Visceral fat vs subcutaneous fat is a distinction most patients have never heard — yet it is one of the most consequential in metabolic medicine. Where fat is stored determines how metabolically dangerous it is, far more than how much you carry. Two patients can have the same BMI, the same body weight, and a completely different metabolic reality — because the location of their fat is not the same.
Two patients. Similar body weight. Similar BMI. One has a triglyceride level of 280 mg/dL, a TG/HDL ratio above 3.0, a fasting insulin of 28 µIU/mL, and an ALT trending upward toward the functional concern threshold. The other has a triglyceride level of 95 mg/dL, a TG/HDL ratio below 1.5, and a fasting insulin that sits comfortably below 7 µIU/mL. Their weights are within five kilograms of each other. Their BMIs are both in the overweight range. Standard screening treats them as equivalent metabolic risk.
They are not. And the reason they are not has nothing to do with how much fat they carry. It has everything to do with where that fat is stored.
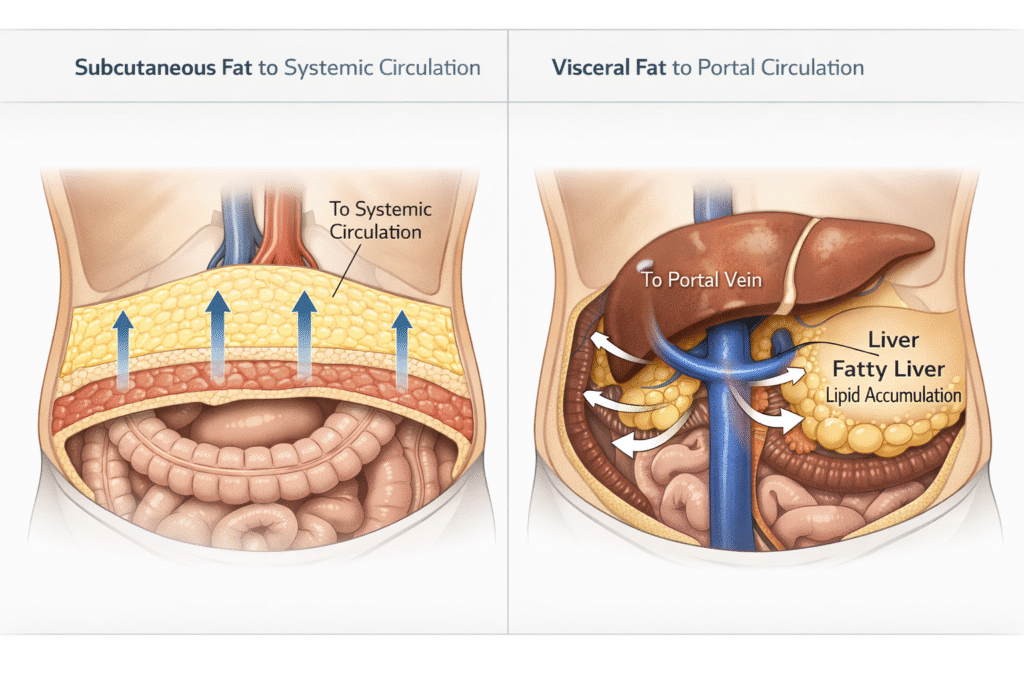
The distinction between visceral fat and subcutaneous fat is not a matter of aesthetics or body shape preference. It is a matter of organ-level biology. Visceral fat — the fat stored around and between the abdominal organs, draining directly into the portal vein — behaves fundamentally differently from subcutaneous fat, which sits beneath the skin and is metabolically far more inert. Visceral fat is an active endocrine organ that secretes inflammatory signals, releases free fatty acids directly to the liver, and drives insulin resistance through mechanisms that standard screening is not designed to detect. Subcutaneous fat, in sufficient quantities, can contribute to metabolic dysfunction — but it does so slowly and indirectly, through a completely different biological pathway.
Understanding this distinction changes what you measure, what you treat, and why the scale and the BMI are so consistently misleading as proxies for metabolic health. It also explains one of the most common and most consequential patterns in modern metabolic dysfunction: the patient who does not look metabolically unwell but is, because everything dangerous is on the inside.
