid Test Cannot
Free fatty acids are almost never measured in standard clinical practice. Even in functional medicine, direct FFA measurement is rarely included in a first-line panel. Yet the clinical picture of adipose tissue insulin resistance and FFA spillover is not invisible — it is reflected, with reasonable precision, in a combination of standard markers that are already present in most routine panels. The challenge is that these markers are almost never read together, and individually each one can appear unremarkable while the mechanism is operating at full intensity.
The TG/HDL ratio is the most accessible and most informative proxy for this process. When adipose tissue is releasing excess free fatty acids into portal circulation and the liver is re-esterifying what it cannot oxidize or export, the consequence appears in the lipid panel as elevated triglycerides and suppressed HDL. The liver packages excess fat as VLDL, raising circulating triglycerides. VLDL particles facilitate cholesteryl ester transfer protein-mediated exchange that depletes HDL particles.
The TG/HDL ratio above 2.0 in mg/dL units reflects this dyslipidemia pattern with specificity for the insulin resistance phenotype that is driving it. It is, in effect, the lipid fingerprint of hepatic insulin resistance — and by extension, of the adipose tissue insulin resistance producing the FFA spillover that initiated it. The full interpretation framework for this marker is covered in the post on the TG/HDL ratio.
Fasting insulin is the direct measure of the compensatory hyperinsulinemia that both reflects and worsens adipose tissue insulin resistance. A fasting insulin above the functional optimal of 5 µIU/mL — and especially above 10 µIU/mL — in the context of the TG/HDL pattern described above confirms that the insulin signaling failure is systemic and that the FFA spillover mechanism is almost certainly active. In Germany, fasting insulin is rarely ordered by GPs and receives almost no attention when it appears in a panel. This is the single most consequential diagnostic gap in this patient population, because without fasting insulin, the upstream driver of the entire hepatic fat accumulation process remains invisible to the clinician managing the case.
ALT and GGT, read against functional medicine thresholds rather than conventional reference ranges, provide the hepatic readout. ALT above 35 U/L reflects hepatocyte stress consistent with intrahepatic fat accumulation. GGT above 20–30 U/L reflects the oxidative stress and metabolic burden that the liver is carrying as a result of the FFA overload and the reactive oxygen species generated by excess fat oxidation and de novo lipogenic activity.
The conventional upper limits for both markers — ALT at 56 U/L, GGT at 55–60 U/L — sit so far above the functional concern thresholds that by the time they are crossed, the FFA spillover mechanism has typically been operating for years. The full interpretation framework for these markers is covered in the post on ALT, AST, and GGT.
Waist circumference provides the anthropometric correlate of visceral fat deposition — the specific fat depot that is most prone to developing insulin resistance and producing portal FFA spillover. Above 94 cm in men and 80 cm in women by IDF criteria, visceral adiposity is clinically significant. In the context of elevated TG/HDL ratio and borderline liver enzymes, a waist circumference above these thresholds confirms that the spatial source of the FFA spillover is visceral, not subcutaneous — which has direct implications for the rate and completeness of response to intervention.
Together — TG/HDL ratio, fasting insulin, ALT, GGT, and waist circumference — these markers provide a reliable indirect picture of adipose tissue insulin resistance and FFA spillover without requiring a direct free fatty acid measurement. No single marker is sufficient. The coherence emerges from reading all five in combination, against functional thresholds, in the context of the full clinical picture.
The TOFI Pattern: When the Fat Is Hidden
The TOFI phenotype — Thin Outside, Fat Inside — deserves specific attention because it is the patient profile most likely to be missed entirely by standard screening and most likely to arrive with adipose tissue insulin resistance that has been operating silently for years. This is the same clinical pattern described in lean fatty liver disease, where normal BMI can hide visceral fat, hepatic fat, and early metabolic dysfunction.
In TOFI individuals, the subcutaneous fat compartment — the fat visible under the skin — is relatively small. BMI is normal or borderline. The patient does not look metabolically unwell. There may be modest central fullness that is attributed to age or dietary habits rather than visceral adiposity. Standard screening, which uses BMI as the primary proxy for metabolic risk, produces no intervention signal.