What you will learn:
The biological differences between visceral and subcutaneous fat at the cellular and organ level | Why visceral fat drives insulin resistance, fatty liver, and systemic inflammation through mechanisms that subcutaneous fat does not | Why BMI and body weight fail as metabolic risk markers and what actually reflects the visceral fat burden | The clinical presentation and lab picture that consistently signals visceral adiposity as the dominant driver | Why the TOFI pattern is rising and what it looks like in practice | Which interventions move visceral fat most effectively and in what order
Visceral Fat vs Subcutaneous Fat: Clinical Perspective and What I See in Practice
The pattern that repeats most consistently across patients with significant visceral adiposity — regardless of whether they are overtly overweight or carrying a relatively unremarkable body weight — is a specific cluster of symptoms and markers that tells the same story in different bodies.
Fatigue is almost universal, and it has a particular quality: not tiredness from exertion, but a low-grade, persistent heaviness that is worst in the afternoon and does not fully resolve with sleep. Brain fog is the second most consistent feature — difficulty concentrating, a sense of cognitive dulling that patients have often normalized over years, attributing it to stress or age, not recognizing it as a metabolic symptom. Abdominal fullness is present even in patients who are not dramatically overweight — a tightness or heaviness in the upper abdomen that reflects not just fat volume but the inflammatory and hormonal signaling that visceral fat generates.
Blood pressure creeping above optimal. Sleep quality declining despite adequate hours. These are not five separate problems. They are one problem expressed across multiple organ systems — and the driving mechanism in the majority of these patients is visceral fat that standard screening is not designed to find.
The lab picture that accompanies this symptom cluster is consistent enough that I can often anticipate it before opening the results. Triglycerides elevated — commonly between 180 and 310 mg/dL on arrival, as seen in several documented cases in this practice. TG/HDL ratio above 2.0, frequently above 3.0. ALT above the functional concern threshold of 35 U/L. GGT trending upward. Fasting insulin — when it has been ordered at all, which is rare — sitting between 18 and 28 µIU/mL. In the documented cases from this practice: one patient arrived with triglycerides at 280 mg/dL and HbA1c at 10.5%; another with triglycerides at 310 mg/dL and ALT at 78 U/L; a third with fasting insulin at 28 µIU/mL.
These are not outliers. They represent the predictable lab consequence of visceral fat that has been metabolically active and unaddressed for years, producing FFA spillover into portal circulation, driving hepatic fat accumulation, and maintaining the compensatory hyperinsulinemia that standard panels consistently fail to flag.
When I explain to patients that the location of their fat — not just the quantity — is what determines how metabolically dangerous it is, the reaction is almost invariably the same: complete surprise. Not vague uncertainty, but genuine surprise that this distinction exists at all. Their GPs have discussed weight. They have discussed BMI. They have discussed total cholesterol.
No one has explained that the fat surrounding their abdominal organs is biologically different from the fat under their skin — that it drains directly into the liver, that it secretes hormones and inflammatory signals that subcutaneous fat does not produce at the same intensity, that this is why their liver enzymes are elevated and their triglycerides will not normalize despite dietary effort that is, by conventional standards, reasonable. That explanation — delivered in the context of their specific lab values — is frequently the first time the metabolic picture coheres for them. And that coherence is what drives genuine engagement with the protocol.
The TOFI presentation — normal or near-normal BMI, normal-looking body, but clinically significant visceral fat and early metabolic dysfunction underneath — is not rare in this practice. It is on a rapid and consistent rise, and it is underestimated at almost every level of standard clinical care. These patients arrive having been told their labs are acceptable. Their weight is fine. Their BMI is in range. The conversation, to the extent there has been one, has focused on total cholesterol and perhaps blood sugar.
What the functional medicine assessment reveals is the pattern described above: a TG/HDL ratio that has never been interpreted, a fasting insulin that has never been ordered, a GGT that has been dismissed as borderline normal, and a waist circumference that, when measured against functional thresholds rather than visual impression, places them firmly in the visceral risk category. The post on normal weight insulin resistance covers the full scope of why BMI fails these patients — and why this phenotype is far more prevalent than standard care recognizes.
The Biological Difference: Two Types of Fat, Two Metabolic Realities
Adipose tissue is not a single uniform tissue. It is a heterogeneous collection of fat depots that differ from one another in their cellular characteristics, their hormonal responses, their secretory profiles, and their anatomical drainage pathways. Of all the distinctions between fat depots, none is more metabolically consequential than the difference between visceral adipose tissue and subcutaneous adipose tissue.
Subcutaneous fat — the fat that sits beneath the skin across the body, most visibly in the thighs, hips, buttocks, and the outer abdominal wall — is the body’s primary long-term energy storage depot. It is relatively inert metabolically compared to visceral fat. Its lipolytic rate is lower. Its response to insulin’s antilipolytic signal is better preserved. Its drainage goes into the systemic circulation rather than the portal vein, meaning that even when it does release free fatty acids, those fatty acids are diluted across the entire circulatory system before reaching the liver, reducing the hepatic fat load per unit of lipolytic activity.
Subcutaneous fat also produces higher levels of adiponectin — the anti-inflammatory, insulin-sensitizing adipokine — relative to visceral fat. In the context of metabolic health, subcutaneous fat is not benign in unlimited quantities, but it is substantially less metabolically disruptive per unit than the visceral depot.
Visceral fat — the fat surrounding the liver, pancreas, intestines, kidneys, and other abdominal organs, including the omental and mesenteric depots — is a different biological entity. Visceral adipocytes are smaller, more numerous per unit volume, more densely innervated by the sympathetic nervous system, and more richly supplied with glucocorticoid receptors. They are more sensitive to catecholamine-stimulated lipolysis and significantly more resistant to insulin’s antilipolytic suppression.
This combination — heightened lipolytic responsiveness and blunted insulin sensitivity — means that visceral fat releases free fatty acids into circulation more readily, more continuously, and with less regulatory control than subcutaneous fat does. The anatomical consequence of this is the portal vein effect: everything that visceral fat releases drains directly into the portal vein and arrives at the liver at high concentration, before it is diluted into systemic circulation.
This anatomical fact — visceral fat draining into the portal vein, subcutaneous fat draining into the systemic circulation — is the single most important structural reason why fat location matters more than fat quantity. A patient with ten kilograms of predominantly visceral fat is delivering a continuous, concentrated FFA load to their liver through a direct anatomical channel. A patient with fifteen kilograms of predominantly subcutaneous fat is delivering a diluted, intermittent FFA supply to the liver via the general circulation. The liver sees a fundamentally different metabolic environment in each case — and the consequences for hepatic fat accumulation, hepatic insulin resistance, and downstream metabolic dysfunction are correspondingly different.
What Visceral Fat Does to the Liver
The liver’s position as the first organ downstream of visceral fat drainage makes it the primary site where the metabolic consequences of visceral adiposity become structural. The mechanism is the same one described in the post on adipose tissue insulin resistance and FFA spillover — but the visceral fat context adds a specific anatomical intensity that subcutaneous fat simply cannot produce.
When visceral adipocytes are insulin resistant and releasing free fatty acids continuously, the liver receives a fat load that exceeds both its oxidative capacity and its export capacity. Beta-oxidation — the process by which the liver burns fatty acids for energy — saturates. VLDL synthesis and secretion — the process by which the liver packages excess fat into triglyceride-rich lipoproteins and exports it into circulation — also has a finite rate. When choline status is inadequate, this VLDL export pathway becomes even more vulnerable. What cannot be burned or exported accumulates inside hepatocytes as triglyceride droplets. This is the direct cellular origin of non-alcoholic fatty liver disease in the visceral adiposity context.
The downstream metabolic consequences of this hepatic fat accumulation are compounding. As intrahepatic diacylglycerol accumulates — a byproduct of the excess FFA flux and triglyceride re-esterification — it activates protein kinase C epsilon, which phosphorylates the insulin receptor and impairs its downstream signaling. The liver develops hepatic insulin resistance: it becomes unable to suppress glucose production in response to insulin, which drives fasting glucose upward, and continues to respond to insulin’s fat-production signal through SREBP-1c activation, which drives de novo lipogenesis and compounds the intrahepatic fat burden further. The full account of this pathway and how it is reflected in liver enzyme interpretation is covered in the post on ALT, AST, and GGT.