What is not visible is the visceral fat component. Visceral adipocytes — smaller, more metabolically active, and more innervated than subcutaneous adipocytes — develop insulin resistance earlier and more severely than subcutaneous fat cells. When visceral fat reaches a critical threshold of insulin resistance, the regulatory brake on lipolysis fails, and FFA spillover into portal circulation begins — at a body weight that registers as entirely unremarkable by conventional standards.
The lab picture in TOFI patients arriving with this mechanism active is consistent: fasting insulin in the range of 10–15 µIU/mL, reported as normal by the lab and ignored by the GP; TG/HDL ratio above 2.0; GGT between 25 and 40 U/L; ALT borderline above the functional concern threshold; waist circumference elevated relative to BMI. HbA1c may be entirely normal. Fasting glucose may be at the upper end of the functional optimal range. The conventional panel produces no flag. The functional medicine panel tells a clear story.
The increasing prevalence of this phenotype in my practice reflects a broader pattern. The TOFI profile is being driven by the same food environment that is producing metabolic dysfunction in overweight individuals — ultra-processed foods, industrial seed oils, high-fructose corn syrup, continuous eating without metabolic rest — but in individuals whose genetic tendency toward subcutaneous fat storage has masked the visceral component from standard screening. It is not a benign variant of normal metabolic health. It is the same upstream mechanism, with better camouflage.
What This Means for Intervention
Understanding FFA spillover as the primary driver of hepatic fat accumulation has specific and direct implications for what the intervention needs to address and in what order.
Dietary fat restriction, as the primary strategy, addresses the wrong input. The dominant source of hepatic fat in this mechanism is not dietary fat — it is adipose-derived fat arriving continuously from insulin-resistant fat cells. Reducing dietary fat intake modestly reduces the dietary FFA contribution to the hepatic fat pool, but it does not touch the adipose tissue FFA spillover that is the dominant driver. Patients who reduce dietary fat while maintaining a high carbohydrate load and continuous eating pattern will continue to drive hyperinsulinemia, which continues to maintain adipose tissue insulin resistance, which continues to drive FFA spillover. The hepatic fat picture does not significantly improve.
The intervention that addresses the upstream mechanism has two primary components. First, reducing the chronic insulin load — through dietary quality improvement, carbohydrate reduction toward the 50g per day range used in active insulin resistance management, elimination of ultra-processed foods and HFCS, and the removal of continuous snacking patterns that prevent insulin from falling between meals. Lower insulin allows adipose tissue insulin sensitivity to begin recovering, which progressively restores the regulatory brake on lipolysis and reduces the FFA spillover reaching the liver.
Second, creating periods of metabolic rest through structured fasting — 16:8 as an entry point, progressing to 18:6 or occasional 24-hour fasts depending on patient profile and progress — reduces the hepatic fat synthesis signal during the fasting window and allows the liver to shift into a fat-clearing mode rather than a fat-accumulating one. The mechanism through which fasting lowers insulin and allows this metabolic shift is covered in detail in the cluster on insulin resistance. What matters in this context is that fasting does not primarily work by reducing caloric intake. It works by creating the insulin environment in which the fat tissue can restore its regulatory function.
Resistance training provides a third lever that is mechanistically distinct from diet and fasting. Skeletal muscle is the largest insulin-sensitive tissue in the body, and increasing its mass and insulin sensitivity through progressive resistance training reduces the peripheral insulin resistance that is maintaining compensatory hyperinsulinemia. Less hyperinsulinemia means less adipose tissue insulin resistance, which means less FFA spillover, which means less hepatic fat accumulation. The exercise effect on the FFA spillover mechanism operates primarily through this insulin sensitivity restoration — not through the caloric expenditure of the training itself.
A Note on Uncertainty
The proportional contribution of adipose tissue FFA spillover to hepatic fat accumulation — approximately 60% in established NAFLD by tracer study estimates — is a population average. In any individual patient, this proportion varies depending on the relative activity of adipose tissue insulin resistance, de novo lipogenesis, and dietary FFA contribution. In patients with primarily metabolic fatty liver driven by hyperinsulinemia and carbohydrate overconsumption, de novo lipogenesis may contribute more than the average. In patients with predominantly visceral adiposity and relatively controlled carbohydrate intake, FFA spillover likely dominates even more than the average figure suggests.