The elevated VLDL export that results from the liver’s attempt to manage its excess fat load is what produces the characteristic dyslipidemia of visceral adiposity: elevated triglycerides and suppressed HDL. The liver packages excess fat as VLDL particles, flooding the circulation with triglyceride-rich lipoproteins. These particles facilitate cholesteryl ester transfer protein-mediated exchange that depletes HDL particles.
The result is the lipid fingerprint that the TG/HDL ratio captures: elevated triglycerides, suppressed HDL, and a ratio above 2.0 that reflects both the hepatic insulin resistance driving VLDL overproduction and the compensatory hyperinsulinemia that is maintaining it. This is the lab signal that standard screening has in every lipid panel and almost never interprets in this context.
Please refer to this post to learn more about the mechanisms by which insulin resistance contributes to fatty liver disease.
What Visceral Fat Does to the Whole Body
The liver is the most immediately affected organ downstream of visceral fat, but it is not the only one. Visceral adipose tissue is an active endocrine organ that secretes a specific profile of adipokines — signaling molecules that communicate the metabolic state of the fat depot to the rest of the body — and the visceral profile differs fundamentally from the subcutaneous one in ways that drive systemic insulin resistance and systemic inflammation simultaneously.
Visceral fat produces significantly higher levels of TNF-alpha and IL-6 than subcutaneous fat. These pro-inflammatory cytokines are not contained locally — they enter portal and systemic circulation and activate the NF-κB inflammatory pathway in liver, muscle, and endothelial tissue. TNF-alpha directly impairs insulin receptor signaling through IRS-1 serine phosphorylation, contributing to systemic insulin resistance independently of the FFA spillover mechanism.
IL-6 activates STAT3 signaling in the liver, suppressing insulin receptor substrate expression and compounding hepatic insulin resistance. The systemic inflammation that patients experience as fatigue, brain fog, and declining sleep quality has a specific biological basis: it is the cytokine output of metabolically active visceral fat tissue operating continuously.
Visceral fat also produces substantially less adiponectin than subcutaneous fat. Adiponectin is the primary insulin-sensitizing adipokine — it activates AMPK in liver and muscle, improves mitochondrial function, suppresses hepatic glucose production, and reduces inflammatory signaling. In a state of visceral adiposity, adiponectin levels fall in proportion to the visceral fat burden.
The loss of adiponectin signaling amplifies the insulin resistance that the inflammatory cytokines and FFA spillover are already driving, removing a regulatory signal that would otherwise partially buffer the metabolic consequences of visceral fat accumulation.
Resistin, another adipokine produced predominantly by visceral fat, impairs insulin signaling in muscle and liver. Plasminogen activator inhibitor-1 — a thrombotic factor whose production is elevated in visceral adiposity — contributes to the cardiovascular risk that accompanies the metabolic dysfunction. The endocrine output of visceral fat is not a single pathway. It is a coordinated set of signals, all of which drive the metabolic environment in the same direction: toward insulin resistance, toward hepatic dysfunction, toward systemic inflammation, and toward the cardiovascular and metabolic disease states that represent the downstream consequences of years of exposure to this signaling environment.
The Subcutaneous Fat Paradox: When More is Less Dangerous
One of the most counterintuitive findings in metabolic research — and one that has direct clinical relevance for patients who are focused on total body weight — is that subcutaneous fat, in the right context, can be metabolically protective. This is not an argument for excess body fat. It is an argument for understanding fat distribution as the primary metabolic variable, not fat quantity.
Research comparing individuals matched for total body fat has consistently demonstrated that those with a higher proportion of subcutaneous fat relative to visceral fat have better insulin sensitivity, lower triglycerides, higher HDL, and lower inflammatory marker levels than those with the same total fat mass distributed predominantly viscerally. The subcutaneous depot acts as a metabolic buffer — sequestering free fatty acids away from the portal circulation, storing lipid in a depot that is less lipolytically active and more responsive to insulin’s antilipolytic signal, and maintaining adiponectin production at levels that support systemic insulin sensitivity.
This is why some patients with a higher body weight and predominantly subcutaneous fat distribution have a metabolic panel that looks entirely unremarkable — normal fasting insulin, normal TG/HDL ratio, normal liver enzymes — while patients with a lower body weight and significant visceral fat accumulation present with the full metabolic dysfunction picture. It is also why interventions that reduce total body weight without specifically targeting visceral fat can produce improvement in the scale number without meaningfully improving the metabolic picture. The weight loss matters less than where the loss is coming from.
How to Identify Visceral Fat Burden Without Imaging
Direct visceral fat measurement requires imaging — CT scanning or MRI provides the most precise quantification of visceral fat volume. These tools are not available in routine clinical practice. What is available — in every standard panel, in every GP office, in every routine consultation — is the combination of markers that consistently reflects the visceral fat burden and its metabolic consequences without requiring imaging.
Waist circumference is the most accessible anthropometric proxy. The IDF thresholds — above 94 cm in men, above 80 cm in women — identify the waist circumference at which visceral adiposity becomes clinically significant. Waist-to-height ratio, where a waist circumference exceeding half of height indicates elevated visceral risk, is an alternative that accounts for body frame differences between individuals. Neither measure is perfect, but both are substantially more informative than BMI for the specific purpose of identifying visceral fat burden.
The TG/HDL ratio, calculated by dividing triglycerides by HDL using both values in mg/dL, is the most informative standard lab marker for visceral adiposity and its hepatic consequences. Above 2.0 reflects compensatory hyperinsulinemia and the dyslipidemia pattern of visceral fat-driven hepatic dysfunction. Above 3.0 reflects a more established pattern of hepatic insulin resistance and active FFA spillover. This ratio is present in every standard lipid panel. It is almost never calculated, reported, or interpreted in the context of visceral fat burden.
Fasting insulin, when it has been measured — which it almost never is in standard practice in Germany — is the most direct quantification of the compensatory hyperinsulinemia that visceral fat drives. Above 10 µIU/mL is above the functional optimal. Above 15 µIU/mL reflects significant insulin resistance. The documented cases in this practice show presenting fasting insulin values of 18 and 28 µIU/mL — both sitting in panels where no clinical action was taken, where the GP noted the value and moved on without connecting it to the visceral fat burden, the liver enzyme pattern, or the dyslipidemia present in the same panel.
ALT above 35 U/L and GGT above 20–30 U/L, read against functional medicine thresholds rather than conventional reference ranges, provide the hepatic readout of what visceral fat is doing to the liver. The conventional upper limit for ALT sits at 56 U/L — a threshold so permissive that by the time it is crossed, the hepatic fat accumulation it reflects has been developing for years.
The functional concern threshold of 35 U/L identifies the earlier stage where hepatic metabolic stress is present and addressable. The documented case of a patient arriving with ALT at 78 U/L and triglycerides at 310 mg/dL represents not an early detection story but a consequence of years of unread signals — all of which were present in earlier panels at lower values, crossing functional thresholds long before conventional ones.
Together — waist circumference, TG/HDL ratio, fasting insulin, ALT, and GGT — these markers form a coherent clinical picture of visceral fat burden and its organ-level consequences. None of them requires imaging. All of them are available in standard practice. The gap is not in the data. It is in the interpretation.
The TOFI Pattern: The Hidden Visceral Burden
The TOFI phenotype — Thin Outside, Fat Inside — represents the clearest expression of why fat location matters more than fat quantity, and why standard screening consistently misses the patients who need the most urgent metabolic attention.
TOFI patients have a normal or near-normal BMI. Their subcutaneous fat is modest. Visually, they do not present as metabolically at-risk individuals by conventional clinical assessment. What they carry that standard screening cannot see is a disproportionate visceral fat depot — fat accumulated around and between the abdominal organs, beneath the superficial fat layer that is visible to inspection, detectable only by imaging or by the lab markers that reflect its metabolic activity.
The TOFI pattern is on a rapid rise, and it is being systematically underestimated in clinical practice. The food environment driving it is not producing this pattern selectively in people who are gaining visible weight. Ultra-processed foods, industrial seed oils, HFCS, and continuous eating without metabolic rest produce hyperinsulinemia that drives preferential visceral fat deposition — particularly in individuals with a genetic tendency toward storing excess energy viscerally rather than subcutaneously.
These individuals can maintain a normal body weight while accumulating metabolically dangerous visceral fat over years. Their GPs reassure them. Their BMI is fine. Their weight has not changed. Standard screening produces no intervention signal. This is the same mechanism that explains lean fatty liver disease, where liver fat develops without obvious obesity.