The clinical implication is not that the mechanism is uncertain — it is well-established — but that the relative contribution of each driver in a given patient must be inferred from the full clinical and lab picture, not assumed from population averages. Treatment prioritization should reflect which mechanism appears dominant in that specific case.
Practical Implications
If your triglycerides are elevated, your TG/HDL ratio is above 2.0, and your liver enzymes are above the functional concern threshold — and no one has connected these findings to a coherent upstream mechanism — the FFA spillover pattern described in this post is almost certainly part of your picture.
The first practical step is to measure fasting insulin if it has not been done. This is not a standard order in most GP consultations, and it must be requested specifically. Without fasting insulin, the degree of compensatory hyperinsulinemia driving both the adipose tissue insulin resistance and the hepatic fat accumulation remains quantitatively invisible. A fasting insulin above 10 µIU/mL in the context described in this post is not a borderline finding. It is confirmation that the upstream mechanism is active and that dietary and lifestyle intervention needs to be directed at insulin load, not at fat intake.
Calculate the TG/HDL ratio from your existing lipid panel if it has not been provided. Divide triglycerides by HDL using both values in mg/dL. Above 2.0 is the functional concern threshold. Above 3.0 reflects significant hepatic insulin resistance and active FFA spillover. This marker is present in every standard lipid panel ordered by every GP. It is almost never interpreted.
Address the dietary drivers of chronic hyperinsulinemia directly and specifically — not through general healthy eating guidance, but through the elimination of the specific inputs that maintain the insulin signal at a level that prevents adipose tissue insulin sensitivity from recovering: ultra-processed foods, industrial seed oils, HFCS, refined carbohydrates, and continuous eating without meaningful fasting windows.
The fat flooding your liver is not primarily coming from your plate. It is coming from your fat tissue — and it is doing so because the signaling system that is supposed to regulate its release has broken down. That breakdown is reversible. But it requires addressing the upstream insulin load, not the downstream hepatic consequence.
People Also Ask
What is adipose tissue insulin resistance?
Adipose tissue insulin resistance is a state in which fat cells lose their sensitivity to insulin’s regulatory signal. In normal physiology, elevated insulin suppresses lipolysis — the release of free fatty acids from fat cells — after meals. When fat cells become insulin resistant, this suppression fails, and they release free fatty acids into circulation continuously, regardless of the nutritional state or the insulin level. The liver is the first organ to receive this unregulated fat load through the portal circulation.
What causes free fatty acid spillover to the liver?
Free fatty acid spillover to the liver is caused primarily by insulin resistance in adipose tissue — particularly visceral adipose tissue, which drains directly into the portal vein. When visceral fat cells lose their sensitivity to insulin’s antilipolytic signal, hormone-sensitive lipase remains active around the clock, releasing excess free fatty acids that exceed the liver’s capacity to oxidize or export them. The liver re-esterifies the excess fat as triglycerides and stores it inside hepatocytes, producing hepatic steatosis.
Can you have fatty liver from fat tissue, not diet?
Yes — and this is one of the most important and least communicated facts about fatty liver pathogenesis. Tracer studies have consistently demonstrated that approximately 60% of the fat stored in the liver in established NAFLD derives from the circulation — primarily from adipose tissue lipolysis — rather than from dietary fat or hepatic de novo lipogenesis. Reducing dietary fat intake without addressing adipose tissue insulin resistance leaves the dominant driver of hepatic fat accumulation entirely untouched.
How do I know if my fatty liver is driven by FFA spillover?
Direct measurement of free fatty acids is rarely performed in standard clinical practice. The combination of markers that reliably infers this mechanism includes: a TG/HDL ratio above 2.0 in mg/dL units, fasting insulin above 10 µIU/mL, ALT above 35 U/L, GGT above 20–30 U/L, and central adiposity with waist circumference above 94 cm in men or 80 cm in women. No single marker is sufficient. The pattern, read across all five in combination, provides a reliable clinical picture of active FFA spillover and adipose tissue insulin resistance.