The functional medicine assessment of these patients tells a different story. TG/HDL ratio above 2.0. Fasting insulin, if it has been ordered, above 10 µIU/mL. GGT above the functional concern threshold. ALT borderline or above 35 U/L. Waist circumference above the IDF threshold despite a normal BMI. The picture is coherent and specific. The standard screening panel saw nothing worth acting on. The functional assessment identifies the mechanism, quantifies the severity, and points directly to the intervention that will address it.
What Actually Moves Visceral Fat
Visceral fat is more metabolically responsive to intervention than subcutaneous fat — this is one of the few genuinely encouraging aspects of its biology. The same properties that make it more lipolytically active and more catecholamine-sensitive also make it more responsive to the metabolic changes that interventions produce. When the right interventions are applied in the right order, visceral fat responds first and most dramatically among the fat depots.
The most effective and most consistently impactful first step is the elimination of the specific dietary inputs that maintain chronic hyperinsulinemia: ultra-processed foods, industrial seed oils, HFCS, and refined carbohydrates. This is not a general instruction to eat less. It is a specific instruction to remove the inputs that keep insulin elevated around the clock and that drive preferential visceral fat deposition through SREBP-1c activation and the continuous eating pattern that prevents insulin from falling between meals.
In clinical practice, patients who make this change with genuine consistency — not partial compliance but real elimination — show the most rapid and most significant early improvements in triglycerides, TG/HDL ratio, and ALT. The liver begins clearing the fat it has accumulated because the primary driver of hepatic fat synthesis has been removed.
The second component is significant carbohydrate reduction — targeting approximately 50 grams of carbohydrate per day in patients actively managing insulin resistance. At this level of carbohydrate intake, insulin remains sufficiently low between meals to allow adipose tissue lipolysis to operate in a regulated fashion, visceral fat to be mobilized for oxidation rather than maintained as a storage depot, and hepatic de novo lipogenesis to fall from the chronically elevated rate that refined carbohydrate loading maintains.
Animal-based, leucine-rich protein at 1.6 grams per kilogram of ideal body weight provides the third component — preserving and building lean mass while the dietary pattern creates the hormonal environment for visceral fat reduction. Protein has a significantly lower insulinogenic effect than carbohydrate, supports satiety through GLP-1 and PYY secretion, and provides the substrate for muscle protein synthesis that makes resistance training metabolically productive. The combination of adequate protein and carbohydrate reduction produces a metabolic environment where visceral fat is preferentially mobilized and lean mass is preferentially maintained or increased — the compositional shift that produces durable improvement in the visceral fat burden and its lab markers.
Resistance training is the intervention that accelerates and sustains the visceral fat reduction that dietary change initiates. Skeletal muscle is the primary tissue responsible for insulin-stimulated glucose disposal. Increasing muscle mass and insulin sensitivity through progressive resistance training reduces the peripheral insulin resistance that maintains compensatory hyperinsulinemia — the same hyperinsulinemia that drives visceral fat deposition. The effect of resistance training on visceral fat is not primarily caloric. It is hormonal: lower circulating insulin means less visceral fat deposition and more visceral fat mobilization, independent of the energy balance effect of the training itself.
Walking after meals — particularly after the evening meal when visceral fat deposition is hormonally favored — provides an accessible additional lever: post-meal glucose disposal through muscular activity reduces the postprandial insulin spike that would otherwise drive visceral fat synthesis, without the cortisol-elevating effect of high-intensity exercise that can, in susceptible individuals, partially counteract the intended metabolic benefit.
A Note on Uncertainty
The distinction between visceral and subcutaneous fat as metabolic risk factors is well-established in the mechanistic and epidemiological literature. The directional principle — that visceral fat location produces metabolically disproportionate consequences relative to its quantity, through the portal vein drainage pathway and the inflammatory secretory profile — is not scientifically contested.
What is less precisely quantified in any individual patient is the relative proportion of their total fat mass that is visceral versus subcutaneous, without imaging. The lab markers described above — TG/HDL ratio, fasting insulin, liver enzymes — provide an indirect picture that is clinically reliable but not a direct fat depot measurement. In patients where the indirect picture is ambiguous, or where the clinical decision depends on precise visceral fat quantification, imaging provides the definitive answer.
Practical Implications
If your BMI is within the normal or overweight range and you have been told your metabolic picture is acceptable — but your triglycerides are above 150 mg/dL, your TG/HDL ratio has never been calculated, your fasting insulin has never been ordered, and your waist circumference has never been measured against functional thresholds — the visceral fat picture described in this post has almost certainly never been assessed in your case.
The practical starting point is not imaging and not a new medication. It is interpreting the markers that are already in your standard panel through the lens of visceral fat burden rather than conventional normal ranges. Calculate your TG/HDL ratio. Request fasting insulin. Measure your waist circumference. Apply the functional medicine thresholds — TG/HDL above 2.0, fasting insulin above 10 µIU/mL, ALT above 35 U/L, GGT above 20–30 U/L, waist above 94 cm in men and 80 cm in women — and assess how many of these signals are present simultaneously.
If the pattern is coherent — if multiple markers are above functional thresholds in the same panel — the visceral fat mechanism is almost certainly active, and the intervention needs to address the upstream insulin load rather than the downstream weight number.
The scale tells you how much. The lab tells you where. And where, in metabolic medicine, is the question that matters.
People Also Ask
What is the difference between visceral fat and subcutaneous fat?
Visceral fat is stored around and between the abdominal organs — liver, pancreas, intestines — and drains directly into the portal vein, delivering a concentrated FFA load to the liver. Subcutaneous fat sits beneath the skin across the body, drains into the systemic circulation rather than the portal vein, and is substantially less lipolytically active. Visceral fat produces higher levels of pro-inflammatory cytokines including TNF-alpha and IL-6, lower adiponectin, and drives insulin resistance through both FFA spillover and inflammatory signaling mechanisms that subcutaneous fat does not produce at equivalent intensity.
Why is visceral fat more dangerous than subcutaneous fat?
Visceral fat is more metabolically dangerous because of its anatomical position and its cellular biology. It drains directly into the portal vein, exposing the liver to a concentrated stream of free fatty acids that drives hepatic fat accumulation and hepatic insulin resistance. Its adipocytes are more lipolytically active, more resistant to insulin’s antilipolytic signal, and secrete a pro-inflammatory cytokine profile — including TNF-alpha and IL-6 — that drives systemic insulin resistance independently of the FFA spillover pathway. Subcutaneous fat drains into systemic circulation, is more insulin-sensitive, and produces higher adiponectin levels that partially buffer metabolic dysfunction.
Can you have visceral fat with a normal BMI?
Yes — this is the TOFI phenotype: Thin Outside, Fat Inside. Individuals with a normal or near-normal BMI can accumulate disproportionate visceral fat relative to their subcutaneous fat, producing the full metabolic dysfunction picture — elevated TG/HDL ratio, above-optimal fasting insulin, borderline liver enzymes, rising fasting glucose — without any conventional screening tool producing an intervention signal. BMI measures mass relative to height. It does not measure fat distribution.
How do I know if I have too much visceral fat?
Without imaging — CT or MRI — direct visceral fat measurement is not available in routine clinical practice. The indirect assessment uses the combination of waist circumference above 94 cm in men and 80 cm in women, TG/HDL ratio above 2.0 in mg/dL units, fasting insulin above 10 µIU/mL, and liver enzymes above functional concern thresholds (ALT above 35 U/L, GGT above 20–30 U/L). When multiple markers in this combination are simultaneously above functional thresholds, visceral adiposity is almost certainly the primary driver.
What reduces visceral fat most effectively?
The most effective first intervention is the elimination of the dietary inputs that maintain chronic hyperinsulinemia: ultra-processed foods, industrial seed oils, HFCS, and refined carbohydrates. Significant carbohydrate reduction to approximately 50 grams per day creates the insulin environment in which visceral fat is preferentially mobilized. Animal-based protein at adequate levels preserves lean mass during fat reduction. Resistance training restores peripheral insulin sensitivity, reducing the compensatory hyperinsulinemia that drives visceral fat deposition. These interventions work most effectively in combination and in this sequence — dietary quality and carbohydrate reduction first, then progressive resistance training to sustain and accelerate the improvement.