What is the TOFI phenotype and how does it relate to fatty liver?
TOFI — Thin Outside, Fat Inside — describes individuals with a normal or near-normal BMI who carry disproportionate visceral fat relative to their subcutaneous fat. Because visceral adipocytes are more metabolically active and develop insulin resistance more readily than subcutaneous fat cells, TOFI individuals can develop significant adipose tissue insulin resistance and FFA spillover at a body weight that standard screening registers as unremarkable. They represent an increasingly common clinical profile in which fatty liver and early metabolic dysfunction exist beneath a BMI that produces no intervention signal.
How long does it take for the liver to improve once FFA spillover is reduced?
In most patients, meaningful improvement in triglycerides and liver enzymes — ALT and GGT — is visible within 8 to 12 weeks of consistently reducing the insulin load through dietary quality improvement, carbohydrate reduction, and structured fasting. In patients who apply the protocol with particular commitment, the response can appear earlier, sometimes within 6 weeks. Progress is not linear and varies between individuals. What is consistent is the direction: when the upstream insulin load is corrected and adipose tissue insulin sensitivity begins to recover, the liver responds measurably and in proportion to the consistency of the intervention.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Donnelly KL, Smith CI, Schwarzenberg SJ, et al. Sources of fatty acids stored in liver and secreted via lipoproteins in patients with nonalcoholic fatty liver disease. Journal of Clinical Investigation. 2005;115(5):1343–1351. 🔗 https://pubmed.ncbi.nlm.nih.gov/15864352/
2. Fabbrini E, Sullivan S, Klein S. Obesity and nonalcoholic fatty liver disease: biochemical, metabolic, and clinical implications. Hepatology. 2010;51(2):679–689. 🔗 https://pubmed.ncbi.nlm.nih.gov/20041406/
3. Samuel VT, Shulman GI. The pathogenesis of insulin resistance: integrating signaling pathways and substrate flux. Journal of Clinical Investigation. 2016;126(1):12–22. 🔗 https://www.jci.org/articles/view/77812
4. Petersen MC, Shulman GI. Mechanisms of insulin action and insulin resistance. Physiological Reviews. 2018;98(4):2133–2223. 🔗 https://pubmed.ncbi.nlm.nih.gov/30067154/
5. Jornayvaz FR, Shulman GI. Diacylglycerol activation of protein kinase Cε and hepatic insulin resistance. Cell Metabolism. 2012;15(5):574–584. 🔗 https://pubmed.ncbi.nlm.nih.gov/22560219/
6. Boden G. Obesity and free fatty acids. Endocrinology and Metabolism Clinics of North America. 2008;37(3):635–646. 🔗 https://pubmed.ncbi.nlm.nih.gov/18775354/
7. Kotronen A, Yki-Järvinen H. Fatty liver: a novel component of the metabolic syndrome. Arteriosclerosis, Thrombosis, and Vascular Biology. 2008;28(1):27–38. 🔗 https://pubmed.ncbi.nlm.nih.gov/17690317/
8. Karpe F, Dickmann JR, Frayn KN. Fatty acids, obesity, and insulin resistance: time for a reevaluation. Diabetes. 2011;60(10):2441–2449. 🔗 https://pubmed.ncbi.nlm.nih.gov/21948998/
9. Stanhope KL, Schwarz JM, Keim NL, et al. Consuming fructose-sweetened, not glucose-sweetened, beverages increases visceral adiposity and lipids and decreases insulin sensitivity in overweight/obese humans. Journal of Clinical Investigation. 2009;119(5):1322–1334. 🔗 https://pubmed.ncbi.nlm.nih.gov/19381015/
10. Fabbrini E, Magkos F, Mohammed BS, et al. Intrahepatic fat, not visceral fat, is linked with metabolic complications of obesity. Proceedings of the National Academy of Sciences. 2009;106(36):15430–15435. 🔗 https://pubmed.ncbi.nlm.nih.gov/19706383/