Why do my triglycerides stay high even when I eat less fat?
Elevated triglycerides in the context of visceral adiposity are driven primarily by hepatic VLDL overproduction — a consequence of the liver receiving excess free fatty acids from visceral fat via the portal vein and packaging the excess as VLDL particles. Reducing dietary fat intake modestly reduces the dietary contribution to this process but does not address the dominant driver: visceral fat-derived FFA spillover. Triglycerides normalize most effectively when the insulin load driving visceral fat insulin resistance is reduced — through carbohydrate restriction, elimination of HFCS and ultra-processed foods, and structured fasting — not when dietary fat is the primary intervention target.
About the Author
Morteza Ariana is a State-Certified Functional Nutritionist based in Germany, specializing in insulin resistance, type 2 diabetes, and root-cause metabolic restoration. He holds advanced training in systems-based physiology and has worked with patients across the U.S. and Europe for over 10 years.
His clinical framework is built around a core principle that mainstream medicine consistently overlooks: chronically elevated insulin — not blood glucose — is the earliest and most actionable driver of metabolic disease. That conviction was shaped in part by his own experience with hyperinsulinemia in 2016, and deepened through a decade of clinical practice and the study of leading researchers in metabolic medicine including Benjamin Bikman, Joseph Kraft, Gerald Reaven, Jason Fung, and Stephen Phinney.
His work focuses on identifying and correcting the upstream metabolic signals — insulin load, liver-gut axis dysfunction, circadian misalignment, and micronutrient gaps — that standard screening misses entirely. Patient outcomes are documented, anonymized, and published on this site.
If this resonates, the next step is clarity
The Metabolic Restoration Blueprint is a structured 12-week framework designed to correct upstream metabolic drivers — not just manage symptoms.
Key References
1. Fontana L, Eagon JC, Trujillo ME, et al. Visceral fat adipokine secretion is associated with systemic inflammation in obese humans. Diabetes. 2007;56(4):1010–1013. 🔗 https://pubmed.ncbi.nlm.nih.gov/17287468/
2. Despres JP, Lemieux I. Abdominal obesity and metabolic syndrome. Nature. 2006;444(7121):881–887. 🔗 https://pubmed.ncbi.nlm.nih.gov/17167477/
3. Fabbrini E, Sullivan S, Klein S. Obesity and nonalcoholic fatty liver disease: biochemical, metabolic, and clinical implications. Hepatology. 2010;51(2):679–689. 🔗 https://pubmed.ncbi.nlm.nih.gov/20041406/
4. Gastaldelli A, Cusi K, Pettiti M, et al. Relationship between hepatic/visceral fat and hepatic insulin resistance in nondiabetic and type 2 diabetic subjects. Gastroenterology. 2007;133(2):496–506. 🔗 https://pubmed.ncbi.nlm.nih.gov/17681172/
5. Pischon T, Boeing H, Hoffmann K, et al. General and abdominal adiposity and risk of death in Europe. New England Journal of Medicine. 2008;359(20):2105–2120. 🔗 https://pubmed.ncbi.nlm.nih.gov/19005195/
6. Tchernof A, Despres JP. Pathophysiology of human visceral obesity: an update. Physiological Reviews. 2013;93(1):359–404. 🔗 https://pubmed.ncbi.nlm.nih.gov/23303913/
7. Jornayvaz FR, Shulman GI. Diacylglycerol activation of protein kinase Cε and hepatic insulin resistance. Cell Metabolism. 2012;15(5):574–584. 🔗 https://pubmed.ncbi.nlm.nih.gov/22560219/
8. Tran TT, Kahn CR. Transplantation of adipose tissue and stem cells: role in metabolism and disease. Nature Reviews Endocrinology. 2010;6(4):195–213. 🔗 https://pubmed.ncbi.nlm.nih.gov/20212495/
9. Ashwell M, Gunn P, Gibson S. Waist-to-height ratio is a better screening tool than waist circumference and BMI for adult cardiometabolic risk factors. Obesity Reviews. 2012;13(3):275–286. 🔗 https://pubmed.ncbi.nlm.nih.gov/22106927/
10. Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006;444(7121):860–867. 🔗 https://pubmed.ncbi.nlm.nih.gov/17167474/
